Label: PREPARATION H- phenylephrine hydrochloride suppository
- NDC Code(s): 0573-1378-00, 0573-1378-01, 0573-1378-06
- Packager: Haleon US Holdings LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated March 2, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
-
Warnings
For rectal use only
Ask a doctor before use if you have
- heart disease
- high blood pressure
- thyroid disease
- diabetes
- difficulty in urination due to enlargement of the prostate gland
-
Directions
- adults: when practical, cleanse the affected area by patting or blotting with an appropriate cleansing wipe. Gently dry by patting or blotting with a tissue or a soft cloth before insertion of this product.
- detach one suppository from the strip; remove the foil wrapper before inserting into the rectum as follows:
- hold suppository with the rounded end up
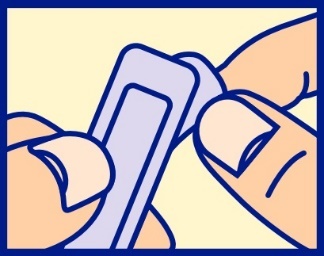
- as shown, carefully separate foil tabs by inserting tip of fingernail at end marked “peel down”
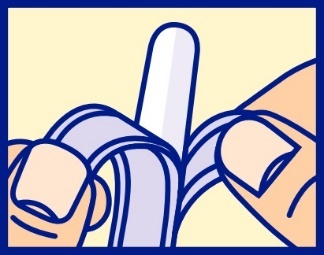
- slowly and evenly peel apart (do not tear) foil by pulling tabs down both sides, to expose the suppository
- remove exposed suppository from the wrapper
- insert one suppository into the rectum up to 4 times daily, especially at night, in the morning or after each bowel movement
- children under 12 years of age: consult a doctor
- Other information
- Inactive ingredients
- Questions or comments?
-
Additional Information
Distributed by: GSK Consumer Healthcare, Warren, NJ 07059
For most recent product information, visit www.preparationh.com
Trademarks are owned by or licensed to the GSK group of companies.
©2022 GSK group of companies or its licensor. Made in GermanyIndividually quality sealed for your protection.
Do Not Use if foil imprinted “PREPARATION H” is torn or damaged.
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
PREPARATION H
phenylephrine hydrochloride suppositoryProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0573-1378 Route of Administration RECTAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE HYDROCHLORIDE 6.25 mg Inactive Ingredients Ingredient Name Strength COCOA BUTTER (UNII: 512OYT1CRR) STARCH, CORN (UNII: O8232NY3SJ) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLPARABEN (UNII: Z8IX2SC1OH) Product Characteristics Color yellow (Light yellow) Score no score Shape BULLET (Bullet shaped suppository) Size 32mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0573-1378-01 12 in 1 CARTON 12/16/2022 1 1 in 1 BLISTER PACK; Type 0: Not a Combination Product 2 NDC:0573-1378-06 24 in 1 CARTON 12/16/2022 2 1 in 1 BLISTER PACK; Type 0: Not a Combination Product 3 NDC:0573-1378-00 48 in 1 CARTON 12/16/2022 3 1 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M015 12/16/2022 Labeler - Haleon US Holdings LLC (079944263)