Label: 62% ETHYL ALCOHOL ANTISEPTIC NASAL- ethyl alcohol swab
- NDC Code(s): 53329-032-29, 53329-032-78
- Packager: Medline Industries, LP
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated November 30, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
-
Warnings
for external use only
flammable, keep away from open flame
Stop and ask a doctor if
- redness, irritation, swelling or pain persists or increases
- infection occurs
-
Directions
NASAL APPLICATION:
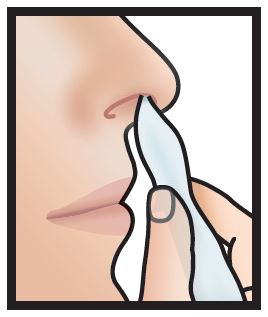
1. Use a tissue to clean the inside of both nostrils, including the inside tip of nostril. Discard.
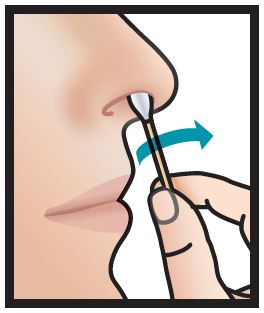
2. Insert right swab comfortably into right nostril and rotate for 30 seconds, covering all surfaces. Discard swabstick.
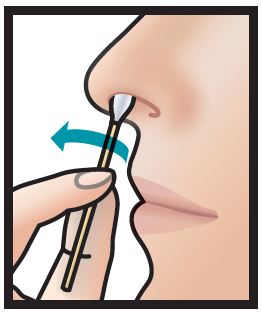
3. Using left swab, repeat step 2 with left nostril.
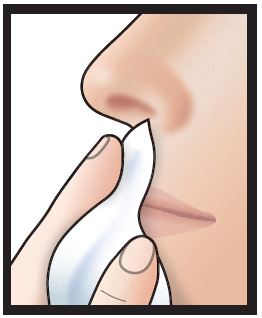
4. Do not blow nose. If solution drips, gently wipe with a tissue.
- Other Information
- Inactive ingredients
- Manufacturing Information
- Package Label
-
INGREDIENTS AND APPEARANCE
62% ETHYL ALCOHOL ANTISEPTIC NASAL
ethyl alcohol swabProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:53329-032 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 62 mL in 100 mL Inactive Ingredients Ingredient Name Strength MENTHOL (UNII: L7T10EIP3A) WATER (UNII: 059QF0KO0R) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) BENZYL ALCOHOL (UNII: LKG8494WBH) ISOPROPYL ALCOHOL (UNII: ND2M416302) POLYAMINOPROPYL BIGUANIDE (UNII: DT9D8Z79ET) ASCORBYL PALMITATE (UNII: QN83US2B0N) CASTOR OIL (UNII: D5340Y2I9G) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:53329-032-78 6 mL in 1 PACKET; Type 0: Not a Combination Product 09/10/2019 2 NDC:53329-032-29 50 in 1 BOX 09/10/2019 2 6 mL in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) 09/10/2019 Labeler - Medline Industries, LP (025460908) Registrant - Medline Industries, LP (025460908)




