Label: ORTHO DF- folic acid and vitamin d3 capsule
- NHRIC Code(s): 69597-301-30
- Packager: Basiem
- Category: DIETARY SUPPLEMENT
- DEA Schedule: None
- Marketing Status: Dietary Supplement
Drug Label Information
Updated October 3, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
STATEMENT OF IDENTITY
Ortho DF Capsules is an orally administered prescription folate product for the dietary management of patients with unique nutritional needs requiring increased folate levels and Vitamin D supplementation due to Vitamin D deficiency and other nutritional supplementation.
Ortho DF should be administered under the supervision of a licensed medical practitioner.
Vitamin D3 (cholecalciferol) is a white, crystalline powder, very soluble in water, with the following structural formula: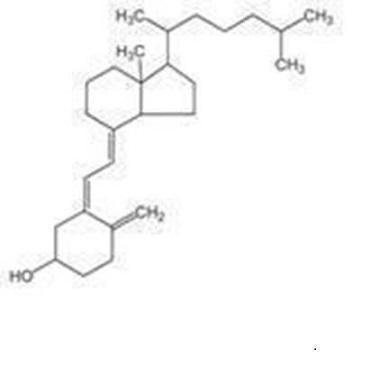
Each capsule contains:
Folic Acid .......................................................................1,666 mcg DFE (1 mg)
Vitamin D3 (Cholecalciferol) .............................................94.3 mcg (3775 IU)Each capsule contains the following inactive ingredients: Gelatin, cellulose, magnesium stearate, silica.
The in vivo synthesis of the major biologically active metabolites of vitamin D occurs in two steps. The first hydroxylation of ergocalciferol takes place in the liver (to 25-hydroxyvitamin D) and the second in the kidneys (to 1,25-dihydroxyvitamin D). Vitamin D metabolites promote the active absorption of calcium and phosphorus by the small intestine, thus elevating serum calcium and phosphate levels sufficiently to permit bone mineralization. Vitamin D metabolites also mobilize calcium and phosphate from bone and probably increase the reabsorption of calcium and perhaps also of phosphate by the renal tubules.
There is a time lag of 10 to 24 hours between the administration of vitamin D and the initiation of its action in the body due to the necessity of synthesis of the active metabolites in the liver and kidneys. Parathyroid hormone is responsible for the regulation of this metabolism in the kidneys.
-
HEALTH CLAIM:
Ortho DF is used for dietary management of patients with unique nutritional needs requiring increased folate levels, Vitamin D deficiency or are in need of Vitamin D supplementation and other nutritional supplementation.
Ortho DF can be taken by women of childbearing age, pregnant women, and lactating and nonlactating mothers. -
PRECAUTIONS:
KEEP OUT OF THE REACH OF CHILDREN.
Tell your doctor if you have: kidney problems, thyroid disease. This medication should be used as directed during pregnancy or while breast-feeding. Consult your doctor about the risks and benefits.
Folic acid alone is improper therapy in the treatment of pernicious anemia and other megaloblastic anemias where vitamin B12 is deficient. Folic acid in doses above 0.1 mg daily may obscure pernicious anemia in that hematologic remission can occur while neurological manifestations progress. - WARNINGS SECTION
- DOSAGE AND ADMINISTRATION:
-
PACKAGE LABEL:

KEEP OUT OF THE REACH OF CHILDREN.Store at 20°-25°C (68°-77°F); excursions permitted to 15°-30°C (59°-86°F) [See USP Controlled Room Temperature.]
Protect from heat, light and moisture.Tamper Evident: Do not use if seal is broken or missing
Ortho DF capsules are supplied as clear capsules printed with “301” dispensed in child-resistant bottles of 30ct (69597-301-30*).
* This product is a prescription-folate with or without other dietary ingredients that – due to increased folate levels (AUG 2 1973 FR 20750), requires an Rx on the label because of increased risk associated with masking of B12 deficiency (pernicious anemia). Based on our assessment of the risk of obscuring pernicious anemia, this product requires licensed medical supervision, an Rx status, and a National Drug Code (NDC) – or similarly-formatted product code, as required by pedigree reporting requirements and supply-chain control as well as – in some cases, for insurance-reimbursement applications.
All prescriptions using this product shall be pursuant to state statutes as applicable. This is not an Orange Book product. This product may be administered only under a physician’s supervision. There are no implied or explicit claims on therapeutic equivalence. -
INGREDIENTS AND APPEARANCE
ORTHO DF
folic acid and vitamin d3 capsuleProduct Information Product Type DIETARY SUPPLEMENT Item Code (Source) NHRIC:69597-301 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength FOLIC ACID (UNII: 935E97BOY8) (FOLIC ACID - UNII:935E97BOY8) FOLIC ACID 1 mg in 1 mg VITAMIN D (UNII: 9VU1KI44GP) (CHOLECALCIFEROL - UNII:1C6V77QF41) VITAMIN D .094 mg in 1 mg Inactive Ingredients Ingredient Name Strength GELATIN (UNII: 2G86QN327L) CELLULOSE ACETATE (UNII: 3J2P07GVB6) MAGNESIUM STEARATE (UNII: 70097M6I30) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NHRIC:69597-301-30 30 mg in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date dietary supplement 11/10/2017 Supplement Facts Serving Size : Serving per Container : Amount Per Serving % Daily Value color shape scoring 1 imprint size (solid drugs) 18 mm Labeler - Basiem (079686680)

