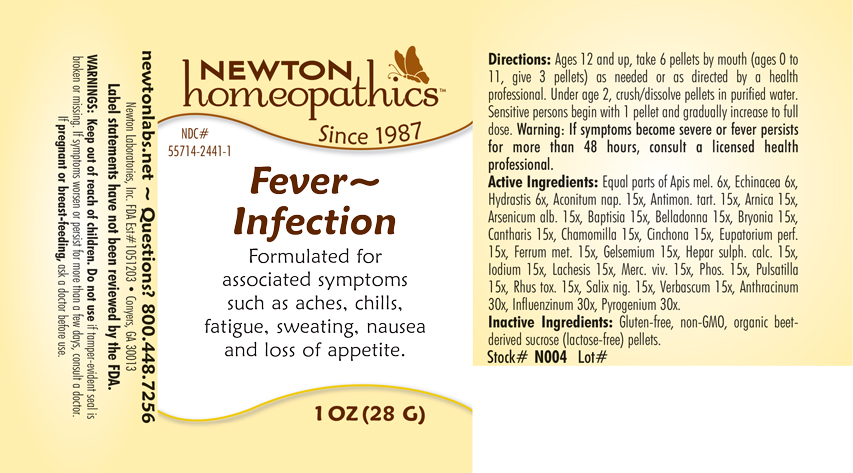
Label: FEVER - INFECTION- anthracinum, influenza a virus, influenza b virus, pyrogenium, aconitum nap., antimon. tart., arnica, arsenicum alb., baptisia, belladonna, bryonia, cantharis, chamomilla, cinchona, eupatorium perf., ferrum metallicum, gelsemium, hepar sulph.calc., iodium, lachesis, merc. viv., phosphorus, pulsatilla, rhus toxicodendron, salix nigra, verbascum, apis mel., echinacea, hydrastis pellet
- NDC Code(s): 55714-2441-1
- Packager: Newton Laboratories, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated January 9, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- INDICATIONS & USAGE SECTION
-
DOSAGE & ADMINISTRATION SECTION
Directions: Ages 12 and up, take 6 pellets mouth (ages 0 to 11, give 3 pellets) as needed or as directed by a health professional. Under age 2, crush or dissolve pellets in purfied water. Sensitive persons begin with 1 pellet and gradually increase to full dose. Warning: If symptoms become severe or fever persists for more than 48 hours, consult a licensed health professional.
-
OTC - ACTIVE INGREDIENT SECTION
Equal parts of Apis mel. 6x, Echinacea 6x, Hydrastis 6x, Aconitum nap. 15x, Antimon. tart. 15x, Arnica 15x, Arsenicum alb. 15x, Baptisia 15x, Belladonna 15x, Bryonia 15x, Cantharis 15x, Chamomilla 15x, Cinchona 15x, Eupatorium perf. 15x, Ferrum metallicum 15x, Gelsemium 15x, Hepar sulph.calc. 15x, Iodium 15x, Lachesis 15x, Merc. viv. 15x, Phosphorus 15x, Pulsatilla 15x, Rhus toxicodendron 15x, Salix nigra 15x, Verbascum 15x, Anthracinum 30x, Influenzinum 30x, Pyrogenium 30x.
- OTC - PURPOSE SECTION
- INACTIVE INGREDIENT SECTION
- QUESTIONS SECTION
- WARNINGS SECTION
- OTC - PREGNANCY OR BREAST FEEDING SECTION
- OTC - KEEP OUT OF REACH OF CHILDREN SECTION
- PACKAGE LABEL
-
INGREDIENTS AND APPEARANCE
FEVER - INFECTION
anthracinum, influenza a virus, influenza b virus, pyrogenium, aconitum nap., antimon. tart., arnica, arsenicum alb., baptisia, belladonna, bryonia, cantharis, chamomilla, cinchona, eupatorium perf., ferrum metallicum, gelsemium, hepar sulph.calc., iodium, lachesis, merc. viv., phosphorus, pulsatilla, rhus toxicodendron, salix nigra, verbascum, apis mel., echinacea, hydrastis pelletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:55714-2441 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BACILLUS ANTHRACIS IMMUNOSERUM RABBIT (UNII: 41LZ22DD4N) (BACILLUS ANTHRACIS IMMUNOSERUM RABBIT - UNII:41LZ22DD4N) BACILLUS ANTHRACIS IMMUNOSERUM RABBIT 30 [hp_X] in 1 g INFLUENZA A VIRUS (UNII: R9HH0NDE2E) (INFLUENZA A VIRUS - UNII:R9HH0NDE2E) INFLUENZA A VIRUS 30 [hp_X] in 1 g RANCID BEEF (UNII: 29SUH5R3HU) (RANCID BEEF - UNII:29SUH5R3HU) RANCID BEEF 30 [hp_X] in 1 g ACONITUM NAPELLUS (UNII: U0NQ8555JD) (ACONITUM NAPELLUS - UNII:U0NQ8555JD) ACONITUM NAPELLUS 15 [hp_X] in 1 g ANTIMONY POTASSIUM TARTRATE (UNII: DL6OZ476V3) (ANTIMONY CATION (3+) - UNII:069647RPT5) ANTIMONY POTASSIUM TARTRATE 15 [hp_X] in 1 g ARNICA MONTANA (UNII: O80TY208ZW) (ARNICA MONTANA - UNII:O80TY208ZW) ARNICA MONTANA 15 [hp_X] in 1 g ARSENIC TRIOXIDE (UNII: S7V92P67HO) (ARSENIC CATION (3+) - UNII:C96613F5AV) ARSENIC TRIOXIDE 15 [hp_X] in 1 g BAPTISIA TINCTORIA (UNII: 5K1UO2888Y) (BAPTISIA TINCTORIA - UNII:5K1UO2888Y) BAPTISIA TINCTORIA 15 [hp_X] in 1 g ATROPA BELLADONNA (UNII: WQZ3G9PF0H) (ATROPA BELLADONNA - UNII:WQZ3G9PF0H) ATROPA BELLADONNA 15 [hp_X] in 1 g BRYONIA ALBA ROOT (UNII: T7J046YI2B) (BRYONIA ALBA ROOT - UNII:T7J046YI2B) BRYONIA ALBA ROOT 15 [hp_X] in 1 g LYTTA VESICATORIA (UNII: 3Q034RO3BT) (LYTTA VESICATORIA - UNII:3Q034RO3BT) LYTTA VESICATORIA 15 [hp_X] in 1 g MATRICARIA RECUTITA (UNII: G0R4UBI2ZZ) (MATRICARIA RECUTITA - UNII:G0R4UBI2ZZ) MATRICARIA RECUTITA 15 [hp_X] in 1 g CINCHONA OFFICINALIS BARK (UNII: S003A158SB) (CINCHONA OFFICINALIS BARK - UNII:S003A158SB) CINCHONA OFFICINALIS BARK 15 [hp_X] in 1 g EUPATORIUM PERFOLIATUM FLOWERING TOP (UNII: 1W0775VX6E) (EUPATORIUM PERFOLIATUM FLOWERING TOP - UNII:1W0775VX6E) EUPATORIUM PERFOLIATUM FLOWERING TOP 15 [hp_X] in 1 g IRON (UNII: E1UOL152H7) (IRON - UNII:E1UOL152H7) IRON 15 [hp_X] in 1 g GELSEMIUM SEMPERVIRENS ROOT (UNII: 639KR60Q1Q) (GELSEMIUM SEMPERVIRENS ROOT - UNII:639KR60Q1Q) GELSEMIUM SEMPERVIRENS ROOT 15 [hp_X] in 1 g CALCIUM SULFIDE (UNII: 1MBW07J51Q) (CALCIUM SULFIDE - UNII:1MBW07J51Q) CALCIUM SULFIDE 15 [hp_X] in 1 g IODINE (UNII: 9679TC07X4) (IODINE - UNII:9679TC07X4) IODINE 15 [hp_X] in 1 g LACHESIS MUTA VENOM (UNII: VSW71SS07I) (LACHESIS MUTA VENOM - UNII:VSW71SS07I) LACHESIS MUTA VENOM 15 [hp_X] in 1 g MERCURY (UNII: FXS1BY2PGL) (MERCURY - UNII:FXS1BY2PGL) MERCURY 15 [hp_X] in 1 g PHOSPHORUS (UNII: 27YLU75U4W) (PHOSPHORUS - UNII:27YLU75U4W) PHOSPHORUS 15 [hp_X] in 1 g PULSATILLA VULGARIS (UNII: I76KB35JEV) (PULSATILLA VULGARIS - UNII:I76KB35JEV) PULSATILLA VULGARIS 15 [hp_X] in 1 g TOXICODENDRON PUBESCENS LEAF (UNII: 6IO182RP7A) (TOXICODENDRON PUBESCENS LEAF - UNII:6IO182RP7A) TOXICODENDRON PUBESCENS LEAF 15 [hp_X] in 1 g SALIX NIGRA BARK (UNII: QU52J3A5B3) (SALIX NIGRA BARK - UNII:QU52J3A5B3) SALIX NIGRA BARK 15 [hp_X] in 1 g VERBASCUM THAPSUS (UNII: C9TD27U172) (VERBASCUM THAPSUS - UNII:C9TD27U172) VERBASCUM THAPSUS 15 [hp_X] in 1 g APIS MELLIFERA (UNII: 7S82P3R43Z) (APIS MELLIFERA - UNII:7S82P3R43Z) APIS MELLIFERA 6 [hp_X] in 1 g ECHINACEA, UNSPECIFIED (UNII: 4N9P6CC1DX) (ECHINACEA, UNSPECIFIED - UNII:4N9P6CC1DX) ECHINACEA, UNSPECIFIED 6 [hp_X] in 1 g GOLDENSEAL (UNII: ZW3Z11D0JV) (GOLDENSEAL - UNII:ZW3Z11D0JV) GOLDENSEAL 6 [hp_X] in 1 g INFLUENZA B VIRUS (UNII: 1314JZ2X6W) (INFLUENZA B VIRUS - UNII:1314JZ2X6W) INFLUENZA B VIRUS 30 [hp_X] in 1 g Inactive Ingredients Ingredient Name Strength SUCROSE (UNII: C151H8M554) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:55714-2441-1 28 g in 1 BOTTLE, GLASS; Type 0: Not a Combination Product 01/31/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 01/31/2019 Labeler - Newton Laboratories, Inc. (788793610) Registrant - Newton Laboratories, Inc. (788793610) Establishment Name Address ID/FEI Business Operations Newton Laboratories, Inc. 788793610 manufacture(55714-2441)