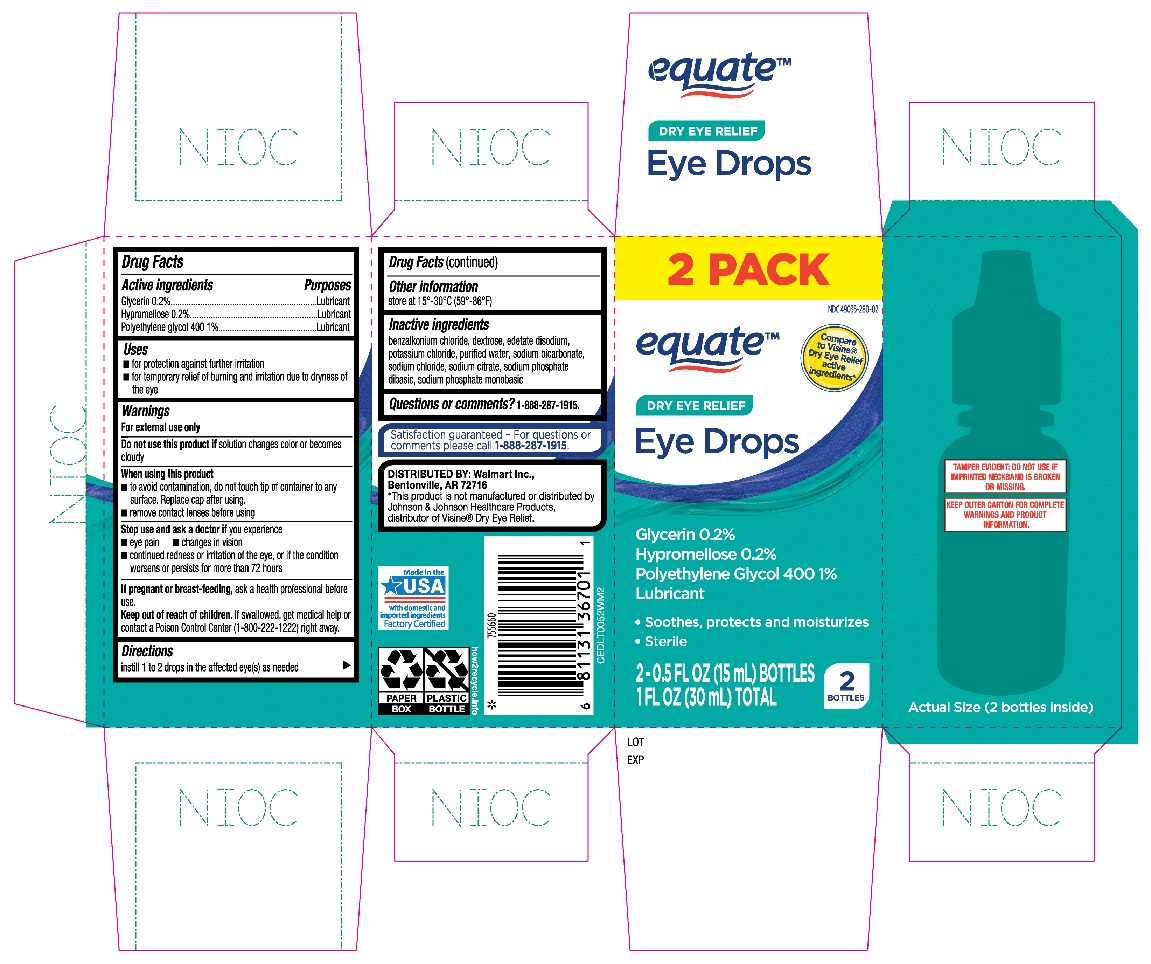
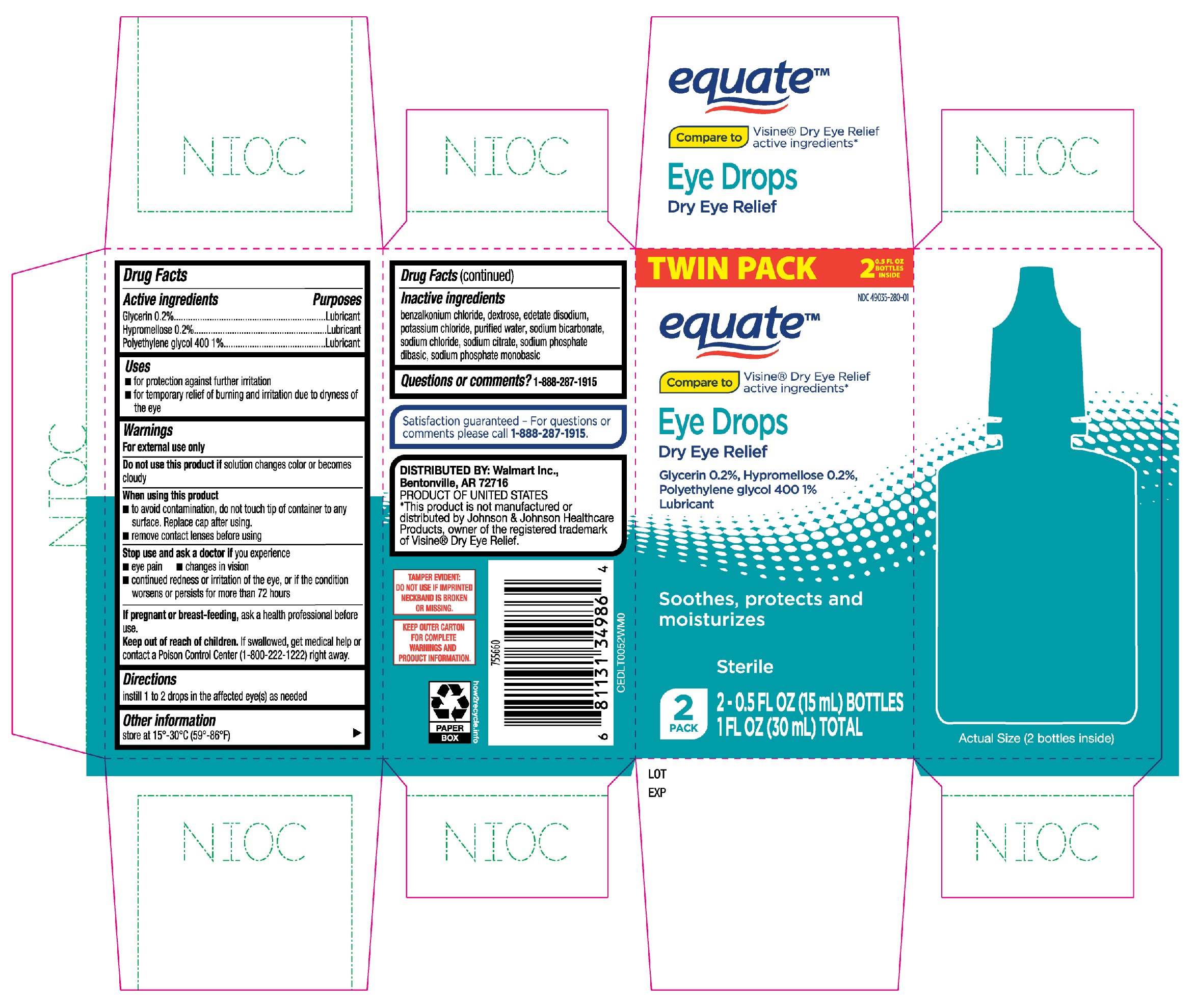
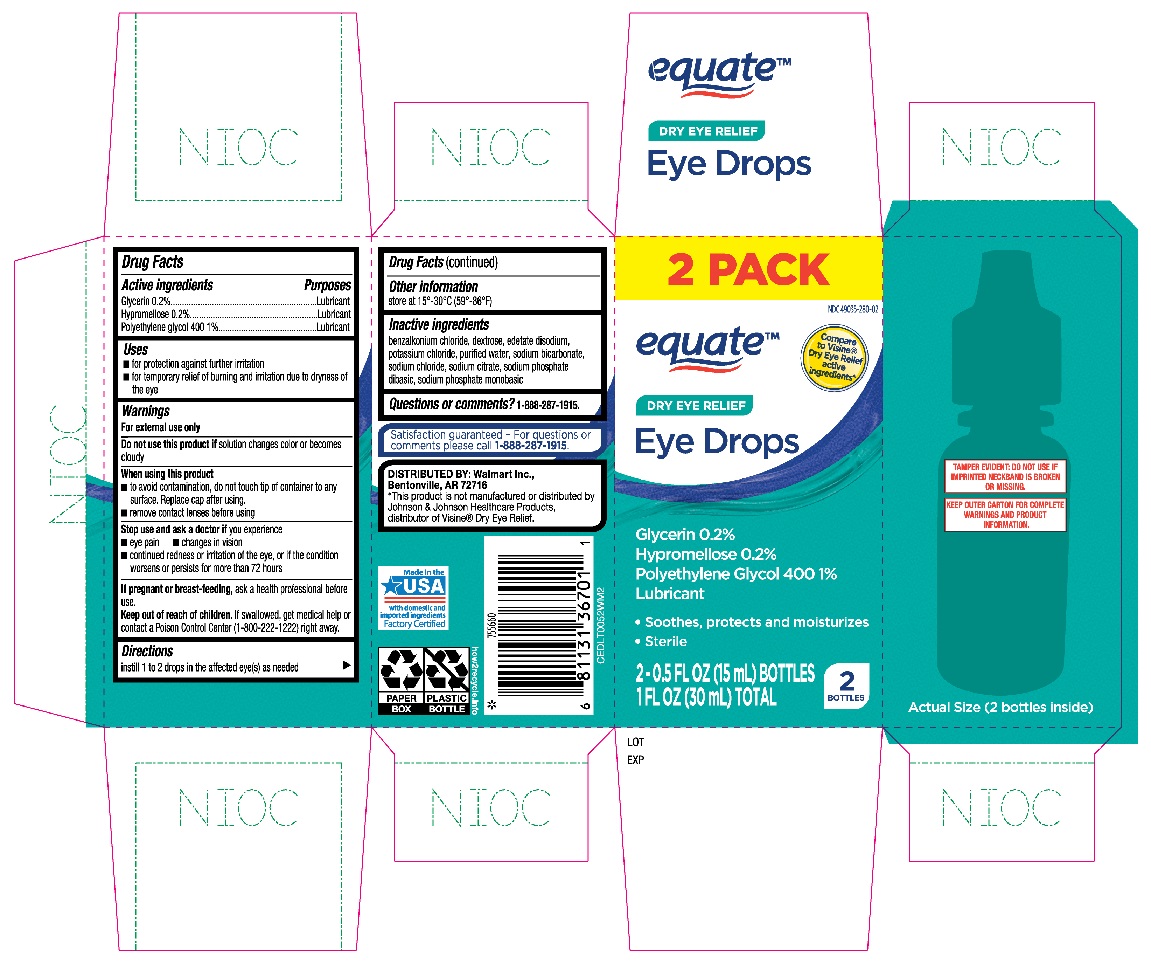
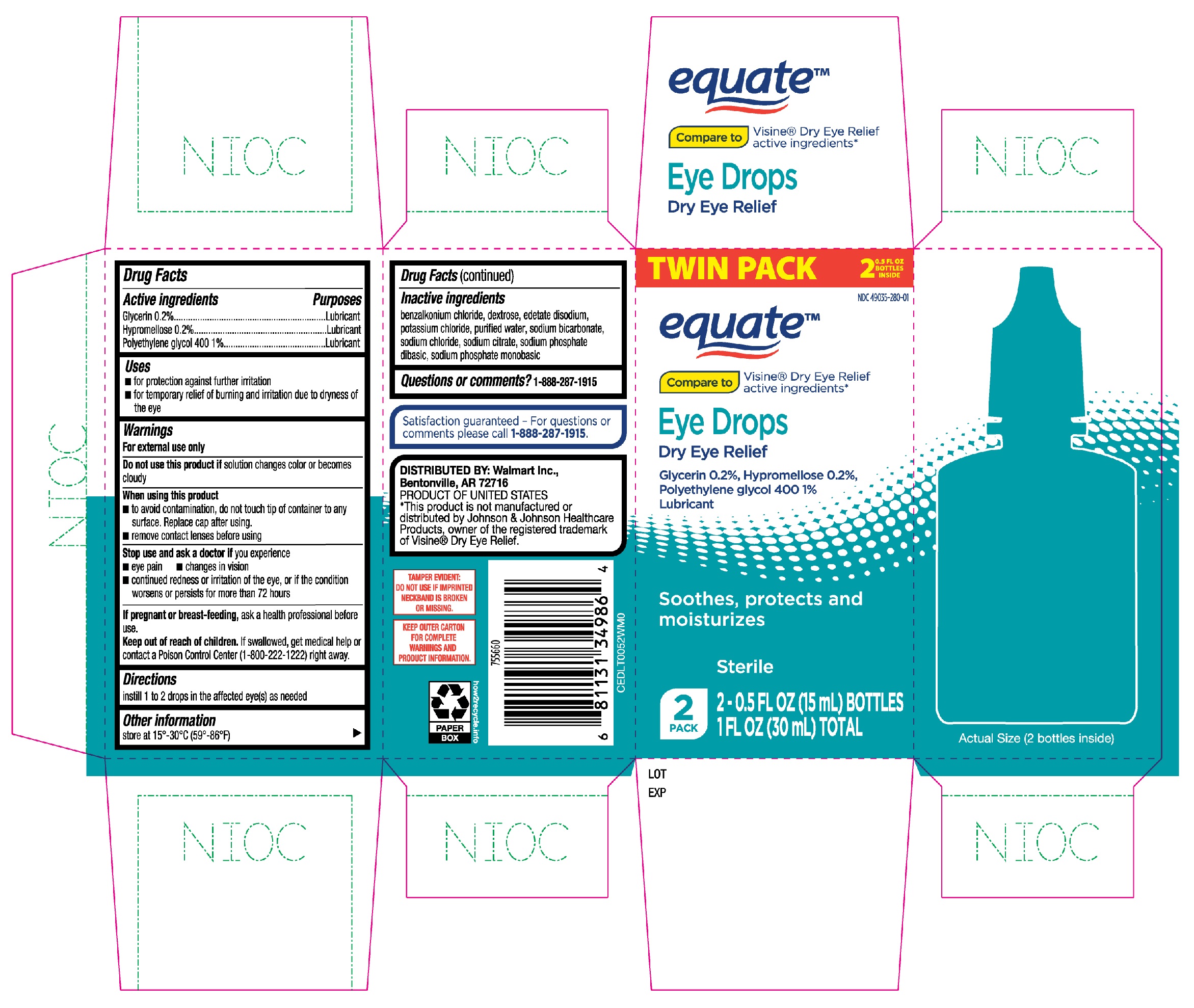
Label: EQUATE EYE DROPS DRY EYE RELIEF- glycerin, hypromellose, polyethylene glycol 400 solution/ drops
- NDC Code(s): 49035-280-01, 49035-280-02
- Packager: Wal-Mart Stores, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 23, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients
- Purposes
- Uses
-
Warnings
For external use only
When using this product
- to avoid contamination, do not touch tip of container to any surface. Replace cap after using.
- remove contact lenses before using
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
- Equate Dry Eye Relief Eye Drops 2-0.5oz bottles
- Equate Eye Drops Dry Eye Relief 2-0.5oz bottles
-
INGREDIENTS AND APPEARANCE
EQUATE EYE DROPS DRY EYE RELIEF
glycerin, hypromellose, polyethylene glycol 400 solution/ dropsProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49035-280 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GLYCERIN (UNII: PDC6A3C0OX) (GLYCERIN - UNII:PDC6A3C0OX) GLYCERIN 0.2 g in 100 mL HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) (HYPROMELLOSE, UNSPECIFIED - UNII:3NXW29V3WO) HYPROMELLOSE, UNSPECIFIED 0.2 g in 100 mL POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) (POLYETHYLENE GLYCOL, UNSPECIFIED - UNII:3WJQ0SDW1A) POLYETHYLENE GLYCOL 400 1 g in 100 mL Inactive Ingredients Ingredient Name Strength SODIUM PHOSPHATE, MONOBASIC, ANHYDROUS (UNII: KH7I04HPUU) POTASSIUM CHLORIDE (UNII: 660YQ98I10) SODIUM BICARBONATE (UNII: 8MDF5V39QO) SODIUM PHOSPHATE DIBASIC DIHYDRATE (UNII: 94255I6E2T) BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) DEXTROSE (UNII: IY9XDZ35W2) EDETATE DISODIUM (UNII: 7FLD91C86K) WATER (UNII: 059QF0KO0R) SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM CITRATE (UNII: 1Q73Q2JULR) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49035-280-01 2 in 1 BOX 11/01/2019 1 15 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product 2 NDC:49035-280-02 2 in 1 BOX 02/01/2021 2 15 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M018 11/01/2019 Labeler - Wal-Mart Stores, Inc. (051957769) Registrant - KC Pharmaceuticals, Inc. (174450460) Establishment Name Address ID/FEI Business Operations KC Pharmaceutical, Inc. 174450460 manufacture(49035-280) , pack(49035-280) , label(49035-280)