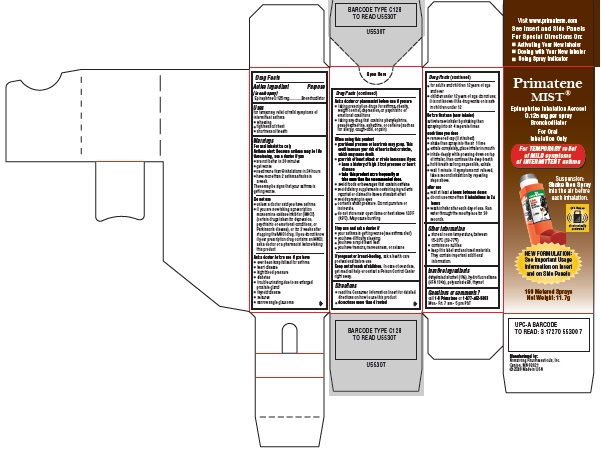
Label: PRIMATENE MIST- epinephrine inhalation aerosol
- NDC Code(s): 17270-5530-0
- Packager: Armstrong Pharmaceuticals, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated July 1, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each spray)
- Purpose
- Uses
-
Warnings
For oral inhalation only
Asthma alert: Because asthma may be life threatening, see a doctor if you
■ are not better in 20 minutes
■ get worse
■ need more than 8 inhalations in 24 hours
■ have more than 2 asthma attacks in a week
These may be signs that your asthma is
getting worse.Do not use
■ unless a doctor said you have asthma
■ if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs taken for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or a pharmacist before taking this product.Ask a doctor before use if you have
■ ever been hospitalized for asthma
■ heart disease
■ high blood pressure
■ diabetes
■ trouble urinating due to an enlarged prostate gland
■ thyroid disease
■ seizures
■ narrow angle glaucomaAsk a doctor or pharmacist before use if you are
■ taking prescription drugs for asthma, obesity, weight control, depression, or psychiatric or emotional conditions
■ taking any drug that contains phenylephrine, pseudoephedrine, ephedrine, or caffeine (such as for allergy, cough-cold, or pain)When using this product
■ your blood pressure or heart rate may go up. This could increase your risk of heart attack or stroke, which may cause death.
■ your risk of heart attack or stroke increases if you:
■ have a history of high blood pressure or heart disease
■ take this product more frequently or take more than the recommended dose.
■ avoid foods or beverages that contain caffeine
■ avoid dietary supplements containing ingredients reported or claimed to have a stimulant effect
■ avoid spraying in eyes
■ contents under pressure. Do not puncture or incinerate.
■ do not store near open flame or heat above 120°F (49°C). May cause bursting. -
Directions
■ read the Consumer Information Insert for detailed directions on how to use this product
■ do not use more than directed■ for adults and children 12 years of age and over
■ children under 12 years of age: do not use; it is not known if the drug works or is safe in children under 12Before first use (new inhaler)
activate new inhaler by shaking then spraying into air 4 separate timeseach time you dose
■ remove red cap (if attached)
■ shake then spray into the air 1 time
■ exhale completely, place inhaler in mouth
■ inhale deeply while pressing down on top of inhaler, then continue the deep breath
■ hold breath as long as possible, exhale
■ wait 1 minute. If symptoms not relieved, take a second inhalation by repeating steps above.after use
■ wait at least 4 hours between doses
■ do not use more than 8 inhalations in 24 hours
■ wash inhaler after each day of use. Run water through the mouthpiece for 30 seconds. - Other information
- Inactive ingredients
- Questions or comments?
-
Primatene MIST® Carton
Principal Display Panel Text:
Primatene MIST®
Epinephrine Inhalation Aerosol
0.125 mg per spray
Bronchodilator
For Oral
Inhalation OnlyFor TEMPORARY relief
of MILD symptoms
of INTERMITTENT asthmaSuspension:
Shake then Spray
into the air before
each inhalation.This item is
electronically
protected
NEW FORMULATION:
See Important Usage
Information on Insert
and on Side Panels160 Metered Sprays
Net Weight: 11.7g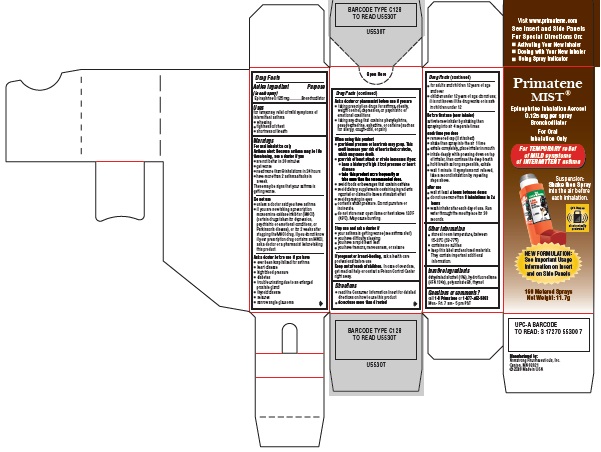
-
INGREDIENTS AND APPEARANCE
PRIMATENE MIST
epinephrine inhalation aerosolProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:17270-5530 Route of Administration RESPIRATORY (INHALATION) Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength EPINEPHRINE (UNII: YKH834O4BH) (EPINEPHRINE - UNII:YKH834O4BH) EPINEPHRINE 125 ug Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) NORFLURANE (UNII: DH9E53K1Y8) POLYSORBATE 80 (UNII: 6OZP39ZG8H) THYMOL (UNII: 3J50XA376E) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:17270-5530-0 1 in 1 CARTON 12/14/2018 1 160 in 1 INHALER; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA205920 12/14/2018 Labeler - Armstrong Pharmaceuticals, Inc. (809773794) Establishment Name Address ID/FEI Business Operations Armstrong Pharmaceuticals, Inc. 809773794 manufacture(17270-5530) , analysis(17270-5530)