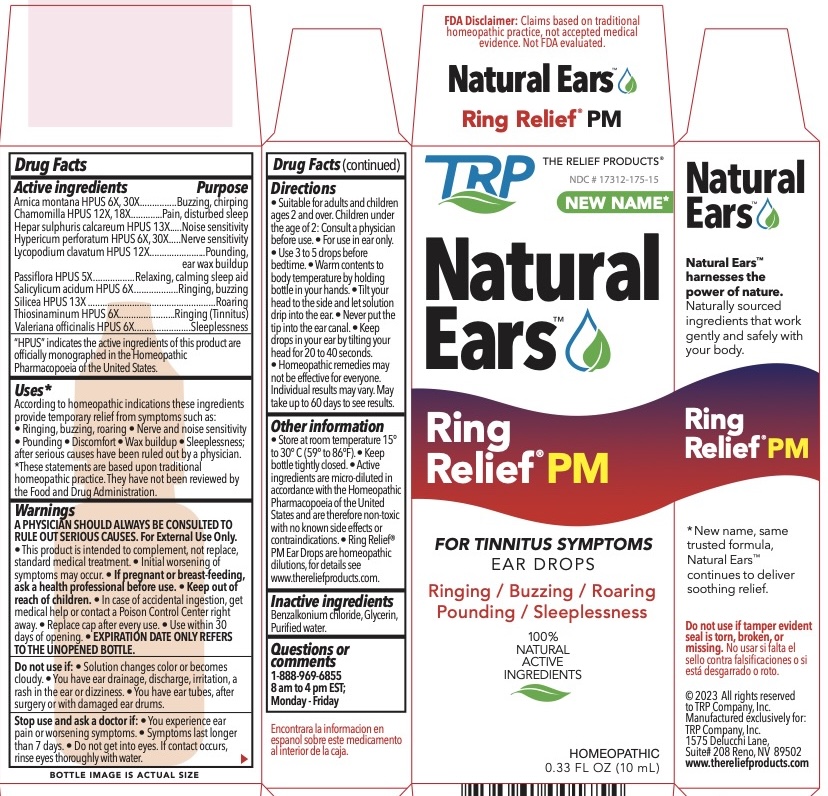
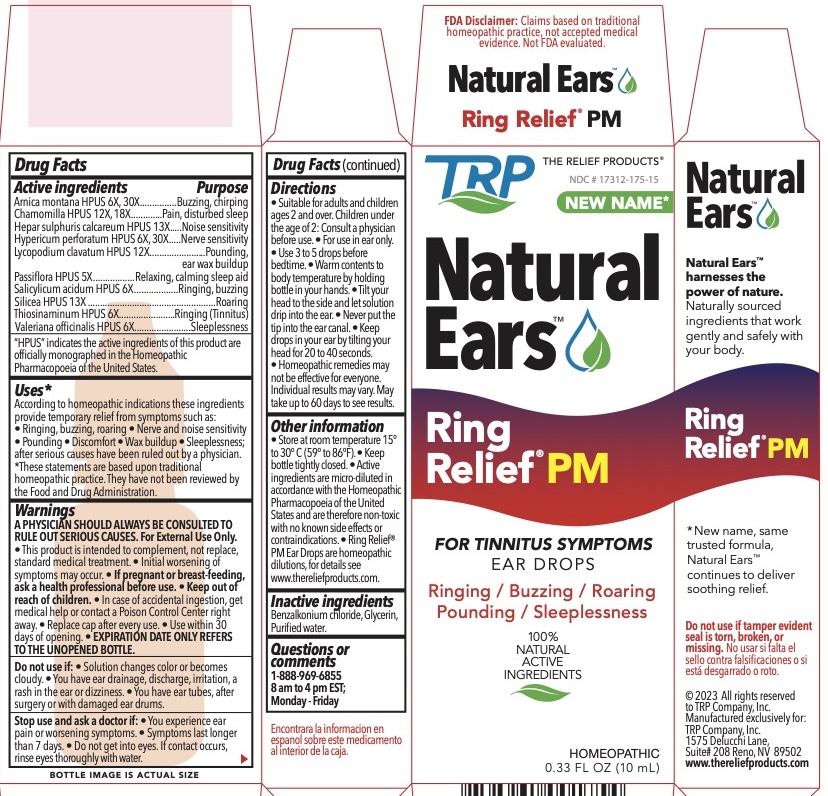
Label: RING RELIEF PM NIGHTTIME FORMULA- arnica montana, calcium sulfide, hypericum perforatum, lycopodium clavatum spore, thiosinaminum, mercurius solubilis, salicylic acid, silicon dioxide, allylthiourea liquid
- NDC Code(s): 17312-175-15
- Packager: TRP Company
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated March 5, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
ACTIVE INGREDIENT
Arnica montana HPUS 6X, 30X................Buzzing, chirping
Chamomilla HPUS 12X, 18X....................Pain, disturbed sleep
Hepar sulphuris calcareum HPUS 13X....Noise sensitivity
Hypericum perforatum HPUS 6X, 30X.....Nerve sensitivity
Lycopodium clavatum HPUS 12X............Pounding, ear wax buildup
Passiflora HPUS 5X.................................Relaxing, calming sleep aid
Salicylicum acidum HPUS 6X..................Ringing, buzzing
Silicea HPUS 13X....................................Roaring
Thiosinaminum HPUS 6X........................Ringing (Tinnitus)
Valeriana officinalis HPUS 6X..................Sleeplessness"HPUS" indicates the active ingredients are in the Homeopathic Pharmacopoeia of the United States.
-
PURPOSE
Arnica montana HPUS- Buzzing, chirping
Chamomilla HPUS - Pain, disturbed sleep
Hepar sulphuris calcareum HPUS - Noise sensitivity
Hypericum perforatum HPUS - Nerve sensitivity
Lycopodium clavatum HPUS - Pounding, ear wax buildup
Passiflora HPUS - Relaxing, calming sleep aid
Salicylicum acidum HPUS - Ringing, buzzing
Silicea HPUS - Roaring
Thiosinaminum HPUS - Ringing (Tinnitus)
Valeriana officinalis HPUS - Sleeplessness -
INDICATIONS & USAGE
Uses:*
According to homeopathic indications these ingredients provide temporary relief from symptoms such as: • Ringing, buzzing, roaring • Nerve and noise sensitivity • Pounding • Discomfort • Wax buildup • Sleeplessness; after serious causes have been ruled out by a physician.
*These statements are based upon traditional homeopathic practice. They have not been reviewed by the Food and Drug Administration. - WARNINGS
- PREGNANCY OR BREAST FEEDING
- DO NOT USE
- Keep out of reach of children
-
DOSAGE & ADMINISTRATION
Directions:
• Suitable for adults and children ages 2 and over. Children under the age of 2: Consult a physician before use. • For use in ear only. • Use 3 to 5 drops before bedtime. • Warm contents to body temperature by holding bottle in your hands. • Tilt your head to the side and let solution drip into the ear. • Never put the tip into the ear canal. • Keep drops in your ear by tilting your head for 20 to 40 seconds. • Homeopathic remedies may not be effective for everyone. Individual results may vary. May take up to 60 days to see results. -
SPL UNCLASSIFIED SECTION
Other information:
• Store at room temperature 15° to 30° C (59° to 86°F). • Keep bottle tightly closed. • Active ingredients are micro-diluted in accordance with the Homeopathic Pharmacopoeia of the United States and are therefore non-toxic with no known side effects or contraindications. • Ring Relief® PM Ear Drops are homeopathic dilutions, for details see www.thereliefproducts.com.
- INACTIVE INGREDIENT
- Questions or comments?
- Stop Use
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
RING RELIEF PM NIGHTTIME FORMULA
arnica montana, calcium sulfide, hypericum perforatum, lycopodium clavatum spore, thiosinaminum, mercurius solubilis, salicylic acid, silicon dioxide, allylthiourea liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:17312-175 Route of Administration AURICULAR (OTIC) Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ARNICA MONTANA (UNII: O80TY208ZW) (ARNICA MONTANA - UNII:O80TY208ZW) ARNICA MONTANA 6 [hp_X] in 1 mL CALCIUM SULFIDE (UNII: 1MBW07J51Q) (CALCIUM SULFIDE - UNII:1MBW07J51Q) CALCIUM SULFIDE 13 [hp_X] in 1 mL HYPERICUM PERFORATUM (UNII: XK4IUX8MNB) (HYPERICUM PERFORATUM - UNII:XK4IUX8MNB) HYPERICUM PERFORATUM 6 [hp_X] in 1 mL LYCOPODIUM CLAVATUM SPORE (UNII: C88X29Y479) (LYCOPODIUM CLAVATUM SPORE - UNII:C88X29Y479) LYCOPODIUM CLAVATUM SPORE 12 [hp_X] in 1 mL MATRICARIA CHAMOMILLA (UNII: G0R4UBI2ZZ) (MATRICARIA CHAMOMILLA - UNII:G0R4UBI2ZZ) MATRICARIA CHAMOMILLA 12 [hp_X] in 1 mL SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 6 [hp_X] in 1 mL SILICON DIOXIDE (UNII: ETJ7Z6XBU4) (SILICON DIOXIDE - UNII:ETJ7Z6XBU4) SILICON DIOXIDE 13 [hp_X] in 1 mL VALERIAN (UNII: JWF5YAW3QW) (VALERIAN - UNII:JWF5YAW3QW) VALERIAN 6 [hp_X] in 1 mL ALLYLTHIOUREA (UNII: 706IDJ14B7) (ALLYLTHIOUREA - UNII:706IDJ14B7) ALLYLTHIOUREA 6 [hp_X] in 1 mL PASSIFLORA INCARNATA FLOWER (UNII: K8F3G29S6Z) (PASSIFLORA INCARNATA FLOWER - UNII:K8F3G29S6Z) PASSIFLORA INCARNATA FLOWER 5 [hp_X] in 1 mL Inactive Ingredients Ingredient Name Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) GLYCERIN (UNII: PDC6A3C0OX) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:17312-175-15 1 in 1 PACKAGE 09/19/2019 1 10 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 09/19/2019 Labeler - TRP Company (105185719) Registrant - TRP Company (105185719)