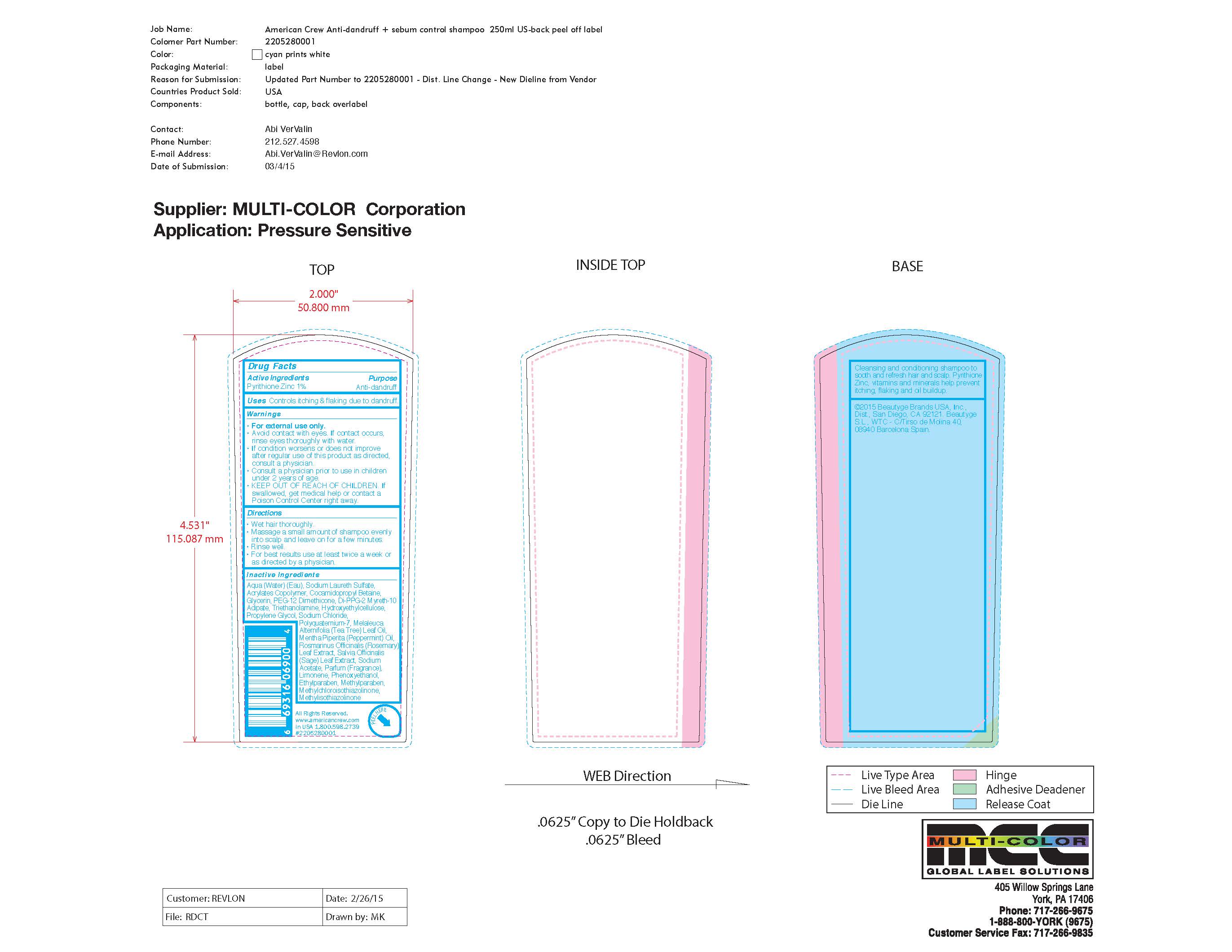
Label: AMERICAN CREW- pyrithione zinc shampoo
- NDC Code(s): 10967-650-25
- Packager: Revlon Consumer Products Corp
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated November 11, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients:
- Purpose
- Uses:
-
Warnings:
For external use only.
• Avoid contact with eyes. If contact occurs,
rinse eyes thoroughly with water.
• If condition worsens or does not improve
after regular use of this product as directed,
consult a physician.
• Consult a physician prior to use in children
under 2 years of age.
• KEEP OUT OF REACH OF CHILDREN. If
swallowed, get medical help or contact a
Poison Control Center right away. - Directions
-
Inactive Ingredients
Aqua (Water) (Eau), Sodium Laureth Sulfate,
Acrylates Copolymer, Cocamidopropyl Betaine,
Glycerin, PEG-12 Dimethicone, Di-PPG-2 Myreth-10
Adipate, Triethanolamine, Hydroxyethylcellulose,
Propylene Glycol, Sodium Chloride,
Polyquaternium-7, Melaleuca
Alternifolia (Tea Tree) Leaf Oil,
Mentha Piperita (Peppermint) Oil,
Rosmarinus Officinalis (Rosemary)
Leaf Extract, Salvia Officinalis
(Sage) Leaf Extract, Sodium
Acetate, Parfum (Fragrance),
Limonene, Phenoxyethanol,
Ethylparaben, Methylparaben - DOSAGE & ADMINISTRATION
- Keep out of Reach of Children
- Label
-
INGREDIENTS AND APPEARANCE
AMERICAN CREW
pyrithione zinc shampooProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:10967-650 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PYRITHIONE ZINC (UNII: R953O2RHZ5) (PYRITHIONE ZINC - UNII:R953O2RHZ5) PYRITHIONE ZINC 1 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) SODIUM LAURETH SULFATE (UNII: BPV390UAP0) COCAMIDOPROPYL BETAINE (UNII: 5OCF3O11KX) GLYCERIN (UNII: PDC6A3C0OX) DI-PPG-2 MYRETH-10 ADIPATE (UNII: 4IN301M0KJ) TROLAMINE (UNII: 9O3K93S3TK) PEG-12 DIMETHICONE (UNII: ZEL54N6W95) HYDROXYETHYL CELLULOSE (100 MPA.S AT 2%) (UNII: R33S7TK2EP) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SODIUM CHLORIDE (UNII: 451W47IQ8X) POLYQUATERNIUM-7 (70/30 ACRYLAMIDE/DADMAC; 1600 KD) (UNII: 0L414VCS5Y) TEA TREE OIL (UNII: VIF565UC2G) PEPPERMINT OIL (UNII: AV092KU4JH) ROSEMARY OIL (UNII: 8LGU7VM393) SAGE OIL (UNII: U27K0H1H2O) SODIUM ACETATE (UNII: 4550K0SC9B) SODIUM HYDROXIDE (UNII: 55X04QC32I) PHENOXYETHANOL (UNII: HIE492ZZ3T) ETHYLPARABEN (UNII: 14255EXE39) METHYLPARABEN (UNII: A2I8C7HI9T) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10967-650-25 250 mL in 1 CONTAINER; Type 0: Not a Combination Product 01/01/2013 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part358H 01/01/2013 Labeler - Revlon Consumer Products Corp (788820165) Establishment Name Address ID/FEI Business Operations REVLON, INC. 809725570 manufacture(10967-650)