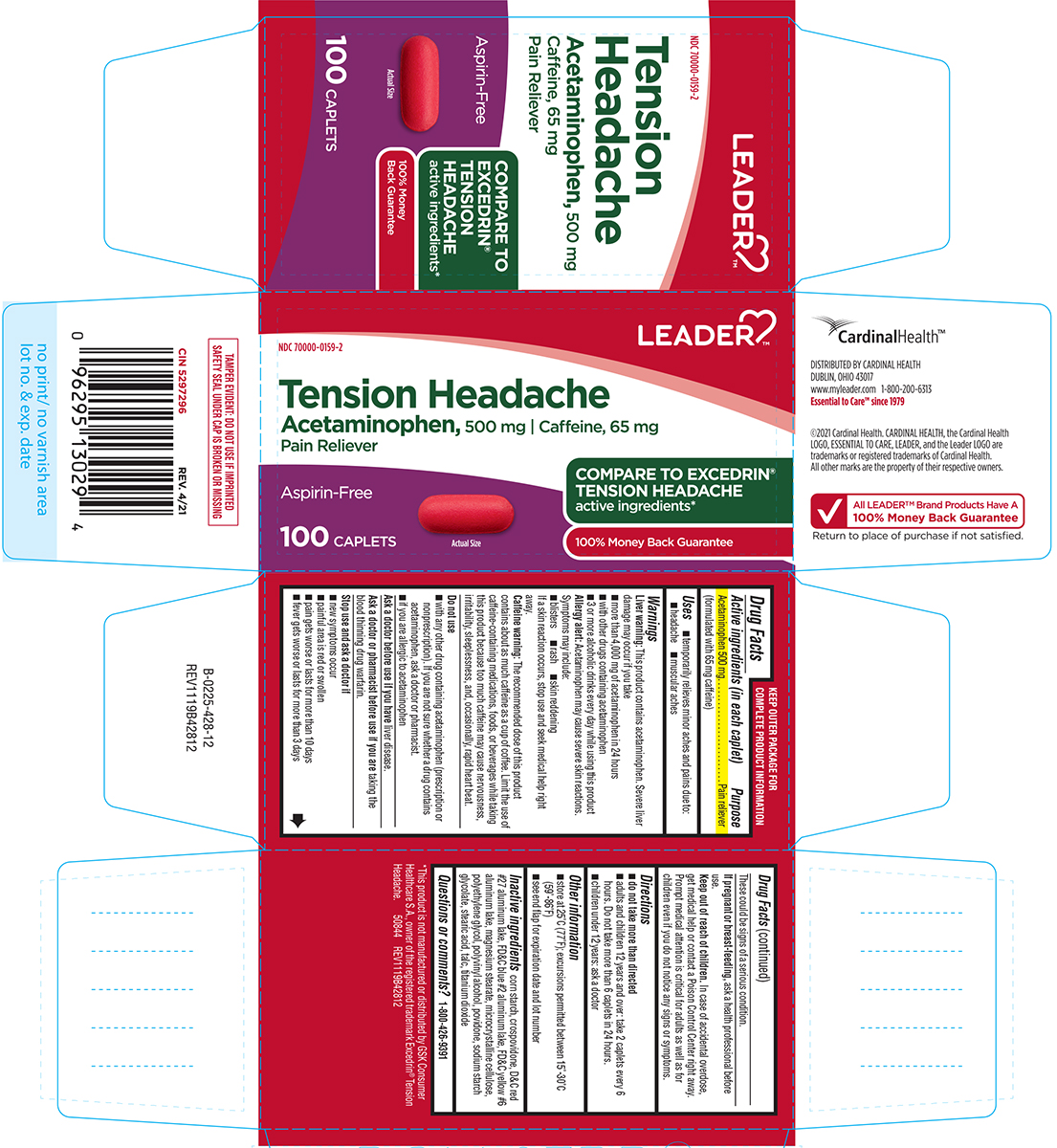
Label: TENSION HEADACHE- acetaminophen, caffeine tablet, film coated
- NDC Code(s): 70000-0159-1, 70000-0159-2
- Packager: Cardinal Health 110, LLC. DBA Leader
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated June 6, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients (in each caplet)
- Purpose
- Uses
-
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take
- more than 4,000 mg of acetaminophen in 24 hours
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- blisters
- rash
- skin reddening
If a skin reaction occurs, stop use and seek medical help right away.
Caffeine warning: The recommended dose of this product contains about as much caffeine as a cup of coffee. Limit the use of caffeine-containing medications, foods, or beverages while taking this product because too much caffeine may cause nervousness, irritability, sleeplessness, and, occasionally, rapid heart beat.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are allergic to acetaminophen
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
Principal Display Panel
LEADER™
NDC 70000-0159-2
Tension Headache
Acetaminophen, 500 mg
Caffeine, 65 mg
Pain relieverAspirin-Free
COMPARE TO
EXCEDRIN®
TENSION
HEADACHE
active ingredients*
100% Money
Back Guarantee100 CAPLETS
Actual Size
TAMPER EVIDENT: DO NOT USE IF IMPRINTED
SAFETY SEAL UNDER CAP IS BROKEN OR MISSING*This product is not manufactured or distributed by GSK Consumer
Healthcare S.A., owner of the registered trademark Excedrin® Tension
Headache.50844 REV1119B42812
CardinalHealth™
DISTRIBUTED BY CARDINAL HEALTH
DUBLIN, OHIO 43017
www.myleader.com 1-800-200-6313
Essential to Care™ since 1979©2021 Cardinal Health. CARDINAL HEALTH, the Cardinal Health
LOGO, ESSENTIAL TO CARE, LEADER, and the Leader LOGO are
trademarks or registered trademarks of Cardinal Health.
All other marks are the property of their respective owners.√ All LEADER™ Brand Products Have A
100% Money Back Guarantee
Return to place of purchase if not satisfied.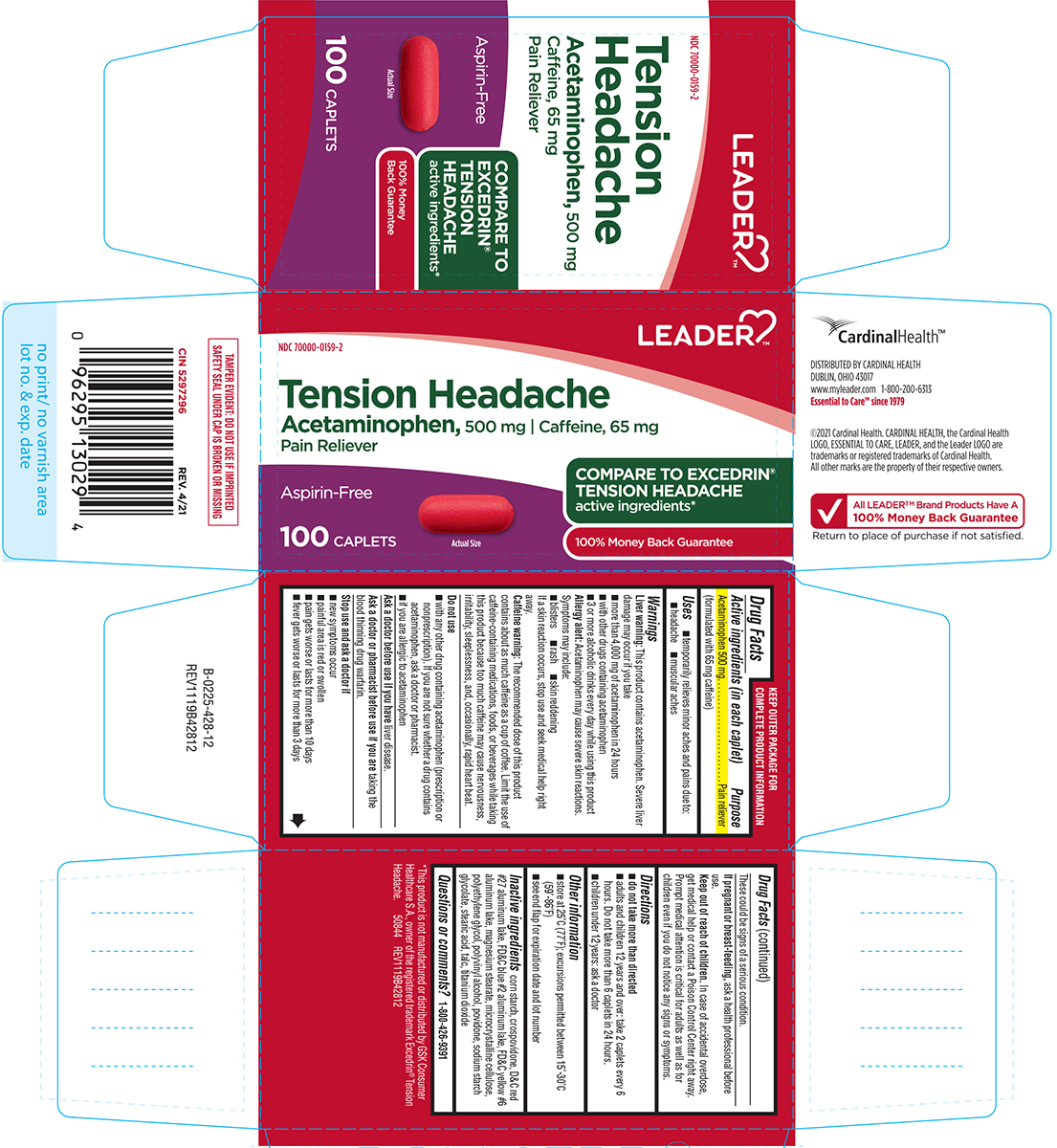
Leader 44-428
-
INGREDIENTS AND APPEARANCE
TENSION HEADACHE
acetaminophen, caffeine tablet, film coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:70000-0159 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 500 mg CAFFEINE (UNII: 3G6A5W338E) (CAFFEINE - UNII:3G6A5W338E) CAFFEINE 65 mg Inactive Ingredients Ingredient Name Strength STARCH, CORN (UNII: O8232NY3SJ) CROSPOVIDONE (UNII: 2S7830E561) D&C RED NO. 27 ALUMINUM LAKE (UNII: ZK64F7XSTX) FD&C BLUE NO. 2 ALUMINUM LAKE (UNII: 4AQJ3LG584) FD&C YELLOW NO. 6 ALUMINUM LAKE (UNII: GYP6Z2JR6Q) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) STEARIC ACID (UNII: 4ELV7Z65AP) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color red Score no score Shape OVAL Size 17mm Flavor Imprint Code 44;428 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70000-0159-1 1 in 1 CARTON 01/17/2007 1 24 in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:70000-0159-2 1 in 1 CARTON 01/17/2007 2 100 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M013 01/17/2007 Labeler - Cardinal Health 110, LLC. DBA Leader (063997360) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 038154464 pack(70000-0159) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867837 manufacture(70000-0159) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867894 manufacture(70000-0159) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 967626305 pack(70000-0159) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 117025878 manufacture(70000-0159)