Label: OMISIRGE- omidubicel-onlv kit
-
NDC Code(s):
73441-100-01,
73441-200-01,
73441-300-01,
73441-400-01, view more73441-800-04
- Packager: Gamida Cell Inc.
- Category: CELLULAR THERAPY
- DEA Schedule: None
- Marketing Status: Biologic Licensing Application
Drug Label Information
Updated February 15, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use OMISIRGE safely and effectively. See full prescribing information for OMISIRGE.
OMISIRGE® (omidubicel-onlv) Suspension for Infusion, for Intravenous Use
Initial U.S. Approval: 2023WARNING: INFUSION REACTIONS, GRAFT VERSUS HOST DISEASE, ENGRAFTMENT SYNDROME, and GRAFT FAILURE
See full prescribing information for complete boxed warning.
- Infusion reactions: Infusion reactions may be fatal. Monitor patients during infusion and discontinue for severe reactions. Use is contraindicated in patients with known allergy to dimethyl sulfoxide (DMSO), Dextran 40, gentamicin, human serum albumin, or bovine material. (4, 5.1, 5.2).
- Graft-vs-Host Disease (GvHD): GvHD may be fatal. Administration of immunosuppressive therapy may decrease the risk of GvHD (5.3).
- Engraftment syndrome: Engraftment syndrome may be fatal. Treat engraftment syndrome promptly with corticosteroids (5.4).
- Graft failure: Graft failure may be fatal. Monitor patients for laboratory evidence of hematopoietic recovery (5.5).
INDICATIONS AND USAGE
OMISIRGE is a nicotinamide modified allogeneic hematopoietic progenitor cell therapy derived from cord blood indicated for use in adults and pediatric patients 12 years and older with hematologic malignancies who are planned for umbilical cord blood transplantation following myeloablative conditioning to reduce the time to neutrophil recovery and the incidence of infection. (1).
DOSAGE AND ADMINISTRATION
For intravenous use only.
Do not irradiate.
- Do not use a leukodepleting filter (2).
- Verify patient's identity upon receipt. Do not open the metal cassettes until time of thaw (2).
- Verify patient's identity prior to thaw and prior to infusion (2).
- Thawing should only take place immediately prior to use (2).
- Premedicate the patient approximately 30 to 60 minutes prior to infusion (2.2).
- The CF (Cultured Fraction) bag must be administered FIRST, and infusion should not exceed 2 hours from the end of dilution. Infusion of the NF (Non-cultured) bag should not exceed 1 hour from the end of dilution.
- Administration of OMISIRGE should be under the supervision of a physician experienced in treatment of hematologic malignancies, in centers with expertise in hematopoietic stem cell transplants (2).
DOSAGE FORMS AND STRENGTHS
OMISIRGE is a cell suspension for intravenous infusion (3).
A single dose of OMISIRGE consists of
- a Cultured Fraction (CF): a minimum of 8.0 × 108 total viable cells of which a minimum of 8.7% is CD34+ cells and a minimum of 9.2 × 107 CD34+ cells, and
- a Non-cultured Fraction (NF): a minimum of 4.0 × 108 total viable cells with a minimum of 2.4 × 107 CD3+ cells (3).
CONTRAINDICATIONS
Known sensitivity to dimethyl sulfoxide (DMSO), Dextran 40, gentamicin, human serum albumin or bovine material (4).
WARNINGS AND PRECAUTIONS
- Malignancies of donor origin: Monitor life-long for secondary malignancies. In the event that a secondary malignancy occurs after treatment with OMISIRGE, contact Gamida Cell at (844) 477-7478 (5.6).
- Transmission of serious infections: Monitor patients closely for serious infections (5.7).
- Transmission of rare genetic diseases: Monitor patients for rare genetic diseases (5.8).
ADVERSE REACTIONS
The most common adverse reactions (incidence > 20%) are infections, GvHD, and infusion reaction (6.1).
To report SUSPECTED ADVERSE REACTIONS, contact Gamida Cell at (844) 477-7478 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
USE IN SPECIFIC POPULATIONS
- Pregnancy: No animal or human data. OMISIRGE should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus (8.1).
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 2/2024
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: INFUSION REACTIONS, GRAFT VERSUS HOST DISEASE, ENGRAFTMENT SYNDROME, and GRAFT FAILURE
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Dose
2.2 Administration
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity Reactions
5.2 Infusion Reactions
5.3 Graft-versus-Host Disease
5.4 Engraftment Syndrome
5.5 Graft Failure
5.6 Malignancies of Donor Origin
5.7 Transmission of Serious Infections
5.8 Transmission of Rare Genetic Diseases
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2. Pharmacodynamics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: INFUSION REACTIONS, GRAFT VERSUS HOST DISEASE, ENGRAFTMENT SYNDROME, and GRAFT FAILURE
- Infusion reactions: Infusion reactions may be fatal. Monitor patients during infusion and discontinue for severe reactions. Use is contraindicated in patients with known allergy to dimethyl sulfoxide (DMSO), Dextran 40, gentamicin, human serum albumin, or bovine material [see Contraindications (4), Warnings and Precautions (5.1, 5.2)].
- Graft-vs-Host Disease (GvHD): GvHD may be fatal. Administration of immunosuppressive therapy may decrease the risk of GvHD [see Warnings and Precautions (5.3)].
- Engraftment syndrome: Engraftment syndrome may be fatal. Treat engraftment syndrome promptly with corticosteroids [see Warnings and Precautions (5.4)].
- Graft failure: Graft failure may be fatal. Monitor patients for laboratory evidence of hematopoietic recovery [see Warnings and Precautions (5.5)].
-
1 INDICATIONS AND USAGE
OMISIRGE is a nicotinamide modified allogeneic hematopoietic progenitor cell therapy derived from cord blood indicated for use in adults and pediatric patients 12 years and older with hematologic malignancies who are planned for umbilical cord blood transplantation following myeloablative conditioning to reduce the time to neutrophil recovery and the incidence of infection.
-
2 DOSAGE AND ADMINISTRATION
For intravenous use only.
Do not irradiate.
- Do NOT use a leukodepleting filter.
- Verify patient's identity upon receipt. Do NOT open the metal cassettes until time of thaw.
- Verify patient's identity prior to thaw and prior to infusion.
- Thawing should only take place immediately prior to use.
- Administration of OMISIRGE should be under the supervision of a physician experienced in treatment of hematologic malignancies, in centers with expertise in hematopoietic stem cell transplants.
2.1 Dose
A single dose of OMISIRGE consists of
- a Cultured Fraction (CF): a minimum of 8.0 × 108 total viable cells of which a minimum of 8.7% is CD34+ cells and a minimum of 9.2 × 107 CD34+ cells, and
- a Non-cultured Fraction (NF): a minimum of 4.0 × 108 total viable cells with a minimum of 2.4 × 107 CD3+ cells
The CF and NF are supplied cryopreserved separately in two bags. OMISIRGE requires thaw and dilution with two infusion solution (IS) bags (one IS bag for the CF, and one IS bag for the NF) prior to administration. Infusion of the NF bag should begin within 1 hour after completion of the CF infusion. For timing of dosing of each fraction, refer to section 2.2 under "Planning prior to OMISIRGE preparation".
2.2 Administration
The patient's identity must match the patient-specific identifiers on the CF and NF metal cassettes, the CF and NF bags, and the respective Infusion Solution for CF and NF bags. Do NOT infuse OMISIRGE if the information on the patient-specific labels does not match the intended patient.
Preparing the Patient for OMISIRGE Infusion
- Confirm the Released For Infusion Certificate (RFI Certificate) is available for OMISIRGE before starting the conditioning regimen.
- Administer an appropriate conditioning regimen before infusion of OMISIRGE, according to institutional guidelines.
- Administer prophylactic and supportive therapies for prevention or treatment of transplant complications (GvHD, infections) according to institutional guidelines.
Confirm emergency medications are available prior to infusion and during the recovery period as per institutional guidelines.
Premedication
- Premedicate the patient approximately 30 to 60 minutes prior to OMISIRGE infusion.
- Premedicate with diphenhydramine 50 mg IV (or 0.5 mg/kg up to a maximum of 50 mg) or dexchlorpheniramine 10 mg IV, hydrocortisone 50 mg IV (or 0.5 mg/kg up to a maximum of 50 mg) and acetaminophen 650 mg PO (or 10 mg/kg up to a maximum of 650 mg).
- Avoid prophylactic use of methylprednisolone in conjunction with OMISIRGE.
- Ensure the patient is adequately hydrated.
Receipt of OMISIRGE
OMISIRGE is shipped directly to the transplant center in 2 shipping containers: [see How supplied/ Storage and Handling (16)]
- A liquid nitrogen dry vapor shipper containing the CF, the NF and a Chimerism Testing Sample(s) at ≤ -150°C.
- A refrigerated shipping container containing the Infusion Solution for CF and the Infusion Solution for NF at 2-8°C.
- Confirm that the batch number and patient-specific identifiers on both shipping container labels match the intended patient and the information on the documents from the Gamida Cell Assist Hospital Portal.
- Confirm receipt of the Release for Shipping Certificate. Confirm patient-specific identifiers on the RFI Certificate and Certificates of Analysis (CoAs) match the patient's identity.
- Ensure that OMISIRGE was received in appropriate conditions and confirm that the temperature of the liquid nitrogen dry vapor shipper upon receipt was ≤ -150°C and the temperature of the refrigerated shipping container was 2-8°C.
- If either of the shippers have expired upon arrival, or if you cannot confirm the patient identity with the patient-specific identifiers on any of the labels, contact Gamida Cell at (844) 477-7478.
- You should receive a total of 4 bags (i.e., CF Drug Product [DP] bag, NF DP bag, IS bag for CF DP and IS bag for NF DP) and vial or segment(s) containing Chimerism Testing Sample(s) in the OMISIRGE shipment.
The liquid nitrogen dry vapor shipper contains two metal cassettes, one labeled for the CF containing the CF cryopreserved bag and one labeled for the NF containing the NF cryopreserved bag. The shipper also contains a Chimerism Testing Sample(s).
- Do NOT open the metal cassettes until time of thaw since the product's overwrap bag may inflate preventing cassette closure.
- Verify that the products are within their expiry date by checking the label located on the front of the metal cassettes and through the cassette windows. Do NOT open the cassettes to locate the expiration date.
- Verify that the patient-specific identifiers on the labels on the outside of the CF and NF metal cassettes and on the CF and NF cryopreserved bags visible through the cassette window (see Figure 1) match the intended patient.
- Transfer the metal cassettes containing the CF and NF cryopreserved bags and the Chimerism Testing Sample(s) to onsite vapor phase of liquid nitrogen storage at ≤ -150°C.
Figure 1: CF or NF Cryopreserved Bag inside closed Metal Cassette.
Patient-specific identifiers are visible on the cryopreserved bag through the cassette window. Do NOT open the cassettes.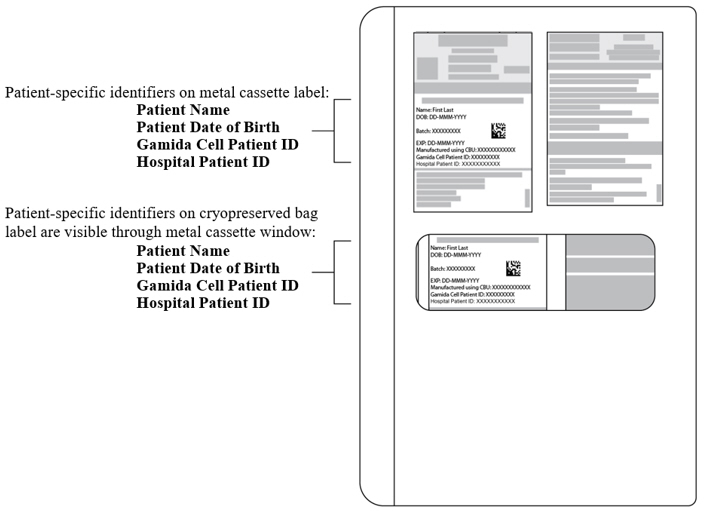
The refrigerated shipping container contains 2 IS bags, the IS for CF and the IS for NF, each with tubing and an attached spike adaptor. Each IS bag is packed inside a sterile bag.
- Ensure that both bags are intact and verify that the Infusion Solutions are within their expiry date by checking the expiration date on the labels located on the IS bags.
- Verify that the patient-specific identifiers on the IS bag labels match the intended patient (see Figure 2).
- Transfer both IS bags to refrigerated storage at 2-8°C.
Figure 2: Infusion Solution for CF Bag with Patient-Specific Label.
The IS bag has tubing with an attached spike adaptor and is packed inside a sterile bag.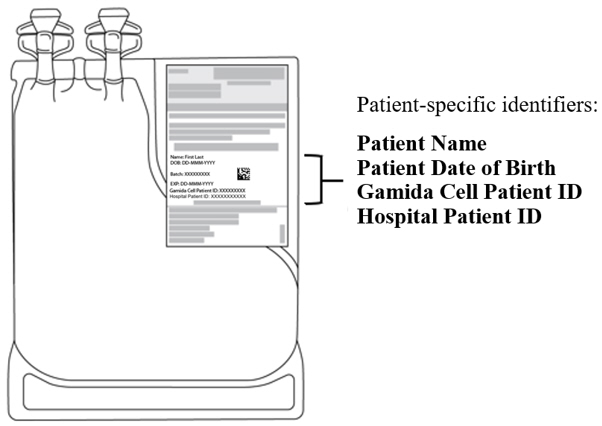
Planning prior to OMISIRGE preparation
- OMISIRGE must not be prepared until after receipt of the RFI Certificate for this patient-specific batch of OMISIRGE. CoAs for the CF, NF and IS batches are attached to the RFI Certificate. The RFI certificate will be issued via Gamida Cell Assist Hospital Portal up to 72 hours after completion of manufacturing.
- Confirm receipt of the RFI Certificate. Confirm patient-specific identifiers on the RFI Certificate and CoAs match the patient's identity.
- The CF bag must be administered FIRST.
- Confirm the infusion time in advance and adjust the start time of CF cryopreserved bag thaw so that it will be available for infusion when the patient is ready.
- Once the CF cryopreserved bag is removed from the metal cassette, thawing and dilution must be carried to completion and the cells administered within 2 hours post-dilution.
- Do not thaw the NF cryopreserved bag until you have determined that the CF has been safely administered.
- Once the NF cryopreserved bag is removed from the metal cassette, thawing and dilution must be carried to completion and the cells administered within 1 hour post-dilution.
- The infusion of the NF bag should begin within 1 hour after completion of the CF infusion.
Preparation of OMISIRGE for Infusion
- Follow universal precautions and local biosafety guidelines for handling and disposal of human cells to avoid potential transmission of infectious diseases.
- Use aseptic technique for all processing steps, including spiking of all transfusion infusion bag ports. No samples should be drawn from OMISIRGE.
The Cultured Fraction
Preparation of the Infusion Solution for CF
- 1.
- Remove the IS for CF bag from the 2-8°C storage location. Remove only the IS for CF bag at this time.
- 2.
- Confirm patient-specific identifiers on the label of the IS for CF match the intended patient.
- 3.
- Wipe the IS for CF sterile bag with 70% alcohol. Place it in the Biological Safety Cabinet (BSC) (if available), for at least 20 minutes with a maximum of 24 hours at room temperature.
- 4.
- Prior to dilution, remove the IS for CF bag from its sterile bag. Check that the pinch clamp is closed.
Thawing and diluting the CF
- 5.
- Remove the CF metal cassette from the liquid nitrogen storage.
- 6.
- Prior to opening the CF metal cassette, verify that the patient-specific identifiers on the label on the outside of the cassette and on the CF cryopreserved bag (visible through the cassette window) match the intended patient (see Figure 1).
- 7.
- Once patient identity has been verified, open the CF metal cassette to remove the CF cryopreserved bag from the cassette. Leave the CF cryopreserved bag in the overwrap bag during thawing and dilution.
- 8.
- Visually inspect the CF cryopreserved bag for damage. If the bag is damaged, contact Gamida Cell at (844) 477-7478. The cryopreserved CF should be white in color.
- 9.
- Once the CF cryopreserved bag is removed from the metal cassette, the thaw and dilution must be carried to completion and the cells administered within 2 hours post-dilution.
- 10.
- Incubate the CF cryopreserved bag for 5 minutes at room temperature.
- 11.
- Place the CF cryopreserved bag in an approximately 37°C water bath until the product reaches a liquid consistency. This generally takes about 3-8 minutes.
Do not massage, knead or apply pressure on the product bag. Keep the bag fully submerged until thawed – do not remove before thawing completion.
- 12.
- Remove the thawed bag from the water bath as soon as the cells have completely thawed. Do not remove the overwrap bag.
- 13.
- Wipe the overwrap with 70% alcohol. Put the bag into the BSC (if available).
- 14.
- Open the overwrap as follows:
- –
- Wipe a pair of clean scissors with 70% alcohol.
- –
- Cut the sealed area at the top of the overwrap.
Be careful not to damage the CF bag or the CF bag's ports/ tubing.
- 15.
- Insert the spike adapter attached to the IS for CF bag into one of the ports of the CF bag, while it remains in the overwrap bag.
- 16.
- Open the pinch clamp on the IS tubing and double the volume of the CF by adding IS for CF (approximately 20 mL) to the CF bag. Gently swirl the bag until mixed well.
- 17.
- Add the remaining IS for CF (approximately 60 mL) to the CF bag. Close the valve and swirl gently.
- 18.
- Remove the overwrap of the CF bag and check the integrity of the CF bag.
- 19.
- Check the appearance of the contents of the CF bag. The thawed and diluted CF should appear as a yellowish suspension, essentially free of visible white clumps and foreign particulates.
- 20.
- Inspect the contents of the thawed and diluted CF bag for any visible cell clumps. If visible cell clumps remain, gently invert and/or massage the bag with fingertips. Small clumps of cellular material should disperse with gentle manual mixing. Do not infuse the CF if clumps are not dispersed, the bag is damaged or leaking, or otherwise appears to be compromised. If this occurs, call Gamida Cell at (844) 477-7478.
- 21.
- Heat seal and detach the emptied Infusion Solution for CF bag.
- 22.
- Connect the Transfusion Infusion Set to the free port on the CF bag. Alternatively, the infusion set may be connected in accordance with internal procedures.
- 23.
- Place the CF bag containing the thawed and diluted CF in a new sterile bag.
Note: Do not wash, spin down, and/or resuspend CF in new media prior to infusion.
- 24.
- Transport the product to the patient at room temperature. Unless prepared at the patient's bedside, transport the product to the bedside in a closed box/bag to protect the product during transport.
The CF bag should be completely infused within 2 hours post-dilution. - 25.
- See the ‘OMISIRGE Administration’ section on how to infuse the CF.
The Non-cultured Fraction
Preparation of the Infusion Solution for NF
- 1.
- Remove the IS for NF bag from the 2-8°C storage location.
- 2.
- Repeat steps 2-4 from the CF process, for the IS for NF.
Thawing and diluting the NF
- 3.
- Repeat steps 5-8 from the CF process, for the NF. The cryopreserved NF should be red in color.
- 4.
- Once the NF cryopreserved bag is removed from the metal cassette, the thaw and dilution must be carried to completion and the cells administered within 1 hour post-dilution.
- 5.
- Repeat steps 10-14 from the CF process, for the NF.
- 6.
- Insert the spike adapter attached to the IS for NF bag into one of the ports of the NF bag, while it remains in the overwrap bag.
- 7.
- Open the pinch clamp on the IS tubing and double the volume of the NF by adding Infusion Solution for NF (approximately 10 mL) to the NF bag. Gently swirl the bag until mixed well.
- 8.
- Add the remaining IS for NF (approximately 30 mL) to the NF bag. Close the valve and swirl gently.
- 9.
- Repeat steps 18-21 from the CF process, for the NF. The thawed and diluted NF should appear as a reddish suspension essentially free of visible clumps and foreign particulates.
- 10.
- Repeat steps 22-25 from the CF process, for the NF. The NF should be completely infused within 1 hour post-dilution.
Do NOT use a leukodepleting filter
- Central venous access is recommended for the infusion of OMISIRGE.
- Confirm that the patient's identity matches the patient-specific identifiers on the CF and NF bags.
- Administer OMISIRGE by gravity infusion.
- Prior to spiking both the CF and NF bags, prime the infusion set tubing with normal saline.
- Infuse the entire contents of the CF and NF bags.
- The rate of infusion should not exceed a maximum of 10 mL per kg per hour.
Administration:
- The thawed and diluted CF bag must be infused FIRST. The infusion time should not exceed 2 hours from the end of dilution to the end of CF infusion. Should an infusion reaction occur, appropriately manage the reaction before thawing the NF.
- The thawed and diluted NF is infused within 1 hour of safely administering the CF infusion. The infusion time should not exceed 1 hour from the end of dilution to the end of infusion.
- In the event of any deviation from the dosing schedule, contact Gamida Cell at (844) 477-7478.
- After the entire contents of the CF and NF bags are each infused, wash the tubing with normal saline at the same infusion rate to ensure as many cells as possible are delivered to the patient.
Follow universal precautions and local biosafety guidelines for handling and disposal of human cells to avoid potential transmission of infectious diseases.
Monitoring
- Monitor the patient for hypersensitivity or other infusion-related reactions during the infusion and post-infusion, per institutional guidelines.
- The infusion rate should be reduced if the fluid load is not tolerated. The infusion should be paused in the event of a hypersensitivity reaction or if the patient develops a moderate to severe infusion reaction. Appropriate medical therapy should be administered as needed. [See Warnings and Precautions (5.1 and 5.2)]
- Monitor for graft failure, GvHD, infections and other post-transplant complications according to institutional guidelines.
-
3 DOSAGE FORMS AND STRENGTHS
OMISIRGE is a cell suspension for intravenous infusion.
A single dose of OMISIRGE consists of:
- a Cultured Fraction: At the time of cryopreservation, the CF contains a minimum of 8.0 × 108 total viable cells with a minimum of 8.7% CD34+ cells and a minimum of 9.2 × 107 CD34+ cells suspended in approximately 10% DMSO.
- a Non-cultured Fraction: At the time of cryopreservation, the NF contains a minimum of 4.0 × 108 total viable cells with a minimum of 2.4 × 107 CD3+ cells suspended in approximately 10% DMSO.
Each fraction is supplied separately in its own cryopreserved bag [see How Supplied/Storage and Handling (16)]. Both bags diluted with their respective IS must be infused to achieve the dose of OMISIRGE.
See the respective CoA for the CF and NF for actual cell counts. The CoAs are attached to the RFI Certificate available via the Gamida Cell Assist Hospital Portal.
Table 1: Appearance of OMISIRGE Product Bags Appearance Immediately before Thawing Appearance Post-Dilution Cultured Fraction White, frozen at the bottom of the cryopreserved bag Yellowish suspension, essentially free of visible white clumps and foreign particulates Non-cultured Fraction Red, frozen at the bottom of the cryopreserved bag Reddish suspension, essentially free of visible clumps and foreign particulates - 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity Reactions
Allergic reactions may occur with the infusion of OMISIRGE. Reactions include bronchospasm, wheezing, angioedema, pruritis and hives. Serious hypersensitivity reactions, including anaphylaxis, may be due to DMSO, residual gentamicin, Dextran 40, human serum albumin (HSA) and bovine material in OMISIRGE.
OMISIRGE may contain residual antibiotics if the cord blood donor was exposed to antibiotics in utero. Patients with a history of allergic reactions to antibiotics should be monitored for allergic reactions following OMISIRGE administration.
5.2 Infusion Reactions
Infusion reactions occurred following OMISIRGE infusion, including hypertension, mucosal inflammation, dysphagia, dyspnea, vomiting and gastrointestinal toxicity. Premedication with antipyretics, histamine antagonists, and corticosteroids may reduce the incidence and intensity of infusion reactions.
In patients transplanted with OMISIRGE in clinical trials, 47% (55/117) patients had an infusion reaction of any severity. Grade 3-4 infusion reactions were reported in 15% (18/117) patients.
Infusion reactions may begin within minutes of the start of infusion of OMISIRGE, although symptoms may continue to intensify and not peak for several hours after the completion of the infusion. Monitor patients for signs and symptoms of infusion reactions during and after OMISIRGE administration. When a reaction occurs, pause the infusion and institute supportive care as needed.
5.3 Graft-versus-Host Disease
Acute and chronic GvHD, including life-threatening and fatal cases, occurred following treatment with OMISIRGE. In patients transplanted with OMISIRGE Grade II-IV acute GvHD was reported in 58% (68/117). Grade III-IV acute GvHD was reported in 17% (20/117). Chronic GvHD occurred in 35% (41/117) of patients. Acute GvHD manifests as maculopapular rash, gastrointestinal symptoms, and elevated bilirubin. Patients treated with OMISIRGE should receive immunosuppressive drugs to decrease the risk of GvHD, be monitored for signs and symptoms of GvHD, and treated if GvHD develops.
5.4 Engraftment Syndrome
Engraftment syndrome may occur because OMISIRGE is derived from umbilical cord blood. Monitor patients for unexplained fever, rash, hypoxemia, weight gain, and pulmonary infiltrates in the peri-engraftment period. Treat with corticosteroids as soon as engraftment syndrome is recognized to ameliorate symptoms. If untreated, engraftment syndrome may progress to multiorgan failure and death.
5.5 Graft Failure
Primary graft failure occurred in 3% (4/117) of patients in OMISIRGE clinical trials. Primary graft failure, which may be fatal, is defined as failure to achieve an absolute neutrophil count greater than 500 per microliter blood by Day 42 after transplantation. Immunologic rejection is the primary cause of graft failure. Patients should be monitored for laboratory evidence of hematopoietic recovery.
5.6 Malignancies of Donor Origin
Two patients treated with OMISIRGE developed post-transplant lymphoproliferative disorder (PTLD) in the second-year post-transplant. PTLD manifests as a lymphoma-like disease favoring non-nodal sites. PTLD is usually fatal if not treated. The etiology is thought to be donor lymphoid cells transformed by Epstein-Barr virus (EBV). Serial monitoring of blood for EBV DNA may be warranted in patients with persistent cytopenias.
One patient treated with OMISIRGE developed a donor-cell derived myelodysplastic syndrome (MDS) during the fourth-year post-transplant. The natural history is presumed to be the same as that for de novo MDS. Monitor life-long for secondary malignancies.
In the event that a secondary malignancy occurs, contact Gamida Cell at (844) 477-7478.
5.7 Transmission of Serious Infections
Transmission of infectious disease may occur because OMISIRGE is derived from umbilical cord blood. Disease may be caused by known or unknown infectious agents. Donors are screened for increased risk of infection with human immunodeficiency virus (HIV), human T-cell lymphotropic virus (HTLV), hepatitis B virus (HBV), hepatitis C virus (HCV), T. pallidum, West Nile Virus (WNV), transmissible spongiform encephalopathy (TSE) agents, vaccinia, and Zika virus (for umbilical cord blood collected since March 2016). Donors are also screened for clinical evidence of sepsis, and communicable disease risks associated with xenotransplantation. Maternal blood samples are tested for HIV types 1 and 2, HTLV types I and II, HBV, HCV, T. pallidum, and WNV. OMISIRGE is tested for sterility. There may be an effect on the reliability of the sterility test results if the cord blood donor was exposed to antibiotics in utero. OMISIRGE is tested for sterility, endotoxin, and mycoplasma. These measures do not totally eliminate the risk of transmitting these or other transmissible infectious diseases and disease agents.
Testing of maternal and infant donor blood is also performed for evidence of donor infection due to cytomegalovirus (CMV).
Test results may be found on the container label and/or in accompanying records.
Product manufacturing includes bovine-derived reagents. While all animal-derived reagents are tested for animal viruses, bacteria, fungi, and mycoplasma before use, these measures do not eliminate the risk of transmitting these or other transmissible infectious diseases and disease agents.
Final sterility test results may not be available at the time of use, but Quality Assurance (QA) will communicate any positive results from sterility testing to the physician. Report the occurrence of transmitted infection to Gamida Cell at (844) 477-7478.
5.8 Transmission of Rare Genetic Diseases
OMISIRGE may transmit rare genetic diseases involving the hematopoietic system because it is derived from umbilical cord blood. Cord blood donors have been screened to exclude donors with sickle cell anemia, and anemias due to abnormalities in hemoglobins C, D, and E. Because of the age of the donor at the time cord blood collection takes place, the ability to exclude rare genetic diseases is severely limited.
-
6 ADVERSE REACTIONS
The following clinically significant adverse events are discussed in greater detail elsewhere in the labeling:
- Hypersensitivity Reactions [See Warnings and Precautions (5.1)]
- Infusion Reactions [See Warnings and Precautions (5.2)]
- Graft versus Host Disease [See Warnings and Precautions (5.3)]
- Engraftment Syndrome [See Warnings and Precautions (5.4)]
- Graft Failure [See Warnings and Precautions (5.5)]
- Malignancies of Donor Origin [see Warnings and Precautions (5.6)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety data described in the WARNINGS AND PRECAUTIONS reflect the exposure of all patients transplanted with OMISIRGE.
The safety of OMISIRGE is based on data from Study P0501 for 52 patients transplanted with OMISIRGE and 56 patients transplanted with umbilical cord blood (UCB) [see Clinical Studies (14)]. The median duration of follow up for the overall safety population was 14 months (range, 1-19 months). All patients received myeloablative preparative regimens and GvHD prophylaxis with tacrolimus or cyclosporin plus mycophenolate mofetil.
Safety data collected included deaths, infusion reactions, infections, GvHD, serious adverse events, and Common Terminology Criteria for Adverse Events (CTCAE) Grade ≥ 3 adverse reactions through the follow-up period.
Fatal adverse reactions occurred in 17% of patients treated with OMISIRGE, including infection (6%), acute GvHD (6%), veno-occlusive disease (VOD)/sinusoidal obstruction syndrome (SOS) (2%), thrombotic thrombocytopenic purpura (TTP)/thrombotic microangiopathy (TMA) (2%), and pulmonary hemorrhage (2%). Fatal adverse reactions occurred in 29% of subjects treated with UCB, including infection/sepsis (11%), respiratory disorders (11%), GvHD (5%), and VOD/SOS (2%).
Infusion Reactions
Infusion reactions occurred in 29 (56%) patients in the OMISIRGE arm and in 40 (71%) patients in the UCB arm, including CTCAE Grade 3-4 events in 17% and 21% of patients, respectively. The most common infusion reactions were hypertension, mucosal inflammation, arrythmia, and fatigue. The most common Grade 3 to 4 infusion reaction was hypertension, reported in 3 (6%) patients treated with OMISIRGE and 9 (16%) patients treated with UCB.
Infections
Infections, including life-threatening and fatal infections, occurred after OMISIRGE infusion. Infections were graded per the Blood and Marrow Transplant Clinical Trials Network (BMT CTN) grading criteria. Data were collected for treated Grade 1 and all Grade 2/3 infections. The incidence of infections following transplantation are shown in the table below.
Table 2: Infections Following Transplantation with OMISIRGE or UCB in Study P0501 OMISIRGE
N = 52UCB
N = 56Grade 1-3
%Grade 2
%Grade 3
%Grade 1-3
%Grade 2
%Grade 3
%Viral infections 75 48 8 80 32 27 Bacterial infections 65 27 8 80 46 23 Fungal Infections 21 4 6 27 0 18 Graft-versus-Host Disease
Acute and chronic GvHD occurred following treatment with OMISIRGE.
Grade II to IV acute GvHD (Przepiorka et al., 1995) was reported in 62% of patients treated with OMISIRGE and 43% in patients treated with UCB. Grade III to IV acute GvHD was reported in 15% versus 21% of patients, respectively.
Chronic GvHD (National Institutes of Health [NIH] Consensus Criteria) was reported in 35% of patients in the OMISIRGE arm and 25% in the UCB arm. Moderate to severe chronic GvHD was reported in 23% versus 20% of patients, respectively.
Graft Failure
Primary graft failure (defined as failure to achieve an absolute neutrophil count greater than or equal to 0.5 Gi/L by Day 42 after transplantation) occurred in 2% of patients treated with OMISIRGE, compared to 11% of patients receiving UCB. One patient treated with OMISIRGE had a secondary graft failure approximately six months following transplantation, concurrent with a diagnosis of acute lymphocytic leukemia (ALL) relapse.
Relapse of Hematologic Malignancy
Disease relapse occurred in 21% of patients treated with OMISIRGE compared to 13% of patients treated with UCB.
Other Adverse Reactions
CTCAE Grade 3-5 non-laboratory adverse reactions in Study P0501 with greater or equal to 10% incidence are summarized in Table 3. The most common Grade 3-5 adverse reactions for patients treated with OMISIRGE, were pain (33%), mucosal inflammation (31%), hypertension (25%), and gastrointestinal toxicity (19%).
Table 3: CTCAE Grade 3 or Higher Adverse Reactions in ≥ 10% of Patients with Hematologic Malignancies Following Transplantation with OMISIRGE or UCB in Study P0501 Adverse Reaction OMISIRGE
N = 52
%UCB
N = 56
%- *
- Fatigue includes asthenia and fatigue
- †
- Hemorrhage include cystitis hemorrhagic, epistaxis, gastrointestinal hemorrhage, hemorrhage, pulmonary alveolar hemorrhage, subarachnoid hemorrhage, and upper gastrointestinal hemorrhage
- ‡
- Renal impairment includes acute kidney injury, blood creatinine increased and renal failure
- §
- Respiratory failure includes acute respiratory distress syndrome, acute respiratory failure, hypoxia, and respiratory failure Abbreviation: CTCAE, common terminology criteria for adverse events; UCB, umbilical cord blood
General disorders and administration site conditions Pain 33 18 Mucosal inflammation 31 34 Fatigue * 4 21 Fever 2 11 Vascular disorders Hypertension 25 38 Hemorrhage † 12 18 Gastrointestinal disorders Gastrointestinal toxicity 19 34 Dysphagia 12 13 Renal and urinary disorders Renal impairment ‡ 12 5 Respiratory, thoracic, and mediastinal disorders Respiratory Failure § 12 30 Dyspnea 8 16 Table 4 summarizes selected chemistry abnormalities by treatment arm for patients treated in Study P0501.
Table 4: Chemistry Laboratory Abnormalities in ≥10% of Patients in Study P0501 OMISIRGE
N = 52UCB
N = 56Laboratory Abnormality Grade 1-4
%Grade 3-4
%Grade 1-4
%Grade 3-4
%Magnesium decreased 94 4 91 2 Aspartate aminotransferase increased 56 13 61 7 Alanine aminotransferase increased 56 13 57 9 Creatinine increased 50 4 57 2 Bilirubin increased 42 12 61 21 Alkaline phosphatase increased 42 0 54 2 Magnesium increased 15 2 29 9 - 7 DRUG INTERACTIONS
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no available data with OMISIRGE use in pregnant women. No animal reproductive and developmental toxicity studies have been conducted with OMISIRGE to assess whether it can cause fetal harm when administered to a pregnant woman.
OMISIRGE should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
In the United States (U.S.) general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
8.2 Lactation
Risk Summary
There is no information regarding the presence of OMISIRGE in human milk, the effect on the breastfed infant, and the effects on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for OMISIRGE and any potential adverse effects on the breastfed infant from OMISIRGE or from the underlying maternal condition.
8.3 Females and Males of Reproductive Potential
Pregnancy Testing
Pregnancy status of females with reproductive potential should be verified. Sexually-active females of reproductive potential should have a pregnancy test prior to starting the conditioning regimen for OMISIRGE.
Contraception
See the prescribing information for the medications used for conditioning for information on the need for effective contraception in patients who receive a conditioning regimen.
There are insufficient exposure data to provide a recommendation concerning duration of contraception following treatment with OMISIRGE.
8.4 Pediatric Use
The safety and efficacy of OMISIRGE have been established in adolescents (12 years to less than 17 years). The efficacy and safety outcomes in Study P0501 suggest consistent efficacy and safety across age groups.
Safety and efficacy of OMISIRGE in pediatric patients below the age of 12 have not been established.
-
11 DESCRIPTION
OMISIRGE (omidubicel-onlv) is a cryopreserved nicotinamide modified allogeneic hematopoietic progenitor cell therapy derived from cord blood consisting of 2 cell fractions; a Cultured Fraction (CF) and a Non-cultured Fraction (NF) which are both derived from the same patient-specific cord blood unit (CBU).
1) The CF is a yellowish suspension consisting of allogeneic, hematopoietic CD34+ progenitor cells.
In addition to the CD34+ hematopoietic progenitor cells (HPCs), the CF consists of other cell populations, including more differentiated myelomonocytic cells, dendritic cells and granulocytes. The CF formulation contains a maximum of 35 mg gentamicin. Following manipulation, the cells are washed, formulated into a suspension, and cryopreserved in a patient specific bag in 10% DMSO. The product is thawed and diluted prior to infusion [see Dosage and Administration (2.2), How Supplied/Storage and Handling (16)]. The CF contains approximately 2.42 mg of DMSO. The diluted CF contains ≤ 2% DMSO.
2) The NF is a reddish suspension consisting of allogeneic, hematopoietic mature myeloid and lymphoid cells that are washed, formulated into a suspension, and cryopreserved in a patient specific bag in 10% DMSO. In addition to the mature myeloid and lymphoid cells, the NF consists of other cell populations, including more lineage committed hematopoietic cells. The product is thawed and diluted prior to infusion [see Dosage and Administration (2.2), How Supplied/Storage and Handling (16)]. The NF contains approximately 1.1 mg of DMSO. The diluted NF contains ≤2% DMSO.
Two Infusion Solution bags are also provided for diluting each fraction after thawing, one specifically for the CF and one specifically for the NF. The Infusion Solutions contain 8% w/v HSA and 6.8% w/v Dextran 40 in 0.9% sodium chloride [see Dosage and Administration (2.2)].
The NF and IS are Released for Shipment (RFS) following full release testing, including sterility test . The CF is RFS at the end of manufacturing, prior to final product testing completion. OMISIRGE is Released for Infusion (RFI) after acceptable results from the CF's Rapid Contamination Test for microbial contamination and quantitative PCR-based mycoplasma test are obtained. The RFI includes all CF DP release testing as detailed in the CF CoA, except the pending Colony Forming Unit (CFU) and final Sterility tests results. RFI certificate is accompanied with the respective IS, NF, and CF CoA's. Receipt of the RFI Certificate for the patient-specific batch of OMISIRGE must be confirmed prior to preparation. RFI Certificate receipt is via the Gamida Cell Assist Hospital Portal, ~approximately 72 h of the end of manufacturing.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
OMISIRGE is a nicotinamide (NAM) modified allogeneic hematopoietic progenitor cell therapy derived from cord blood used as an allogeneic stem cell donor source. OMISIRGE is manufactured utilizing a proprietary NAM based technology producing enriched HPCs.
NAM technology overcomes the induction of accelerated proliferation, differentiation, cellular stress and signaling pathways that are typically activated when HPCs are removed from their natural environment.
Ex-vivo culturing of cord blood derived HPCs in the presence of NAM leads to preservation of their stemness, homing to the bone marrow (BM) and retained engraftment capacity as demonstrated by rapid neutrophil engraftment and multi lineage immune reconstitution as observed in the clinical trials with OMISIRGE.
12.2. Pharmacodynamics
Transplantation with OMISIRGE resulted in rapid and broad immune reconstitution of dendritic cells, monocytes, Natural Killer (NK), CD4+ T cells and CD8+ T cells as early as one-week post-transplantation, and B cells 28 days post transplantation and all lineages throughout the one-year follow-up period. Robust positive linear correlations between the CD34(+) cell content in the OMISIRGE CF, and the reconstitution of T-cells and NK cells were identified. Additionally, dose-response analyses demonstrated a strong correlation between the total CD34+ cell counts and dose / kg for OMISIRGE with the kinetics of neutrophil recovery. The model demonstrated that days to neutrophil recovery decreased with an increase in OMISIRGE CD34(+) cell dose.
- 13 NONCLINICAL TOXICOLOGY
-
14 CLINICAL STUDIES
OMISIRGE was evaluated in Study P0501 (NCT02730299), an open-label, multicenter, randomized study of OMISIRGE transplantation or UCB transplantation following myeloablative conditioning in patients with hematologic malignancies.
In total, 125 patients were randomized to the study, 62 patients were randomized to receive OMISIRGE and 63 patients were randomized to the UCB group. The minimum specifications of OMISIRGE were a Total Nucleated Viable Cell (TNVC) count of 8.0 × 108 cells and CD34+ cell count of 5.6 × 107. Fifty-two patients were transplanted with OMISIRGE at a median CD34+ cell dose of 9.0 × 106 cells/kg (range 2.1 – 47.6 × 106 cells/kg). Fifty-six patients were transplanted in the UCB arm with one or two cord units (66% received two cord units); among patients in whom the post-thaw cell dose was reported (n = 42), the median CD34+ cell dose was 0.2 × 106 cells/kg (range 0.0 – 0.8 × 106 cells/kg). Multiple conditioning regimens were used, including Total Body Irradiation (TBI)-based or chemotherapy-based options.
Demographic and baseline patient characteristics were similarly distributed among the treatment arms. The overall study population included 72 males (58%) and 53 females (42%) with median age 41 years (range: 13–65). Fifty-eight percent of patients were White, 16% were Black, 14% were Asian and 13% were of other races or unknown. Thirteen percent of patients identified as Hispanic or Latino. Forty-eight percent of patients had Acute Myelogenous Leukemia (AML), 33% had Acute Lymphoblastic Leukemia (ALL), 7% had Myelodysplastic Syndrome (MDS), 5% had Chronic myeloid leukemia (CML), 4% had lymphoma and 3% had other rare leukemias. Baseline disease status (remission vs. overt disease) varied depending on the hematologic malignancy. Disease risk index was high/very high for 34% and moderate for 42%. HCT specific comorbidity index was ≥ 3 in 51% and 1-2 in 28% of patients.
Of the patients randomized to OMISIRGE, 8% of patients (5/62) were not able to receive OMISIRGE due to manufacturing failure.
The efficacy of OMISIRGE was established based on time to neutrophil recovery following transplantation and the incidence of BMT CTN Grade 2/3 bacterial or Grade 3 fungal infections through Day 100 following transplantation. The efficacy outcomes are summarized below.
Table 5: Efficacy Results in Patients Randomized to Receive OMISIRGE or UCB in Study P0501 (ITT Population) OMISIRGE
N=62UCB
N=63Absolute Difference
(95% CI)Abbreviation: CI: Confidence interval; UCB: umbilical cord blood - *
- Time to neutrophil recovery was defined as the time from transplantation to the earliest of 3 consecutive measurements on different days with absolute neutrophil count greater than or equal to 0.5 Gi/L assessed with 42 days of follow-up.
- †
- Median time to neutrophil recovery was estimated by the Kaplan-Meier estimator.
Median time to neutrophil recovery *,† 12 days
(95% CI: 10-15 days)22 days
(95% CI: 19-25 days)10 days
(95% CI: 6-14 days)Incidence of Grade 2/3 bacterial or Grade 3 fungal infections through 100 days following transplantation 39% 60% 22%
(95% CI: 4%-39%)Eighty-seven percent of patients in the OMISIRGE arm and 83% in the UCB arm achieved neutrophil recovery. The median time to neutrophil recovery was 12 days in the OMISIRGE arm and 22 days in the UCB arm. BMT CTN Grade 2/3 bacterial or Grade 3 fungal infections through Day 100 following transplantation occurred in 39% of patients in the OMISIRGE arm and 60% of patients in the UCB arm.
Per Protocol Population:
Among the patients treated with OMISIRGE (n=52), neutrophil recovery with 42 days of follow-up was achieved in 94% of patients at a median of 10 days (95% CI: 8, 12), compared to 89% of patients treated with UCB (n=56), at a median of 20 days (95% CI: 18, 24). BMT CTN Grade 2/3 bacterial or Grade 3 fungal infections by 100 days were reported in 35% of patients treated with OMISIRGE and 61% of patients treated with UCB, respectively.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
OMISIRGE (NDC 73441-800-04) is shipped in two shipping containers, a liquid nitrogen dry vapor shipper at ≤ -150°C, containing the two cryopreserved cell fractions (CF NDC 73441-100-01 and NF NDC 73441-200-01) and a Chimerism Testing Sample(s) and a refrigerated shipping container at 2-8°C, containing two Infusion Solutions (Infusion Solution for CF NDC 73441-300-01 and Infusion Solution for NF NDC 73441-400-01). OMISIRGE is shipped to the transplant center for a specific patient.
The Cryopreserved Cell Fractions
OMISIRGE is comprised of two cryopreserved cell fractions, a Cultured Fraction (CF) and a Non-cultured Fraction (NF) each in a separate cryopreserved bag labeled for the specific patient. Each cryopreserved bag is protected by a corresponding transparent overwrap bag and each cryopreserved bag enclosed in its overwrap bag is individually packed in a metal cassette. The cassettes are NOT to be opened upon receipt. Both cryopreserved OMISIRGE cell fractions are shipped together in the vapor phase of liquid nitrogen in a liquid nitrogen dry vapor shipper with the Prescribing Information (PI) and a Chimerism Testing Sample(s).
At the time of cryopreservation, the CF contains a minimum of 8.0 × 108 total viable cells with a minimum of 8.7% CD34+ cells and a minimum of 9.2 × 107 CD34+ cells suspended in 20 mL of a cryopreservation solution containing 10% DMSO.
See the CoA for the CF for actual cell counts. CoAs are attached to the RFI Certificate available via the Gamida Cell Assist Hospital Portal.
Upon cryopreservation, the CF appears white and is frozen at the bottom of the cryopreserved bag. The cassette may not be opened until the time of thaw for the specific fraction.
At the time of cryopreservation, the NF contains a minimum of 4.0 × 108 total viable cells with a minimum of 2.4 × 107 CD3+ cells suspended in 10 mL cryopreservation solution containing 10% DMSO.
See the CoA for the NF for actual cell counts.
Upon cryopreservation, the NF appears red and is frozen at the bottom of the cryopreserved bag. The cassette may not be opened until the time of thaw for the specific fraction.
- Do NOT open the metal cassettes until the time of thaw.
- Match the identity of the patient with the patient-specific identifiers on the cassettes and cryopreserved bag labels (visible through the cassette window) upon receipt.
- Store OMISIRGE frozen in the vapor phase of liquid nitrogen (≤ -150°C) in a temperature-controlled system.
- Thaw immediately prior to use [see Dosage and Administration (2.2)]
- Use closed containers when transporting the bags within the facility.
The Refrigerated Infusion Solutions
The Infusion Solutions (IS) used to dilute OMISIRGE CF and NF are provided in two IS bags labeled for the specific patient and for diluting the specific fraction. The IS for diluting the CF contains approximately 80 mL and the IS for diluting the NF contains approximately 40 mL of IS consisting of 6.8% Dextran 40 and 8% HSA. The Infusion Solutions are shipped in a refrigerated shipping container with the PI.
- Match the identity of the patient with the patient-specific identifiers on the IS bag labels upon receipt.
- Store the IS bags in a 2-8°C refrigerated storage until the time of thaw of the OMISIRGE CF and NF.
-
17 PATIENT COUNSELING INFORMATION
Discuss the following with the patient receiving OMISIRGE:
The recommended course of therapy for OMISIRGE is a single dose for infusion, which is provided by the manufacturer as 2 separate components (CF and NF). OMISIRGE CF and OMISIRGE NF are infused one after the other once the patient has received an appropriate conditioning regimen.
Each OMISIRGE unit is specific to each patient. Ensure that patients understand the risk of manufacturing failure (8% in Study P0501) of OMISIRGE. In case of a manufacturing failure, a second manufacturing attempt may be considered. In this case, while the patient awaits the product, additional chemotherapy (in addition to the preparative conditioning) may be necessary and may increase the risk of adverse events during the pre-infusion period.
Prior to infusion, advise patients of the following risks:
Hypersensitivity Reactions:
Report immediately any signs and symptoms of hypersensitivity reactions including wheezing, swelling, itching, or hives [See Warnings and Precautions (5.1)].
Infusion Reactions:
Report immediately any signs and symptoms of infusion reactions including fever, chills, fatigue, tachycardia, hypoxia, severe nausea, severe vomiting, diarrhea, muscle pain, joint pain, low blood pressure, high blood pressure, or dizziness/lightheadedness [see Warnings and Precautions (5.2), Adverse Reactions (6.1)].
Graft-versus-Host-Disease:
Report immediately any signs and symptoms suggestive of graft vs host disease, including rash, diarrhea or yellowing of the eyes [see Warnings and Precautions (5.3), Adverse Reactions (6.1)].
Engraftment Syndrome:
Report immediately any signs and symptoms suggestive of engraftment syndrome including fever, rash, or unexplained weight gain [See Warnings and Precautions (5.4)].
Graft Failure:
Advise patients that primary graft failure, which may be fatal, can occur [See Warnings and Precautions (5.5), Adverse Reactions (6.1)].
Malignancies of Donor Origin:
Advise patients of the need to contact Gamida Cell at (844)-477-7478 if they are diagnosed with a secondary malignancy after treatment with OMISIRGE [See Warnings and Precautions (5.6), Adverse Reactions (6.1)].
Transmission of Serious Infections:
Advise patients of the risk of transmission of infectious disease [See Warnings and Precautions (5.7)].
Transmission of Rare Genetic Diseases:
Advise patients of the risk of transmission of rare genetic diseases [See Warnings and Precautions (5.8)].
- SPL UNCLASSIFIED SECTION
-
PRINCIPAL DISPLAY PANEL - 20 mL and 10 mL Shipping Label
NDC 73441-100-01
NDC 73441-200-01Gamida Cell Patient ID: 123456789
Hospital Patient ID: 123456789
Batch: 1234567890
Expiry Date: DD-MMM-CCYYomidubicel-onlv
OMISIRGE®Rx Only
Container 1 of 2Cultured Fraction
Non-cultured FractionSHIP TO:
Transplant Center Name:
Insert Name: XXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXX
Address:
Insert Address: XXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXX
Telephone Number: Insert Number
Receiver Name(s): List NamesProduct of Israel.
Manufactured For and Distributed By: Gamida Cell, Inc. Boston, MA, 02116
Telephone Number: (844) 477-7478Ship and Store in Vapor Phase Liquid Nitrogen ≤ -150°C
DO NOT X-RAY
Exempt Human SpecimenPK000113-7

-
PRINCIPAL DISPLAY PANEL - 80 mL and 40 mL Shipping Label
NDC 73441-300-01
NDC 73441-400-01Gamida Cell Patient ID: 123456789
Hospital Patient ID: 123456789
Batch: 1234567890
Expiry Date: DD-MMM-CCYYomidubicel-onlv
OMISIRGE®Rx Only
Container 2 of 2Infusion Solution for
Cultured Fraction
Infusion Solution for
Non-cultured FractionSHIP TO:
Transplant Center Name:
Insert Name: XXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXX
Address:
Insert Address: XXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXXX
Telephone Number: Insert Number
Receiver Name(s): List NamesProduct of Israel.
Manufactured For and Distributed By: Gamida Cell, Inc. Boston, MA, 02116
Telephone Number: (844) 477-7478Ship and store at 2–8°C
DO NOT X-RAY
Exempt Human SpecimenPK000112-7

-
INGREDIENTS AND APPEARANCE
OMISIRGE
omidubicel-onlv kitProduct Information Product Type CELLULAR THERAPY Item Code (Source) NDC:73441-800 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:73441-800-04 1 in 1 KIT Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 BAG 20 mL Part 2 1 BAG 80 mL Part 3 1 BAG 10 mL Part 4 1 BAG 40 mL Part 1 of 4 OMIDUBICEL CULTURED FRACTION
omidubicel cultured fraction suspensionProduct Information Item Code (Source) NDC:73441-100 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OMIDUBICEL (UNII: ET4JC4S66E) (OMIDUBICEL - UNII:ET4JC4S66E) OMIDUBICEL 800000000 in 100 mL Inactive Ingredients Ingredient Name Strength DIMETHYL SULFOXIDE (UNII: YOW8V9698H) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:73441-100-01 20 mL in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125738 05/01/2023 Part 2 of 4 INFUSION SOLUTION FOR CF
infusion solution solutionProduct Information Item Code (Source) NDC:73441-300 Route of Administration INTRAVENOUS Inactive Ingredients Ingredient Name Strength ALBUMIN HUMAN (UNII: ZIF514RVZR) DEXTRAN 40 (UNII: K3R6ZDH4DU) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:73441-300-01 80 mL in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125738 05/01/2023 Part 3 of 4 OMIDUBICEL NON-CULTURED FRACTION
omidubicel non-cultured fraction suspensionProduct Information Item Code (Source) NDC:73441-200 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OMIDUBICEL NON-CULTURED FRACTION (UNII: MAH7ZHD7ZJ) (OMIDUBICEL NON-CULTURED FRACTION - UNII:MAH7ZHD7ZJ) OMIDUBICEL NON-CULTURED FRACTION 400000000 in 50 mL Inactive Ingredients Ingredient Name Strength DIMETHYL SULFOXIDE (UNII: YOW8V9698H) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:73441-200-01 10 mL in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125738 05/01/2023 Part 4 of 4 INFUSION SOLUTION FOR NF
infusion solution solutionProduct Information Item Code (Source) NDC:73441-400 Route of Administration INTRAVENOUS Inactive Ingredients Ingredient Name Strength ALBUMIN HUMAN (UNII: ZIF514RVZR) DEXTRAN 40 (UNII: K3R6ZDH4DU) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:73441-400-01 40 mL in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125738 05/01/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125738 05/01/2023 Labeler - Gamida Cell Inc. (093828795) Registrant - Gamida Cell LTD (532501574) Establishment Name Address ID/FEI Business Operations Gamida Cell LTD 532501574 API MANUFACTURE(73441-800) , MANUFACTURE(73441-800) , LABEL(73441-800) , PACK(73441-800) , ANALYSIS(73441-800) Establishment Name Address ID/FEI Business Operations Hy Laboratories Ltd. 600013676 ANALYSIS(73441-800) Establishment Name Address ID/FEI Business Operations Alcami Carolinas Corporation 831351445 ANALYSIS(73441-800) Establishment Name Address ID/FEI Business Operations Alcami Carolinas Corporation 832394535 ANALYSIS(73441-800) Establishment Name Address ID/FEI Business Operations Eurofins Biolab S.r.l 429117112 ANALYSIS(73441-800)


