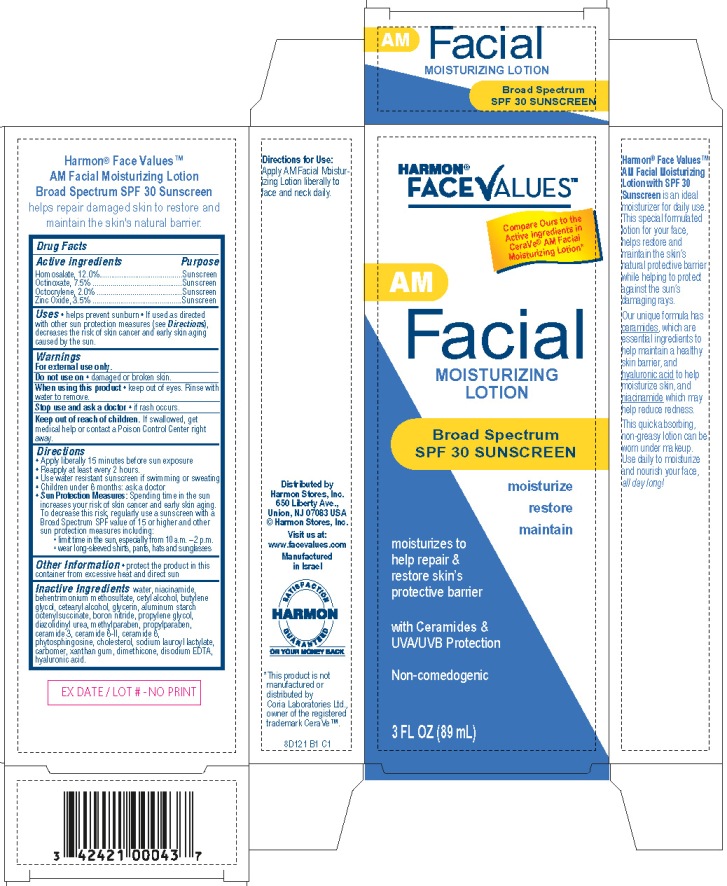
Label: FACE VALUES - AM FACIAL MOISTURIZING BROAD SPECTRUM SPF30 SUNSCREEN- homosalate, octinoxate, octocrylene, zinc oxide lotion
-
Contains inactivated NDC Code(s)
NDC Code(s): 63940-121-11 - Packager: Harmon Stores Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated November 12, 2013
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
- Warnings
- Do not use on
- When using this product
- Stop use and ask a doctor
- Keep Out of Reach of Children.
-
Directions
- Apply liberally 15 minutes before sun exposure.
- Reapply at least every 2 hours
- Use water resistant sunscreen if swimming or sweating
- Children under 6 months of age: ask a doctor
-
Sun Protection Measures: Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m. - 2 p.m.
- wear long-sleeved shirts, pants, hats and sunglasses
- Other Information
-
Inactive ingredients
water, niacinamide, behentrimonium methosulfate, cetyl alcohol, butylene glycol, cetearyl alcohol, glycerin, aluminum starch octenylsuccinate, boron nitride, propylene glycol, diazolidinyl urea, methylparaben, propylparaben, ceramide 3, ceramide 6- II, ceramide 1, phytosphingosine, cholesterol, sodium lauroyl lactylate, carbomer, xanthan gum, dimethicone, disodium EDTA, hyaluronic acid
-
Package/Label Principal Display Panel
Harmon® FaceValuesTM AM Facial Moisturizing Lotion
Broad Spectrum SPF30 Sunscreen
moisturize
restore
maintainmoisturizes to help repair & restore skin's protective barrier
with Ceramides & UVA/UVB Protection
Non-comedogenic
Compare Ours to the Active ingredients in CeraVe®
AM Facial Moisturizing Lotion*3 Fl Oz (89 mL)
Distributed by
Harmon Stores, Inc.
650 Liberty Ave.
Union, NJ 07083 USA
©Harmon Stores, Inc.
Visit us at:
www.facevalues.com
Manufactured in Israel
8D121 B1 C1
*This product is not manufactured or distributed by Coria Laboratories Ltd,
owner of the registered trademark CeraVeTM.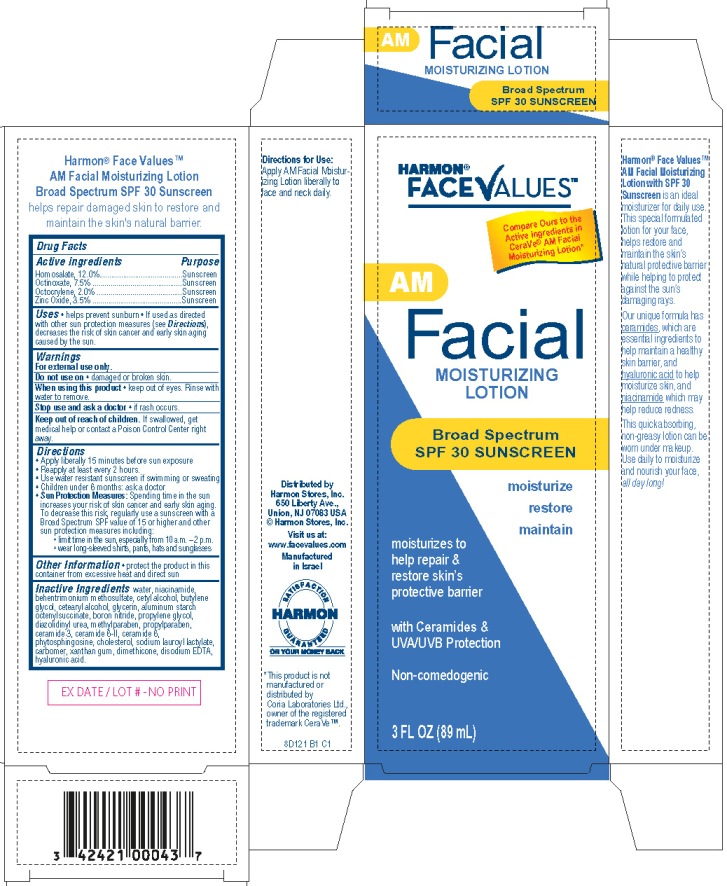
Label
-
INGREDIENTS AND APPEARANCE
FACE VALUES - AM FACIAL MOISTURIZING BROAD SPECTRUM SPF30 SUNSCREEN
homosalate, octinoxate, octocrylene, zinc oxide lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:63940-121 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 12 g in 100 mL OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 7.5 g in 100 mL OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 2 g in 100 mL ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 3.5 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) NIACINAMIDE (UNII: 25X51I8RD4) BEHENTRIMONIUM METHOSULFATE (UNII: 5SHP745C61) CETYL ALCOHOL (UNII: 936JST6JCN) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) GLYCERIN (UNII: PDC6A3C0OX) ALUMINUM STARCH OCTENYLSUCCINATE (UNII: I9PJ0O6294) BORON NITRIDE (UNII: 2U4T60A6YD) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) DIAZOLIDINYL UREA (UNII: H5RIZ3MPW4) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLPARABEN (UNII: Z8IX2SC1OH) CERAMIDE 3 (UNII: 4370DF050B) CERAMIDE 6 II (UNII: F1X8L2B00J) CERAMIDE 1 (UNII: 5THT33P7X7) PHYTOSPHINGOSINE (UNII: GIN46U9Q2Q) CHOLESTEROL (UNII: 97C5T2UQ7J) SODIUM LAUROYL LACTYLATE (UNII: 7243K85WFO) CARBOMER COPOLYMER TYPE A (ALLYL PENTAERYTHRITOL CROSSLINKED) (UNII: 71DD5V995L) XANTHAN GUM (UNII: TTV12P4NEE) DIMETHICONE (UNII: 92RU3N3Y1O) EDETATE DISODIUM (UNII: 7FLD91C86K) HYALURONIC ACID (UNII: S270N0TRQY) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63940-121-11 1 in 1 CARTON 1 89 mL in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 11/15/2013 Labeler - Harmon Stores Inc (804085293)