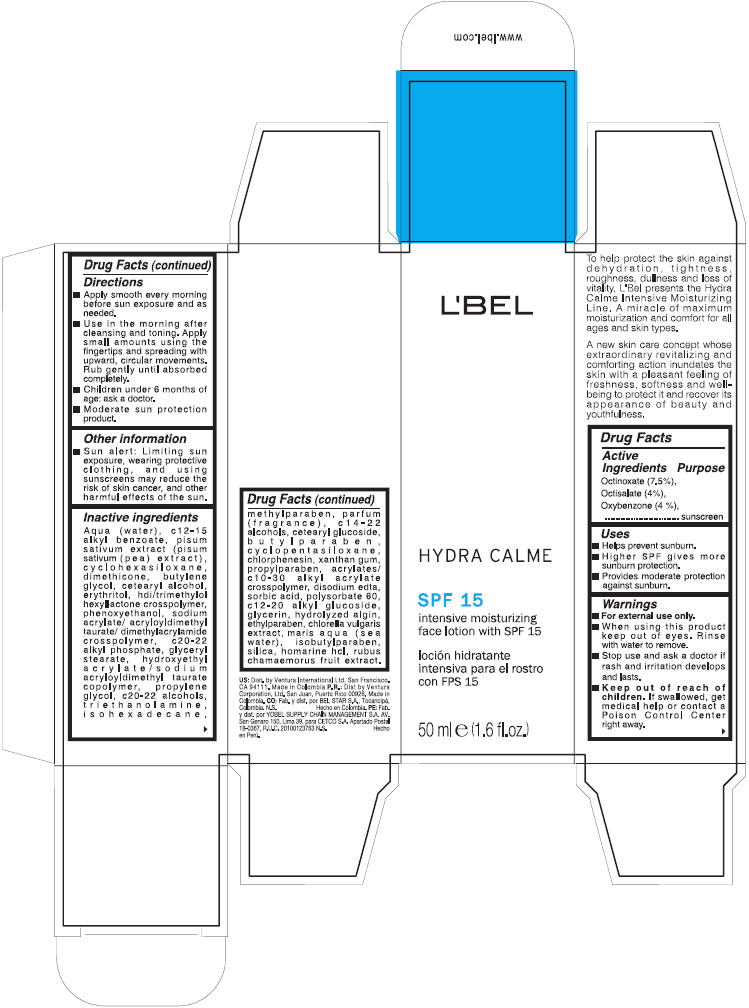
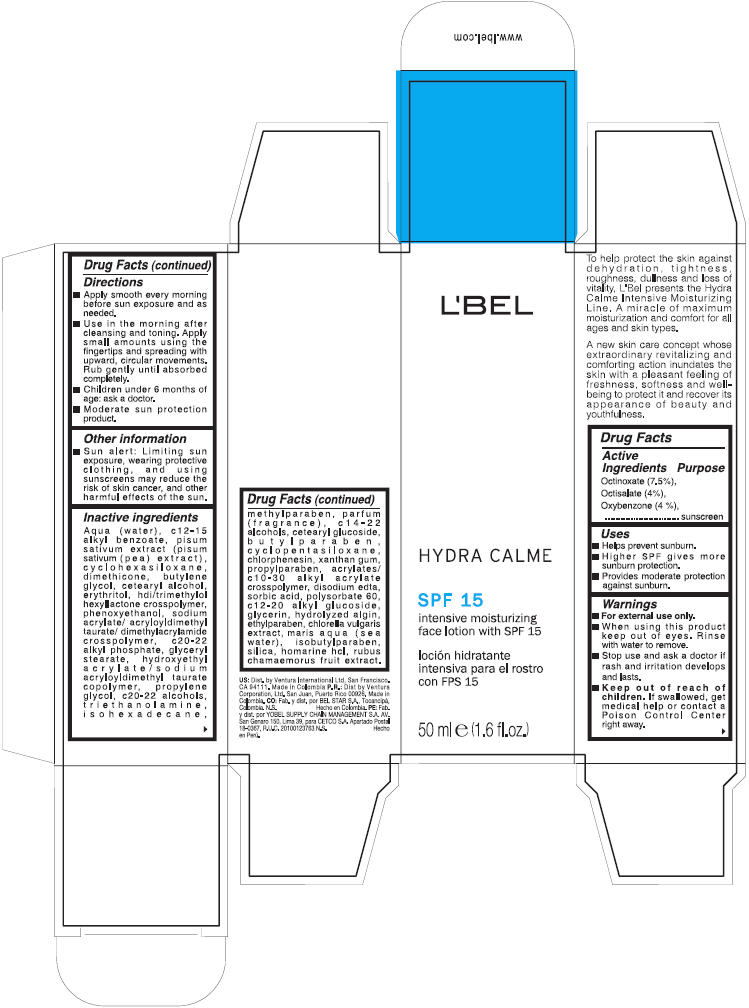
Label: LBEL- octinoxate, octisalate, and oxybenzone lotion
-
Contains inactivated NDC Code(s)
NDC Code(s): 14783-400-01, 14783-400-02, 14783-400-03, 14783-400-04, view more14783-400-05 - Packager: Ventura International Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated September 6, 2011
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- Uses
- Warnings
-
Directions
- Apply smoothly every morning before sun exposure and as needed.
- Use in the morning after cleansing and toning. Apply small amounts using the fingertips and spreading with upward, circular movements. Rub gently until absorbed completely.
- Children under 6 months of age: ask a doctor.
- Moderate sun protection product
- Other information
-
Inactive ingredients
aqua (water), c12-15 alkyl benzoate, pisum sativum extract (pisum sativum (pea) extract), cyclohexasiloxane, dimethicone, butylene glycol, cetearyl alcohol, erythritol, hdi/trimethylol hexyllactone crosspolymer, phenoxyethanol, sodium acrylate/ acryloyldimethyltaurate/ dimethylacrylamide crosspolymer, c20-22 alkyl phosphate, glyceryl stearate, hydroxyethyl acrylate/ sodium acryloyldimethyl taurate copolymer, propylene glycol, c20-22 alcohols, triethanolamine, isohexadecane, methylparaben, parfum (fragrance), c14-22 alcohols, cetearyl glucoside, butylparaben, cyclopentasiloxane, chlorphenesin, xanthan gum, propylparaben, acrylates/ c10-30 alkyl acrylate crosspolymer, disodium edta, sorbic acid, polysorbate 60, c12-20 alkyl glucoside, glycerin, hydrolyzed algin, ethylparaben, chlorella vulgaris extract, maris aqua (sea water), isobutylparaben, silica, homarine hcl, rubus chamaemorus fruit extract
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 50 ml Bottle Carton
-
INGREDIENTS AND APPEARANCE
LBEL HYDRA CALME SPF 15
octinoxate, octisalate, and oxybenzone lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:14783-400 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Octinoxate (UNII: 4Y5P7MUD51) (Octinoxate - UNII:4Y5P7MUD51) Octinoxate 0.075 g in 1 mL Octisalate (UNII: 4X49Y0596W) (Octisalate - UNII:4X49Y0596W) Octisalate 0.04 g in 1 mL Oxybenzone (UNII: 95OOS7VE0Y) (Oxybenzone - UNII:95OOS7VE0Y) Oxybenzone 0.04 g in 1 mL Inactive Ingredients Ingredient Name Strength water (UNII: 059QF0KO0R) alkyl (c12-15) benzoate (UNII: A9EJ3J61HQ) pea (UNII: W4X7H8GYFM) cyclomethicone 6 (UNII: XHK3U310BA) dimethicone (UNII: 92RU3N3Y1O) butylene glycol (UNII: 3XUS85K0RA) cetostearyl alcohol (UNII: 2DMT128M1S) erythritol (UNII: RA96B954X6) phenoxyethanol (UNII: HIE492ZZ3T) c20-22 alkyl phosphate (UNII: L4VKP0Y7RP) glyceryl monostearate (UNII: 230OU9XXE4) propylene glycol (UNII: 6DC9Q167V3) c20-22 alcohols (UNII: O4M0347C6A) trolamine (UNII: 9O3K93S3TK) isohexadecane (UNII: 918X1OUF1E) methylparaben (UNII: A2I8C7HI9T) c14-22 alcohols (UNII: B1K89384RJ) cetearyl glucoside (UNII: 09FUA47KNA) butylparaben (UNII: 3QPI1U3FV8) cyclomethicone 5 (UNII: 0THT5PCI0R) chlorphenesin (UNII: I670DAL4SZ) xanthan gum (UNII: TTV12P4NEE) propylparaben (UNII: Z8IX2SC1OH) edetate disodium (UNII: 7FLD91C86K) sorbic acid (UNII: X045WJ989B) polysorbate 60 (UNII: CAL22UVI4M) c12-20 alkyl glucoside (UNII: K67N5Z1RUA) glycerin (UNII: PDC6A3C0OX) ethylparaben (UNII: 14255EXE39) chlorella vulgaris (UNII: RYQ4R60M02) isobutylparaben (UNII: 0QQJ25X58G) silicon dioxide (UNII: ETJ7Z6XBU4) rubus chamaemorus fruit (UNII: 74CW8G72Q6) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:14783-400-02 1 in 1 BOX 1 NDC:14783-400-01 50 mL in 1 TUBE 2 NDC:14783-400-04 1 in 1 BOX 2 NDC:14783-400-03 5 mL in 1 TUBE 3 NDC:14783-400-05 1 mL in 1 PACKET Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH NOT FINAL part352 09/01/2011 Labeler - Ventura International Ltd. (603192787) Establishment Name Address ID/FEI Business Operations Bel Star, S.A. 880160197 MANUFACTURE Establishment Name Address ID/FEI Business Operations Yobel Supply Chain Management S.A. 934116930 MANUFACTURE