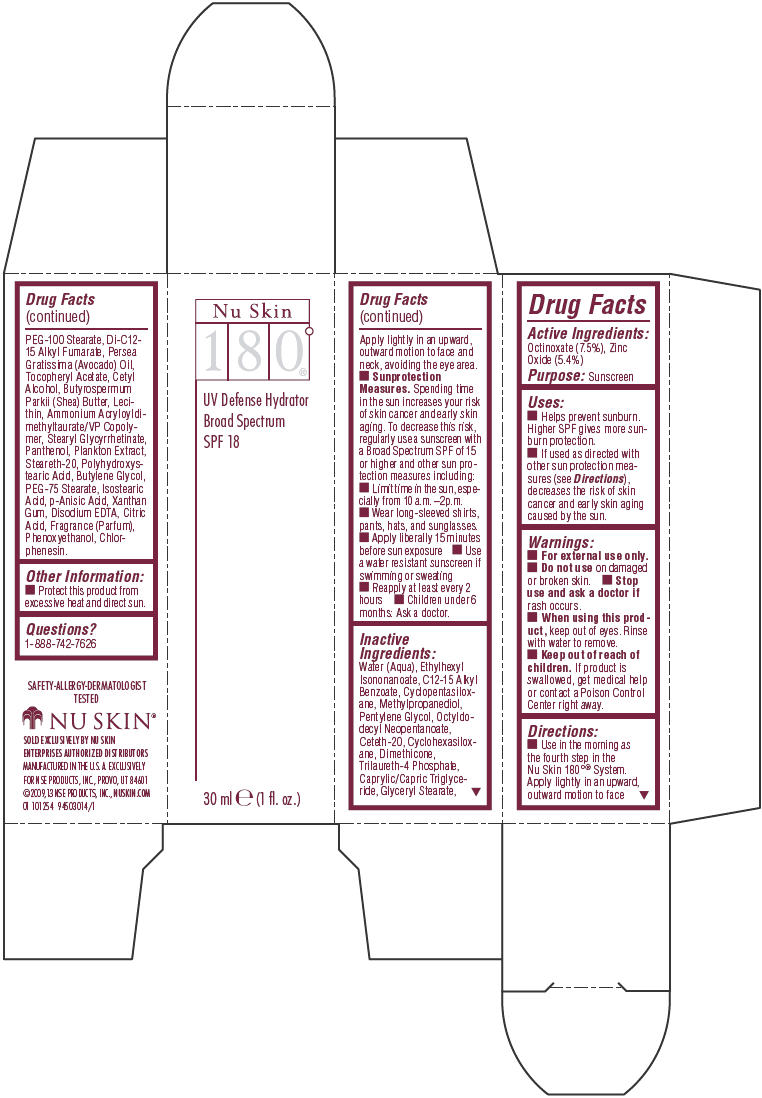
Label: NU SKIN 180 DEGREE UV DEFENSE HYDRATOR SPF 18- octinoxate and zinc oxide lotion
- NDC Code(s): 62839-1254-1
- Packager: NSE Products, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 10, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active Ingredients
- Purpose
-
Uses
- Helps prevent sunburn. Higher SPF gives more sunburn protection.
- If used as directed with other sun protection measures (see Directions), decreases the risk of skin cancer and early skin aging caused by the sun.
- Warnings
-
Directions
- Use in the morning as the fourth step in the Nu Skin 180°® System. Apply lightly in an upward, outward motion to face
Apply lightly in an upward, outward motion to face and neck, avoiding the eye area. -
Sunprotection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF of 15 or higher and other sun protection measures including:
- Limit time in the sun, especially from 10 a.m. –2p.m.
- Wear long-sleeved shirts, pants, hats, and sunglasses.
- Apply liberally 15 minutes before sun exposure
- Use a water resistant sunscreen if swimming or sweating
- Reapply at least every 2 hours
- Children under 6 months: Ask a doctor.
- Use in the morning as the fourth step in the Nu Skin 180°® System. Apply lightly in an upward, outward motion to face
-
Inactive Ingredients
Water (Aqua), Ethylhexyl Isononanoate, C12-15 Alkyl Benzoate, Cyclopentasiloxane, Methylpropanediol, Pentylene Glycol, Octyldodecyl Neopentanoate, Ceteth-20, Cyclohexasiloxane, Dimethicone, Trilaureth-4 Phosphate, Caprylic/Capric Triglyceride, Glyceryl Stearate, PEG-100 Stearate, Di-C12-15 Alkyl Fumarate, Persea Gratissima (Avocado) Oil, Tocopheryl Acetate, Cetyl Alcohol, Butyrospermum Parkii (Shea) Butter, Lecithin, Ammonium Acryloyldimethyltaurate/VP Copolymer, Stearyl Glycyrrhetinate, Panthenol, Plankton Extract, Steareth-20, Polyhydroxystearic Acid, Butylene Glycol, PEG-75 Stearate, Isostearic Acid, p-Anisic Acid, Xanthan Gum, Disodium EDTA, Citric Acid, Fragrance (Parfum), Phenoxyethanol, Chlorphenesin.
- Other Information
- Questions?
- PRINCIPAL DISPLAY PANEL - 30 ml Bottle Carton
-
INGREDIENTS AND APPEARANCE
NU SKIN 180 DEGREE UV DEFENSE HYDRATOR SPF 18
octinoxate and zinc oxide lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:62839-1254 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Octinoxate (UNII: 4Y5P7MUD51) (Octinoxate - UNII:4Y5P7MUD51) Octinoxate 75 mg in 1 mL Zinc Oxide (UNII: SOI2LOH54Z) (Zinc Oxide - UNII:SOI2LOH54Z) Zinc Oxide 54 mg in 1 mL Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Ethylhexyl Isononanoate (UNII: I6KB4GE3K4) Alkyl (C12-15) Benzoate (UNII: A9EJ3J61HQ) Cyclomethicone 5 (UNII: 0THT5PCI0R) Methylpropanediol (UNII: N8F53B3R4R) Pentylene Glycol (UNII: 50C1307PZG) Octyldodecyl Neopentanoate (UNII: X8725R883T) Ceteth-20 (UNII: I835H2IHHX) Cyclomethicone 6 (UNII: XHK3U310BA) Dimethicone (UNII: 92RU3N3Y1O) Trilaureth-4 Phosphate (UNII: M96W2OLL2V) Medium-Chain Triglycerides (UNII: C9H2L21V7U) Glyceryl Monostearate (UNII: 230OU9XXE4) PEG-100 Stearate (UNII: YD01N1999R) Di-C12-15 Alkyl Fumarate (UNII: A1CB3Z898P) Avocado Oil (UNII: 6VNO72PFC1) Shea Butter (UNII: K49155WL9Y) Ammonium Acryloyldimethyltaurate/VP Copolymer (UNII: W59H9296ZG) Stearyl Glycyrrhetinate (UNII: 3YYE6VJS0P) Panthenol (UNII: WV9CM0O67Z) Steareth-20 (UNII: L0Q8IK9E08) Butylene Glycol (UNII: 3XUS85K0RA) PEG-75 Stearate (UNII: OT38R0N74H) Isostearic Acid (UNII: X33R8U0062) p-Anisic Acid (UNII: 4SB6Y7DMM3) Xanthan Gum (UNII: TTV12P4NEE) Edetate Disodium (UNII: 7FLD91C86K) Citric Acid Monohydrate (UNII: 2968PHW8QP) Phenoxyethanol (UNII: HIE492ZZ3T) Chlorphenesin (UNII: I670DAL4SZ) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:62839-1254-1 1 in 1 CARTON 08/01/2013 1 30 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 08/01/2013 Labeler - NSE Products, Inc. (803486393)