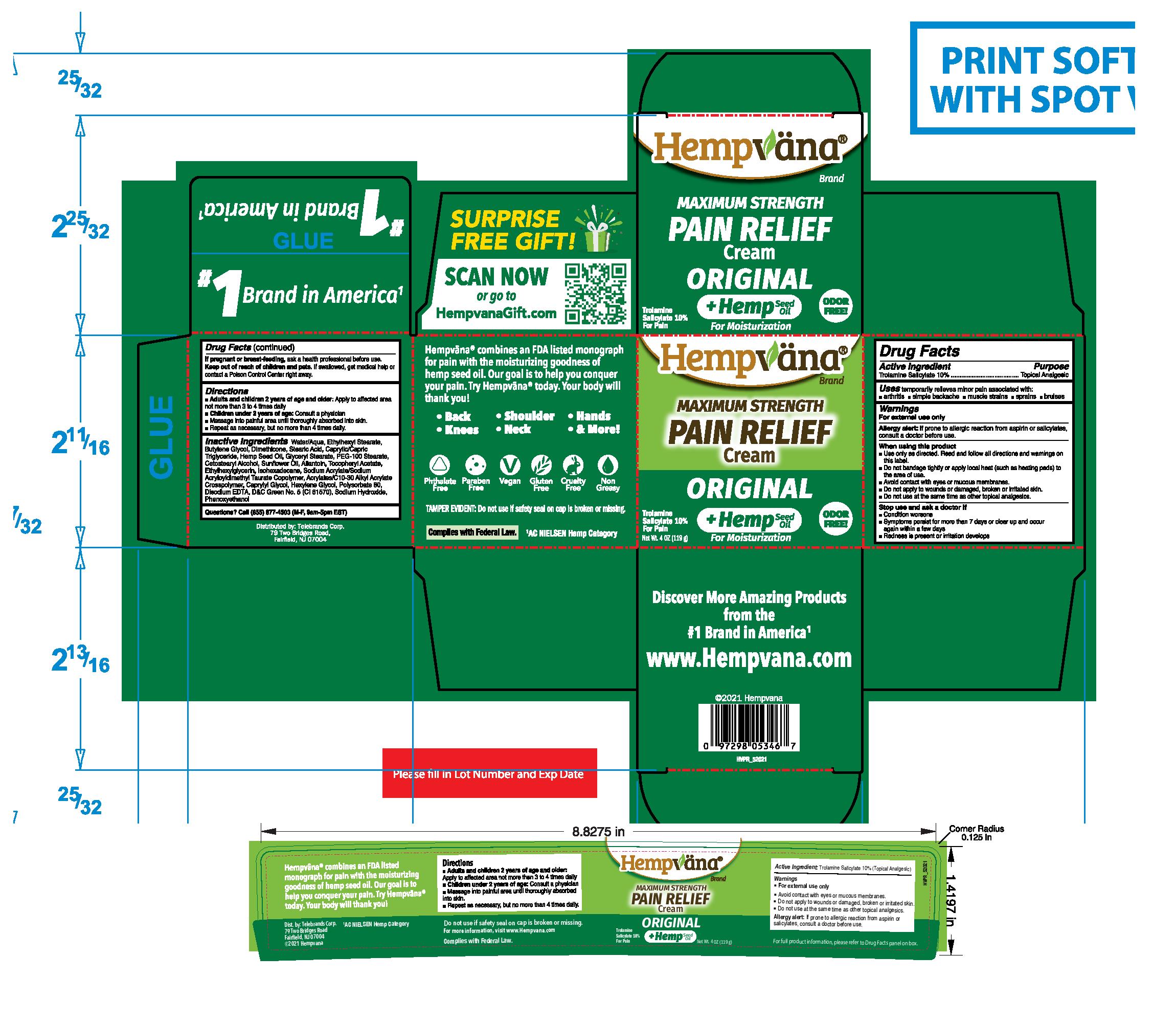
Label: HEMPVANA PAIN RELIEF- trolamine salicylate cream
- NDC Code(s): 73287-001-01, 73287-001-02
- Packager: Telebrands Corp
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated February 22, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active Ingredient:
- Purpose
- Uses
-
Warnings
For external use only
Allergy alert:If prone to allergic reaction from aspirin or salicylates, consult a doctor before use.
When using this product
- Use only as directed. Read and follow all directions and warnings on this label.
- Do not bandage tightly or apply local heat (such as heating pads) to the area of use.
- Avoid contact with eyes or mucous membranes.
- Do not apply to wounds or damaged, broken or irritated skin.
- Do not use at the same time as other topical analgesics.
- Directions
-
Inactive Ingredients
Water/Aqua, Ethylhexyl Stearate, Butylene Glycol, Dimethicone, Stearic Acid, Caprylic/Capric Triglyceride, Hemp Seed Oil (Hemp Seed Oil), Glyceryl Stearate, PEG-100 Stearate, Cetostearyl Alcohol, Sunflower Oil, Allantoin, Tocopheryl Acetate, Ethylhexylglycerin, Isohexadecane, Sodium Acrylate/Sodium Acryloyldimethyl Taurate Copolymer, Acrylates/C10-30 Alkyl Acrylate Crosspolymer, Caprylyl Glycol, Hexylene Glycol, Polysorbate 80, Disodium EDTA, D&C Green No. 5 (CI 61570), Sodium Hydroxide, Phenoxyethanol
- Questions?
-
DESCRIPTION
Hempvana ®Pain Relief Cream absorbs quickly and goes to work instantly. Its unique formula includes a proven active ingredient plus the moisturizing benefit of cold pressed hemp seed* extract for soothing relief to back, neck, shoulders, feet, wrists, calves and more.
- Maximum Strength
- Odor Free
- Non-Irritating
- No Burning
- Non Greasy
TAMPER EVIDENT: Do not use if safety seal on cap is broken or missing.
For more information, visit www.HempvanaStore.com
*Complies with Federal Law. Hemp seed extract in the form of oil made by cold-pressing industrial hemp seeds.
cultivar.
Dist. by: Telebrands Corp.
79 Two Bridges Road,
Fairfield, NJ 07004
- Packaging
-
INGREDIENTS AND APPEARANCE
HEMPVANA PAIN RELIEF
trolamine salicylate creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:73287-001 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TROLAMINE SALICYLATE (UNII: H8O4040BHD) (SALICYLIC ACID - UNII:O414PZ4LPZ) TROLAMINE SALICYLATE 10 g in 100 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) ETHYLHEXYL STEARATE (UNII: EG3PA2K3K5) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) DIMETHICONE (UNII: 92RU3N3Y1O) STEARIC ACID (UNII: 4ELV7Z65AP) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) CANNABIS SATIVA SEED OIL (UNII: 69VJ1LPN1S) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) PEG-100 STEARATE (UNII: YD01N1999R) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) SUNFLOWER OIL (UNII: 3W1JG795YI) ALLANTOIN (UNII: 344S277G0Z) .ALPHA.-TOCOPHEROL ACETATE, DL- (UNII: WR1WPI7EW8) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) ISOHEXADECANE (UNII: 918X1OUF1E) SODIUM ACRYLATE/SODIUM ACRYLOYLDIMETHYLTAURATE COPOLYMER (4000000 MW) (UNII: 1DXE3F3OZX) CARBOMER COPOLYMER TYPE A (ALLYL PENTAERYTHRITOL CROSSLINKED) (UNII: 71DD5V995L) CAPRYLYL GLYCOL (UNII: 00YIU5438U) HEXYLENE GLYCOL (UNII: KEH0A3F75J) POLYSORBATE 80 (UNII: 6OZP39ZG8H) EDETATE DISODIUM ANHYDROUS (UNII: 8NLQ36F6MM) D&C GREEN NO. 5 (UNII: 8J6RDU8L9X) SODIUM HYDROXIDE (UNII: 55X04QC32I) PHENOXYETHANOL (UNII: HIE492ZZ3T) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:73287-001-01 1 in 1 CARTON 01/10/2019 1 119 g in 1 JAR; Type 0: Not a Combination Product 2 NDC:73287-001-02 524 g in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 02/22/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 01/10/2019 Labeler - Telebrands Corp (177266558) Establishment Name Address ID/FEI Business Operations Neutraderm, Inc. 146224444 manufacture(73287-001)