Label: ALLERGY RELIEF- diphenhydramine hcl tablet, film coated
- NDC Code(s): 70000-0136-1, 70000-0136-2, 70000-0136-3
- Packager: Cardinal Health 110, LLC. DBA Leader
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated November 9, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each tablet)
- Purpose
- Uses
-
Warnings
Do not use
- to make a child sleepy
- with any other product containing diphenhydramine, even one used on skin
Ask a doctor before use if you have
- a breathing problem such as emphysema or chronic bronchitis
- glaucoma
- difficulty in urination due to enlargement of the prostate gland
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
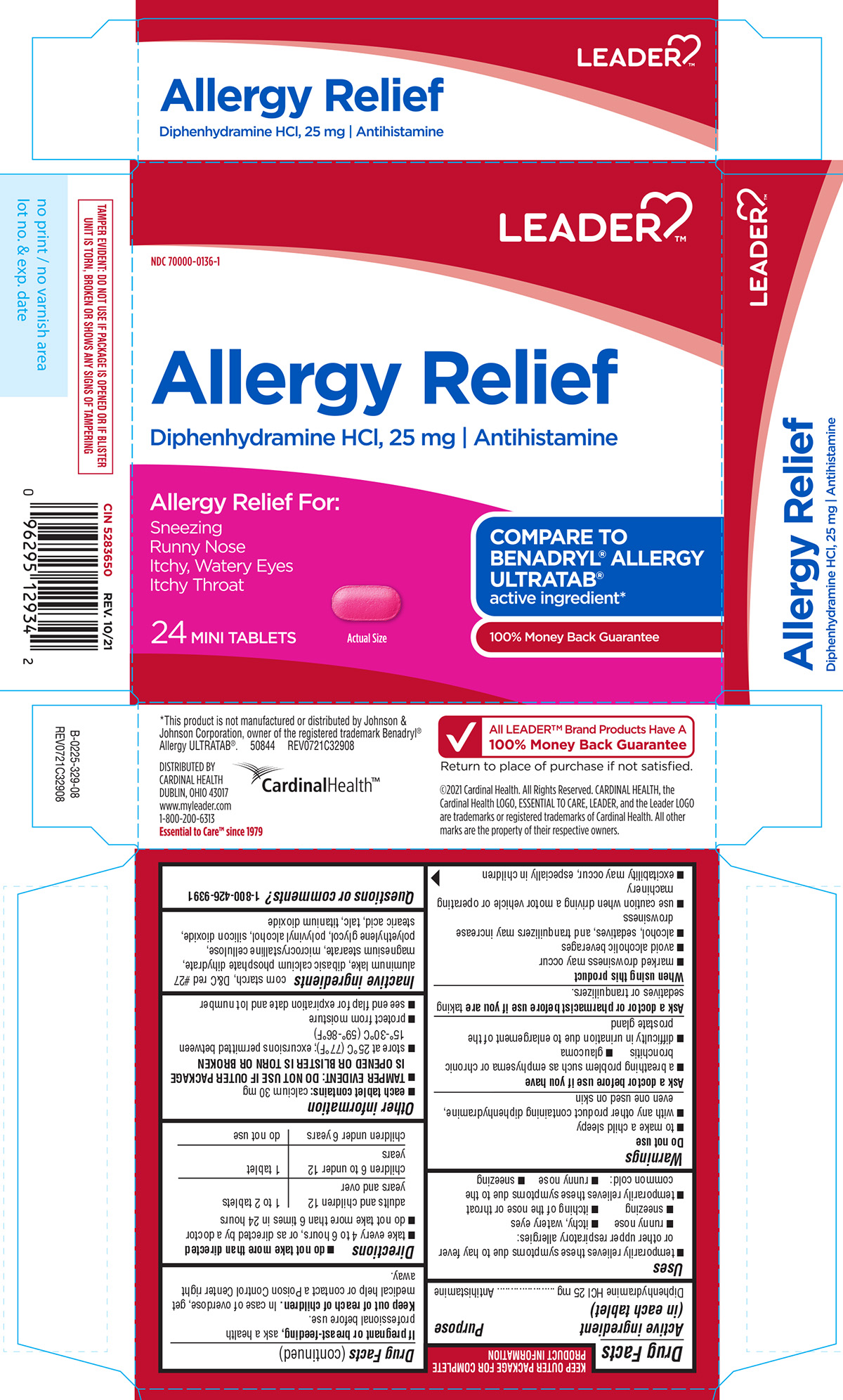
Principal Display Panel
LEADER™
NDC 70000-0136-1
Allergy Relief
Diphenhydramine HCl, 25 mg I Antihistamine
Allergy Relief For:
Sneezing
Runny Nose
Itchy, Watery Eyes
Itchy Throat24 MINI TABLETS
Actual Size
COMPARE TO BENADRYL® ALLERGY ULTRATAB®
active ingredient*
100% Money Back GuaranteeTAMPER EVIDENT: DO NOT USE IF PACKAGE IS OPENED OR IF BLISTER
UNIT IS TORN, BROKEN OR SHOWS ANY SIGNS OF TAMPERING*This product is not manufactured or distributed by Johnson &
Johnson Corporation, owner of the registered trademark Benadryl®
Allergy ULTRATAB®.50844 REV0721C32908
DISTRIBUTED BY
CARDINAL HEALTH
DUBLIN, OHIO 43017
www.myleader.com
1-800-200-6313
Essential to Care™ since 1979All LEADER™ Brand Products Have A
100% Money Back GuaranteeReturn to place of purchase if not satisfied.
©2021 Cardinal Health. All Rights Reserved. CARDINAL HEALTH, the
Cardinal Health LOGO, ESSENTIAL TO CARE, LEADER, and the Leader
LOGO are trademarks or registered trademarks of Cardinal Health. All other
marks are the property of their respective owners.
Leader 44-329
-
INGREDIENTS AND APPEARANCE
ALLERGY RELIEF
diphenhydramine hcl tablet, film coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:70000-0136 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIPHENHYDRAMINE HYDROCHLORIDE (UNII: TC2D6JAD40) (DIPHENHYDRAMINE - UNII:8GTS82S83M) DIPHENHYDRAMINE HYDROCHLORIDE 25 mg Inactive Ingredients Ingredient Name Strength STARCH, CORN (UNII: O8232NY3SJ) D&C RED NO. 27 ALUMINUM LAKE (UNII: ZK64F7XSTX) DIBASIC CALCIUM PHOSPHATE DIHYDRATE (UNII: O7TSZ97GEP) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) STEARIC ACID (UNII: 4ELV7Z65AP) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color pink Score no score Shape OVAL Size 11mm Flavor Imprint Code 44;329 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70000-0136-2 4 in 1 CARTON 03/02/1990 1 12 in 1 BLISTER PACK; Type 0: Not a Combination Product 2 NDC:70000-0136-3 1 in 1 CARTON 03/02/1990 2 100 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 3 NDC:70000-0136-1 2 in 1 CARTON 03/02/1990 3 12 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M012 03/02/1990 Labeler - Cardinal Health 110, LLC. DBA Leader (063997360) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 038154464 pack(70000-0136) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867837 manufacture(70000-0136) , pack(70000-0136) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867894 manufacture(70000-0136) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 868734088 manufacture(70000-0136) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 967626305 pack(70000-0136)