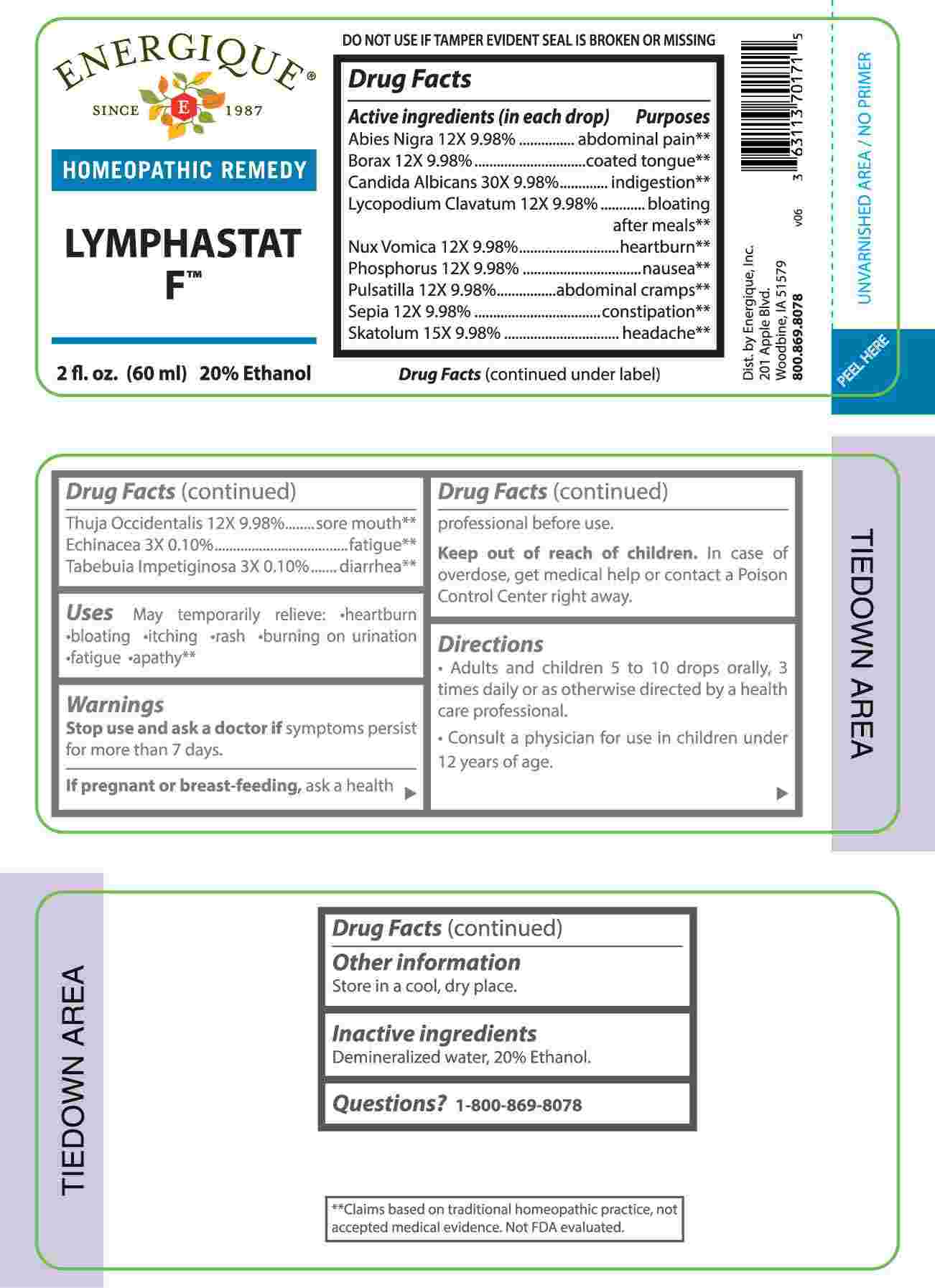
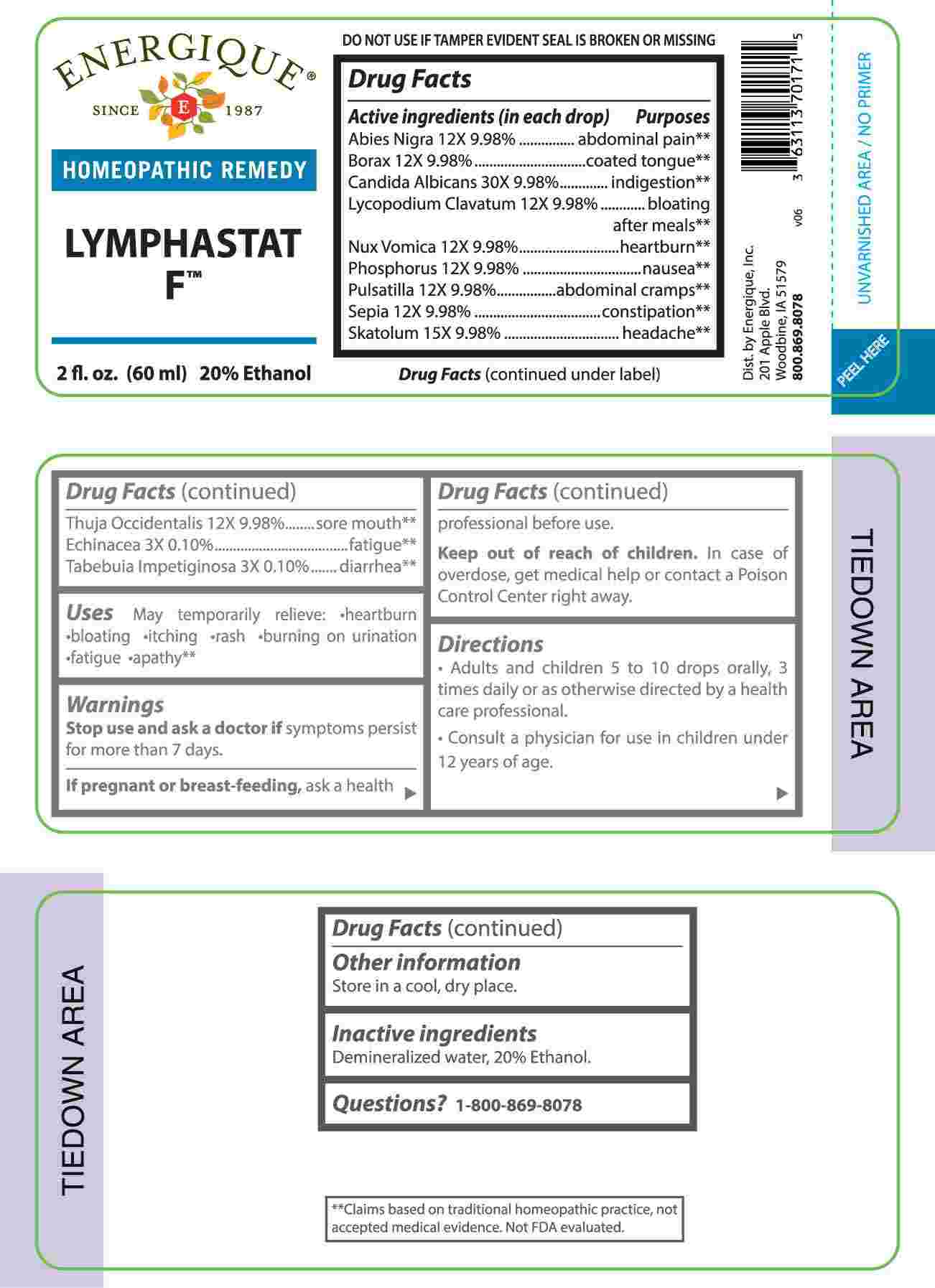
Label: LYMPHASTAT F (echinacea (angustifolia), tabebuia impetiginosa, abies nigra, borax, lycopodium clavatum, nux vomica, phosphorus, pulsatilla- vulgaris, sepia, thuja occidentalis, skatolum, candida albicans liquid
- NDC Code(s): 44911-0708-1
- Packager: Energique, Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated March 8, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENTS:
-
PURPOSE
Abies Nigra – abdominal pain,** Borax – coated tongue,** Candida Albicans - indigestion,** Lycopodium Clavatum – bloating after meals,** Nux Vomica - heartburn,** Phosphorus - nausea,** Pulsatilla – abdominal cramps,** Sepia - constipation,** Skatolum - headache,** Thuja Occidentalis – sore mouth,** Echinacea - fatigue,** Tabebuia Impetiginosa – diarrhea**
**Claims based on traditional homeopathic practice, not accepted medical evidence. Not FDA evaluated.
- USES:
-
WARNINGS:
Stop use and ask a doctor if symptoms persist for more than 7 days.
If pregnant or breast-feeding, ask a health professional before use.
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
DO NOT USE IF TAMPER EVIDENT SEAL IS BROKEN OR MISSING
Store in a cool, dry place.
- KEEP OUT OF REACH OF CHILDREN:
- DIRECTIONS:
- INACTIVE INGREDIENTS:
- QUESTIONS:
- PACKAGE LABEL DISPLAY:
-
INGREDIENTS AND APPEARANCE
LYMPHASTAT F
echinacea (angustifolia), tabebuia impetiginosa, abies nigra, borax, lycopodium clavatum, nux vomica, phosphorus, pulsatilla (vulgaris), sepia, thuja occidentalis, skatolum, candida albicans liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:44911-0708 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ECHINACEA ANGUSTIFOLIA WHOLE (UNII: VB06AV5US8) (ECHINACEA ANGUSTIFOLIA - UNII:VB06AV5US8) ECHINACEA ANGUSTIFOLIA WHOLE 3 [hp_X] in 1 mL HANDROANTHUS IMPETIGINOSUS BARK (UNII: 6GLA1946WX) (HANDROANTHUS IMPETIGINOSUS BARK - UNII:6GLA1946WX) HANDROANTHUS IMPETIGINOSUS BARK 3 [hp_X] in 1 mL PICEA MARIANA RESIN (UNII: 71AOV0W131) (PICEA MARIANA RESIN - UNII:71AOV0W131) PICEA MARIANA RESIN 12 [hp_X] in 1 mL SODIUM BORATE (UNII: 91MBZ8H3QO) (BORATE ION - UNII:44OAE30D22) SODIUM BORATE 12 [hp_X] in 1 mL LYCOPODIUM CLAVATUM SPORE (UNII: C88X29Y479) (LYCOPODIUM CLAVATUM SPORE - UNII:C88X29Y479) LYCOPODIUM CLAVATUM SPORE 12 [hp_X] in 1 mL STRYCHNOS NUX-VOMICA SEED (UNII: 269XH13919) (STRYCHNOS NUX-VOMICA SEED - UNII:269XH13919) STRYCHNOS NUX-VOMICA SEED 12 [hp_X] in 1 mL PHOSPHORUS (UNII: 27YLU75U4W) (PHOSPHORUS - UNII:27YLU75U4W) PHOSPHORUS 12 [hp_X] in 1 mL PULSATILLA VULGARIS WHOLE (UNII: I76KB35JEV) (ANEMONE PULSATILLA - UNII:I76KB35JEV) PULSATILLA VULGARIS WHOLE 12 [hp_X] in 1 mL SEPIA OFFICINALIS JUICE (UNII: QDL83WN8C2) (SEPIA OFFICINALIS JUICE - UNII:QDL83WN8C2) SEPIA OFFICINALIS JUICE 12 [hp_X] in 1 mL THUJA OCCIDENTALIS LEAFY TWIG (UNII: 1NT28V9397) (THUJA OCCIDENTALIS LEAFY TWIG - UNII:1NT28V9397) THUJA OCCIDENTALIS LEAFY TWIG 12 [hp_X] in 1 mL SKATOLE (UNII: 9W945B5H7R) (SKATOLE - UNII:9W945B5H7R) SKATOLE 15 [hp_X] in 1 mL CANDIDA ALBICANS (UNII: 4D7G21HDBC) (CANDIDA ALBICANS - UNII:4D7G21HDBC) CANDIDA ALBICANS 30 [hp_X] in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) ALCOHOL (UNII: 3K9958V90M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:44911-0708-1 60 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product 03/08/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 03/08/2024 Labeler - Energique, Inc (789886132) Registrant - Apotheca Company (844330915) Establishment Name Address ID/FEI Business Operations Apotheca Company 844330915 manufacture(44911-0708) , api manufacture(44911-0708) , label(44911-0708) , pack(44911-0708)