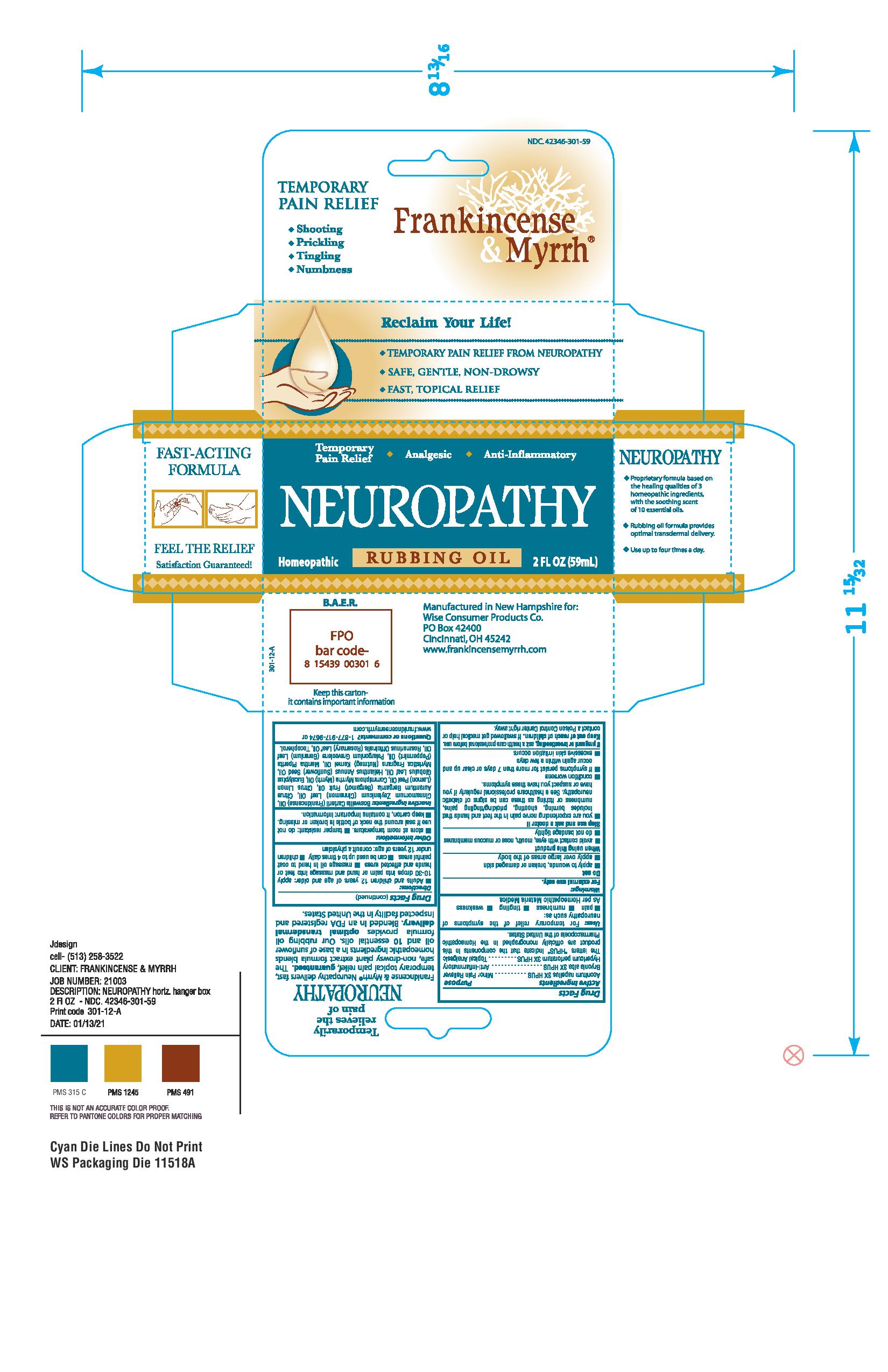
Label: FRANKINCENSE AND MYRRH NEUROPATHY- aconitum napellus, bryonia alba, hypericum perforatum oil
- NDC Code(s): 42346-301-59
- Packager: Wise Consumer Products
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated December 3, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients
- Purpose
- Uses:
-
Warnings:
For external use only.
When using this product
- avoid contact with eyes, mouth, nose or mucous membranes
- do not bandage tightly
Stop use and ask a doctor if
- you are experiencing nerve pain in the feet and hands that includes burning, shooting, prickling/tingling pains, numbness or itching as these can be signs of diabetic neuropathy. See a healthcare professional regularly if you have or suspect you have these symptoms.
- condition worsens
- if symptoms persist for more than 7 days or clear up and occur again within a few days
- excessive skin irritation occurs
- Directions:
- Other Information:
-
Inactive Ingredients:
Boswellia Carterii (Frankincense) Oil, Cinnamomum Zeylanicum (Cinnamon) Leaf Oil, Citrus Aurantium Bergamia (Bergamot) Fruit Oil, Citrus Limon (Lemon) Peel Oil, Commiphora Myrrha (Myrrh) Oil, Eucalyptus Globulus Leaf Oil, Helianthus Annuus (Sunflower) Seed Oil, Myristica Fragrans (Nutmeg) Kernel Oil, Mentha Piperita (Peppermint) Oil, Pelargonium Graveolens (Geranium) Leaf Oil, Rosmarinus Officinalis (Rosemary) Leaf Oil, Tocopherol.
- Questions or comments?
-
DESCRIPTION
Manufactured in New Hampshire for:
Wise Consumer Products Co.
PO Box 42400
Cincinnati, OH 45242
www.frankincensemyrrh.com
Keep this carton - it contains important information
Temporarily relieves the pain of NEUROPATHY
Frankincense and Myrrh TM Neuropathy is specifically formulated to deliver temporary and topical relief for Neuropathy. The plant extract formula blends homeopathic ingredients in a base of sunflower oil and 10 essential oils. Blended in an FDA registered and inspected facility in the United States. -
Principal Display Panel
NDC. 42346-300-59
TEMPORARY PAIN RELIEF
* Shooting
* Prickling
* Tingling
* Numbness
Frankincense and
MyrrhTM
Reclaim Your Life!
* TEMPORARY PAIN RELIEF FROM NEUROPATHY
* SAFE, GENTLE, NON-DROWSY
* FAST, TOPICAL RELIEF
Temporary Pain Relief - Analgesic - Anti-Inflammatory
NEUROPATHY
Homeopathic RUBBING OIL 2 FL OZ (59 mL)

-
INGREDIENTS AND APPEARANCE
FRANKINCENSE AND MYRRH NEUROPATHY
aconitum napellus, bryonia alba, hypericum perforatum oilProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:42346-301 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACONITUM NAPELLUS (UNII: U0NQ8555JD) (ACONITUM NAPELLUS - UNII:U0NQ8555JD) ACONITUM NAPELLUS 3 [hp_X] in 59 mL BRYONIA ALBA ROOT (UNII: T7J046YI2B) (BRYONIA ALBA ROOT - UNII:T7J046YI2B) BRYONIA ALBA ROOT 3 [hp_X] in 59 mL HYPERICUM PERFORATUM (UNII: XK4IUX8MNB) (HYPERICUM PERFORATUM - UNII:XK4IUX8MNB) HYPERICUM PERFORATUM 3 [hp_X] in 59 mL Inactive Ingredients Ingredient Name Strength BERGAMOT OIL (UNII: 39W1PKE3JI) CINNAMON OIL (UNII: E5GY4I6YCZ) EUCALYPTUS OIL (UNII: 2R04ONI662) FRANKINCENSE OIL (UNII: 67ZYA5T02K) GERANIUM OIL, ALGERIAN TYPE (UNII: 5Q1I94P4WG) LEMON OIL (UNII: I9GRO824LL) MYRRH OIL (UNII: H74221J5J4) NUTMEG OIL (UNII: Z1CLM48948) PEPPERMINT OIL (UNII: AV092KU4JH) ROSEMARY OIL (UNII: 8LGU7VM393) SUNFLOWER OIL (UNII: 3W1JG795YI) .ALPHA.-TOCOPHEROL (UNII: H4N855PNZ1) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:42346-301-59 1 in 1 CARTON 06/11/2013 1 59 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 06/11/2013 Labeler - Wise Consumer Products (006459643) Establishment Name Address ID/FEI Business Operations PJ Noyes Co. Inc. 018927392 manufacture(42346-301)