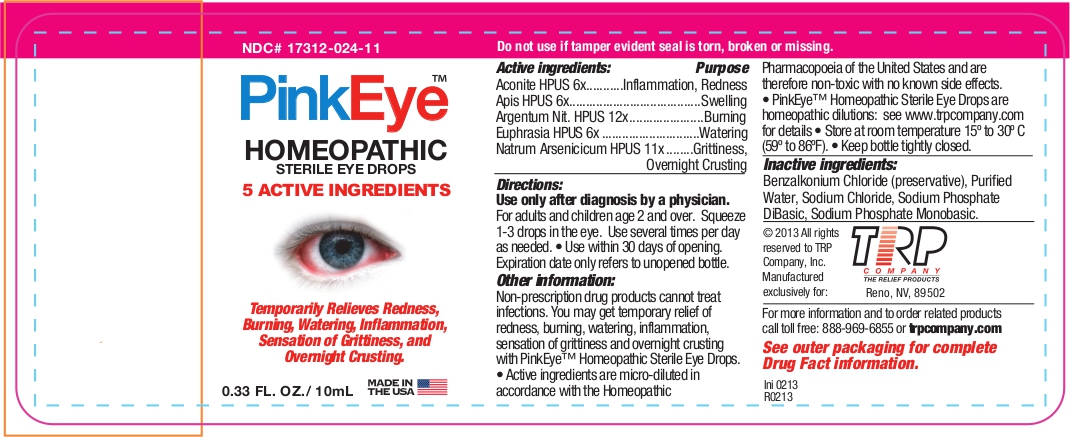
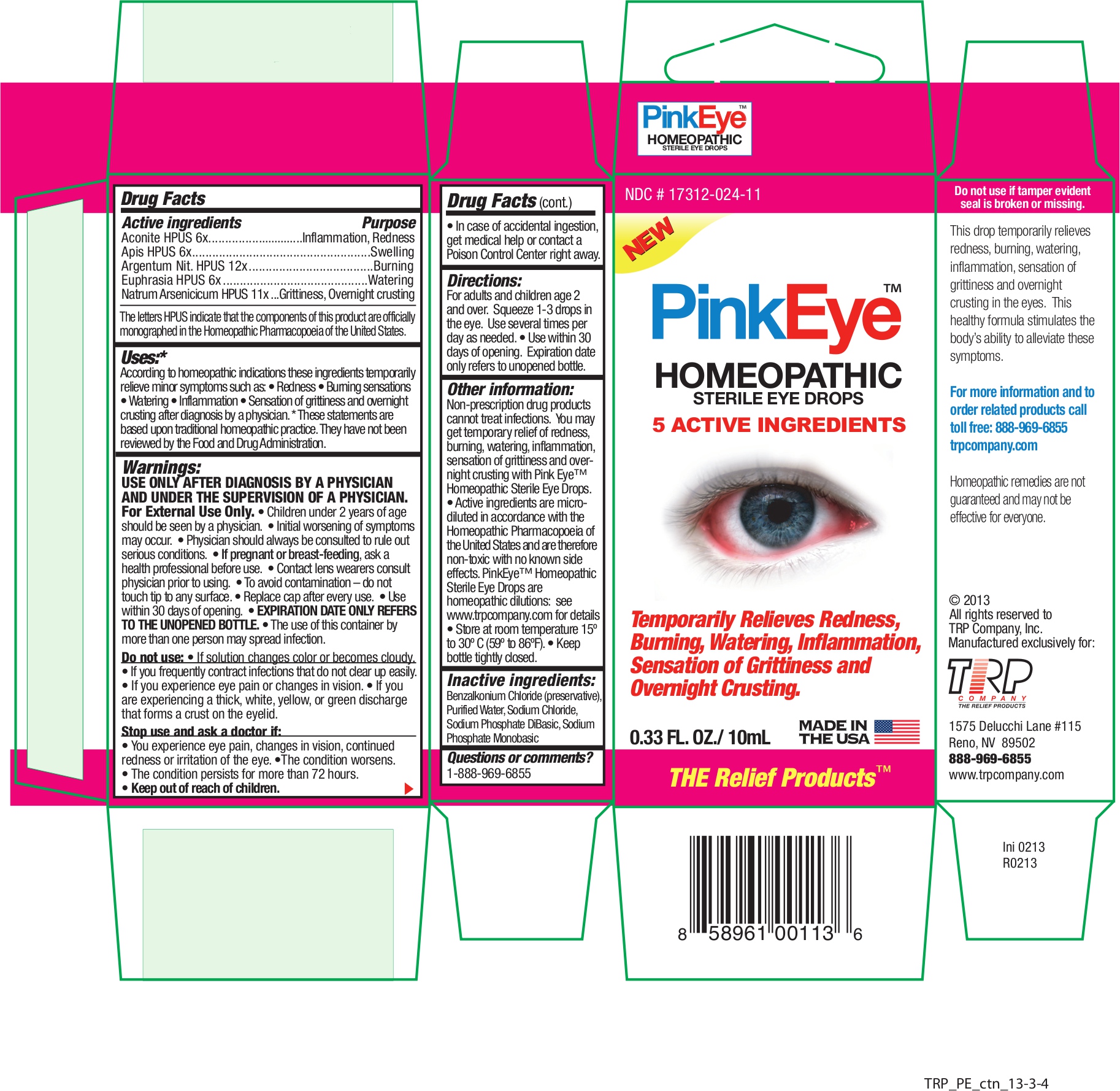
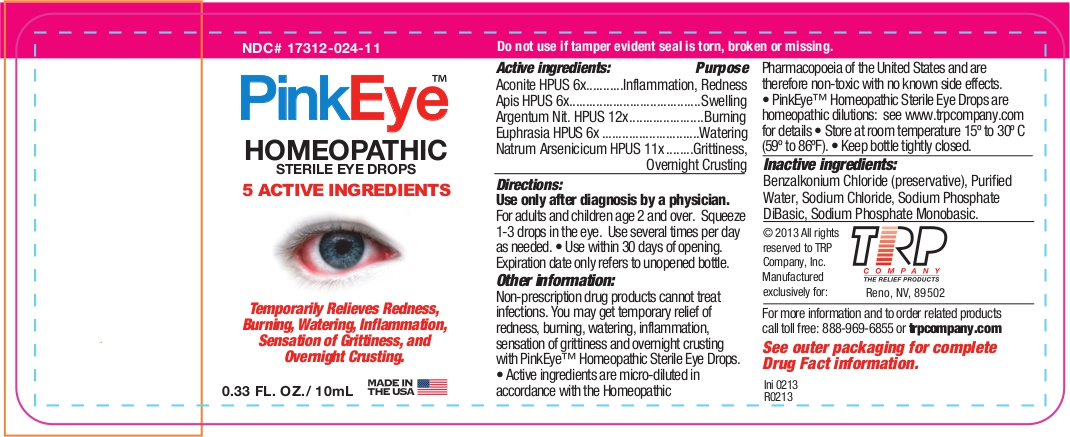
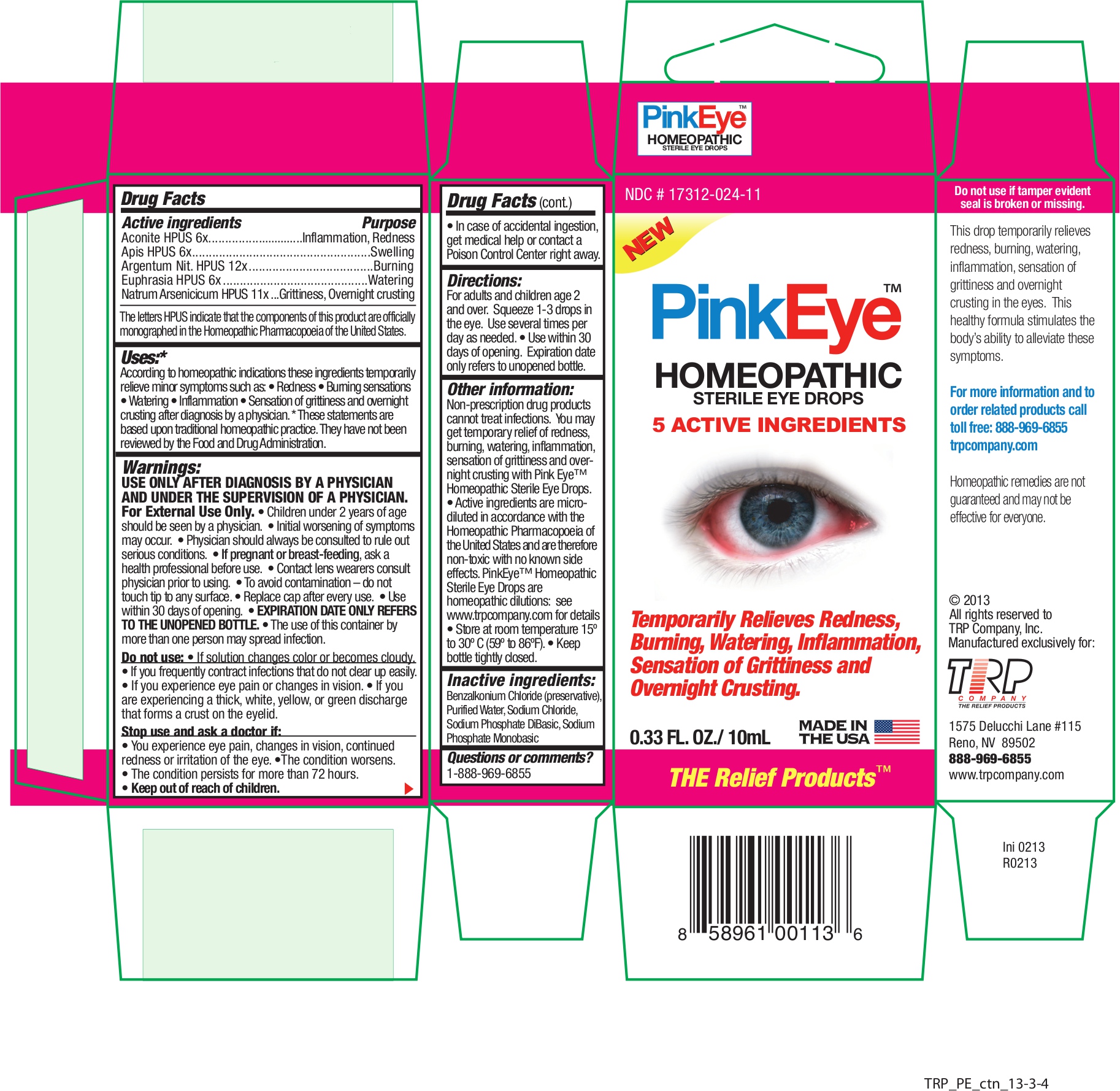
Label: PINK EYE- aconitum napellus - apis mellifera - silver nitrate - euphrasia stricta - sodium arsenate, dibasic, heptahydrate - liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 17312-024-11 - Packager: TRP Company
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated March 4, 2013
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
ACTIVE INGREDIENT
Active Ingredients Purpose Aconite HPUS 6x Inflammation, Redness Apis HPUS 6x Swelling Argentum Nit. HPUS 12x Burning Euphrasia HPUS 6x Watering Natrum Arsenicicum HPUS 11x Grittiness, Overnight crusting The letters HPUS indicate that the components of this product are officially monographed in the Homeopathic Pharmacopoeia of the United States.
- PURPOSE
-
Uses*
According to homeopathic indications these ingredients temporarily relieve minor symptoms such as:
• Redness • Burning sensations • Watering • Inflammation • Sensation of grittiness and overnight crusting after diagnosis by a physician. * These statements are based on traditional homeopathic practice. They have not been reviewed by the Food and Drug Administration. - Warnings
- PREGNANCY OR BREAST FEEDING
- WARNINGS
- Do not use:
- Stop use and ask a doctor if:
- Keep out of reach of children
- Directions
-
Other information
Non-prescription drug products cannot treat infections. You may get temporary relief of redness, burning, watering, inflammation, sensation of grittiness and overnight crusting with Pink Eye™ Relief Homeopathic Sterile Eye Drops.
- Active ingredients are micro-diluted in accordance with the Homeopathic Pharmacopoeia of the United States and are therefore non-toxic with no known side effects. PinkEye™; Homeopathic Sterile Eye Drops are homeopathic dilutions: see www.trpcompany.com for details.
- Store at room temperature 15° to 30° C (59° to 84° F).
- Keep bottle tightly closed
- Inactive Ingredients
- Questions or comments?
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
PINK EYE
aconitum napellus - apis mellifera - silver nitrate - euphrasia stricta - sodium arsenate, dibasic, heptahydrate - liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:17312-024 Route of Administration INTRAOCULAR Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACONITUM NAPELLUS (UNII: U0NQ8555JD) (ACONITUM NAPELLUS - UNII:U0NQ8555JD) ACONITUM NAPELLUS 6 [hp_X] in 1 mL APIS MELLIFERA (UNII: 7S82P3R43Z) (APIS MELLIFERA - UNII:7S82P3R43Z) APIS MELLIFERA 6 [hp_X] in 1 mL SILVER NITRATE (UNII: 95IT3W8JZE) (SILVER CATION - UNII:57N7B0K90A) SILVER NITRATE 12 [hp_X] in 1 mL EUPHRASIA STRICTA (UNII: C9642I91WL) (EUPHRASIA STRICTA - UNII:C9642I91WL) EUPHRASIA STRICTA 6 [hp_X] in 1 mL SODIUM ARSENATE, DIBASIC, HEPTAHYDRATE (UNII: 90ZQ7G407W) (ARSENATE ION - UNII:G1DBI405OK) SODIUM ARSENATE, DIBASIC, HEPTAHYDRATE 11 [hp_X] in 1 mL Inactive Ingredients Ingredient Name Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) WATER (UNII: 059QF0KO0R) SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM PHOSPHATE, DIBASIC (UNII: GR686LBA74) SODIUM PHOSPHATE, MONOBASIC (UNII: 3980JIH2SW) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:17312-024-11 1 in 1 Package 1 10 mL in 1 Bottle, Dropper Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 09/01/2012 Labeler - TRP Company (105185719)