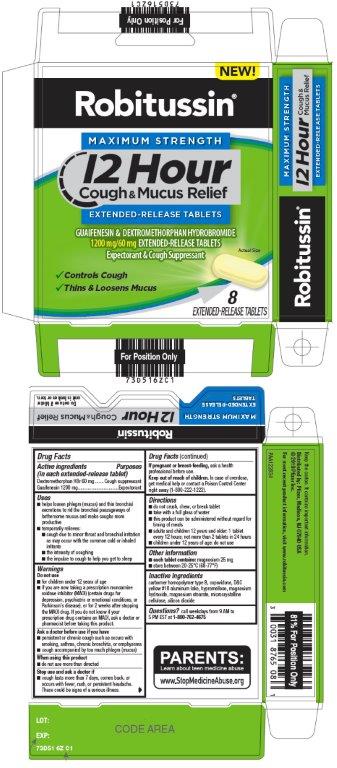
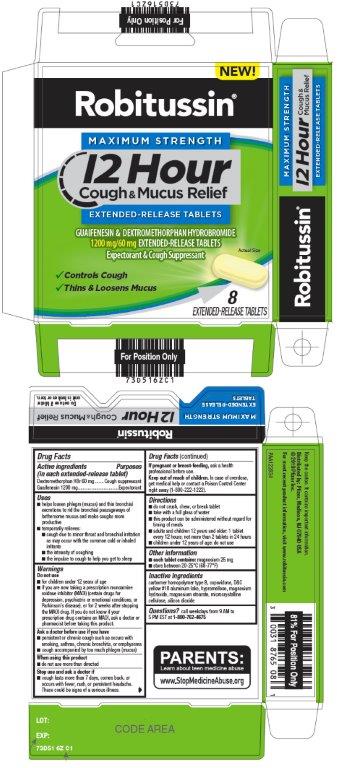
Label: ROBITUSSIN MAXIMUM STRENGTH 12 HOUR COUGH AND MUCUS RELIEF- dextromethorphan hydrobromide, guaifenesin tablet, extended release
- NDC Code(s): 0031-8765-04, 0031-8765-08, 0031-8765-16
- Packager: Haleon US Holdings LLC
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated April 7, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients (in each extended-release tablet)
- Purposes
-
INDICATIONS & USAGE
Uses
- helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive
- temporarily relieves:
- cough due to minor throat and bronchial irritation as may occur with the common cold or inhaled irritants
- the intensity of coughing
- the impulse to cough to help you get to sleep
-
WARNINGS
Warnings
Do not use
- for children under 12 years of age
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema
- cough accompanied by too much phlegm (mucus)
- DOSAGE & ADMINISTRATION
- STORAGE AND HANDLING
- INACTIVE INGREDIENT
- QUESTIONS
- SPL UNCLASSIFIED SECTION
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
ROBITUSSIN MAXIMUM STRENGTH 12 HOUR COUGH AND MUCUS RELIEF
dextromethorphan hydrobromide, guaifenesin tablet, extended releaseProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0031-8765 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DEXTROMETHORPHAN HYDROBROMIDE (UNII: 9D2RTI9KYH) (DEXTROMETHORPHAN - UNII:7355X3ROTS) DEXTROMETHORPHAN HYDROBROMIDE 60 mg GUAIFENESIN (UNII: 495W7451VQ) (GUAIFENESIN - UNII:495W7451VQ) GUAIFENESIN 1200 mg Inactive Ingredients Ingredient Name Strength CARBOMER HOMOPOLYMER TYPE B (ALLYL PENTAERYTHRITOL OR ALLYL SUCROSE CROSSLINKED) (UNII: K6MOM3T5YL) COPOVIDONE K25-31 (UNII: D9C330MD8B) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MAGNESIUM HYDROXIDE (UNII: NBZ3QY004S) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) Product Characteristics Color yellow Score no score Shape OVAL Size 22mm Flavor Imprint Code 2424 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0031-8765-04 1 in 1 CARTON 06/14/2019 1 4 in 1 BLISTER PACK; Type 0: Not a Combination Product 2 NDC:0031-8765-08 1 in 1 CARTON 06/14/2019 2 8 in 1 BLISTER PACK; Type 0: Not a Combination Product 3 NDC:0031-8765-16 2 in 1 CARTON 06/14/2019 3 8 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA207602 06/14/2019 Labeler - Haleon US Holdings LLC (079944263)