Label: POSLUMA- flotufolastat f-18 injection
- NDC Code(s): 69932-002-50
- Packager: Blue Earth Diagnostics
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated September 24, 2025
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use POSLUMA ® safely and effectively. See full prescribing information for POSLUMA.
POSLUMA (flotufolastat F 18) injection, for intravenous use
Initial U.S. Approval: 2023INDICATIONS AND USAGE
POSLUMA is a radioactive diagnostic agent indicated for positron emission tomography (PET) of prostate-specific membrane antigen (PSMA) positive lesions in men with prostate cancer:
- with suspected metastasis who are candidates for initial definitive therapy
- with suspected recurrence based on elevated serum prostate-specific antigen (PSA) level. ( 1)
DOSAGE AND ADMINISTRATION
- Recommended amount of radioactivity of POSLUMA is 296 MBq (8 mCi) administered as an intravenous bolus injection. ( 2.2)
- Initiate imaging approximately 60 minutes after administration. Scanning should start from mid-thigh and proceed to base of skull. ( 2.4)
- See full prescribing information for additional preparation, handling, administration, imaging, and radiation dosimetry information. ( 2.3, 2.4)
DOSAGE FORMS AND STRENGTHS
Injection: 296 MBq/mL to 5,846 MBq/mL (8 mCi/mL to 158 mCi/mL) as flotufolastat F 18 gallium in approximately 25 mL at end of synthesis in a multiple-dose vial. ( 3)
CONTRAINDICATIONS
None. ( 4)
WARNINGS AND PRECAUTIONS
- Risk of Image Misinterpretation: Image interpretation errors can occur with POSLUMA imaging. Interpretation of POSLUMA PET may differ depending on imaging readers in patients with suspected recurrence of prostate cancer. Consider multidisciplinary consultation and histopathological confirmation. ( 5.1, 14.2)
- Radiation risk: POSLUMA contributes to a patient's long-term cumulative radiation exposure. Ensure safe handling to protect patients and health care workers from unintentional radiation exposure. ( 2.1, 5.2)
ADVERSE REACTIONS
The most common adverse reactions (≥0.4%) are diarrhea, blood pressure increase, and injection site pain. ( 6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Blue Earth Diagnostics Ltd at 1-844-POSLUMA (1-844-767-5862) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 5/2023
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Radiation Safety - Drug Handling
2.2 Recommended Dose and Administration Instructions
2.3 Patient Preparation
2.4 Image Acquisition
2.5 Image Display and Interpretation
2.6 Radiation Dosimetry
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Risk of Image Misinterpretation
5.2 Radiation Risks
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
11 DESCRIPTION
11.1 Chemical Characteristics
11.2 Physical Characteristics
11.3 External Radiation
10 OVERDOSAGE
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Imaging Prior to Initial Definitive Therapy of Prostate Cancer
14.2 Imaging for Suspected Recurrence of Prostate Cancer
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
POSLUMA is indicated for positron emission tomography (PET) of prostate-specific membrane antigen (PSMA) positive lesions in men with prostate cancer
- with suspected metastasis who are candidates for initial definitive therapy.
- with suspected recurrence based on elevated serum prostate-specific antigen (PSA) level.
-
2 DOSAGE AND ADMINISTRATION
2.1 Radiation Safety - Drug Handling
Handle POSLUMA with safety measures to minimize radiation exposure [see Warnings and Precautions ( 5.2)] . Use waterproof gloves, effective radiation shielding, including syringe shields, and other appropriate safety measures when handling and administering POSLUMA.
Radiopharmaceuticals should be used by or under the control of physicians who are qualified by specific training and experience in the safe use and handling of radionuclides, and whose experience and training have been approved by the appropriate governmental agency authorized to license the use of radionuclides.
2.2 Recommended Dose and Administration Instructions
Recommended Dose
The recommended amount of radioactivity to be administered in adults is 296 MBq (8 mCi) as an intravenous bolus injection.
Preparation and Administration Instructions
- Inspect POSLUMA visually for particulate matter and discoloration before administration. Do not use the drug if the solution contains particulate matter or is discolored.
- Use aseptic technique and radiation shielding when withdrawing and administering POSLUMA.
- Calculate the necessary volume to administer based on calibration time and required dose.
- The recommended maximum volume of undiluted POSLUMA is 5 mL.
- POSLUMA may be diluted with 0.9% Sodium Chloride Injection, USP.
- Assay the dose in a dose calibrator before administration.
2.3 Patient Preparation
Instruct patients to drink water prior to administration of POSLUMA to ensure adequate hydration and to continue drinking and voiding frequently for the first few hours following administration to reduce radiation exposure.
2.4 Image Acquisition
- Patients should void immediately prior to imaging.
- Position the patient supine with arms above the head.
- Begin image acquisition approximately 60 minutes after POSLUMA injection.
- Image acquisition should start from mid-thigh and proceed to the base of the skull.
- Scan duration is approximately 20 minutes depending on the number of bed positions and acquisition time per bed position (typically 3 minutes). Adapt imaging technique according to the equipment used and patient characteristics in order to obtain the best image quality possible.
2.5 Image Display and Interpretation
POSLUMA binds to PSMA. PET images obtained using POSLUMA indicate the presence of PSMA in tissues [see Clinical Pharmacology ( 12.1)] . Lesions should be considered suspicious if uptake is greater than physiologic uptake in that tissue or greater than adjacent background if no physiologic uptake is expected. Tumors that do not express PSMA will not be visualized. Increased uptake in tumors is not specific for prostate cancer [see Warnings and Precautions ( 5.1)].
2.6 Radiation Dosimetry
Estimated absorbed radiation doses for adult patients following intravenous injection of POSLUMA are shown in Table 1. The effective radiation dose resulting from the administration of the recommended activity of 296 MBq of POSLUMA is 4.1 mSv. The radiation absorbed doses to the critical organs of adrenal glands, kidneys, and submandibular glands for the recommended activity of 296 MBq are 54.3 mGy, 51 mGy, and 43.8 mGy, respectively. When PET/CT is performed, exposure to radiation will increase by an amount dependent on the settings used in the CT acquisition.
Table 1: Estimated Radiation Absorbed Doses in Organs/Tissues in Adults who Received POSLUMA *The absorbed dose value reflects self-irradiation only; no dose contribution from other regions to the glands is added. **A 1-hour bladder voiding interval is assumed.
Organ/Tissue Absorbed Dose per Unit Administered Activity (mGy/MBq) Mean Adrenal glands 0.184 Brain 0.002 Breasts 0.004 Gallbladder wall 0.017 Lower large intestine wall 0.007 Upper large intestine wall 0.01 Heart wall 0.02 Kidneys 0.172 Lacrimal glands 0.08 * Liver 0.062 Lungs 0.01 Muscle 0.006 Osteogenic cells 0.012 Ovaries 0.005 Pancreas 0.028 Parotid glands 0.114 * Red bone marrow 0.01 Skin 0.002 Small intestine 0.012 Spleen 0.083 Stomach wall 0.012 Sublingual glands 0.065 * Submandibular glands 0.148 * Testes 0.005 Thymus gland 0.01 Thyroid 0.01 Urinary bladder wall 0.006 ** Uterus 0.011 Effective dose (mSv/MBq) 0.014** - 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Risk of Image Misinterpretation
Image interpretation errors can occur with POSLUMA PET. A negative image does not rule out the presence of prostate cancer and a positive image does not confirm the presence of prostate cancer. The performance of POSLUMA for imaging metastatic pelvic lymph nodes in patients prior to initial definitive therapy seems to be affected by serum PSA levels and risk grouping [See Clinical Studies ( 14.1)] . The performance of POSLUMA for imaging patients with biochemical evidence of recurrence of prostate cancer seems to be affected by serum PSA levels [See Clinical Studies ( 14.2)] . Flotufolastat F 18 uptake is not specific for prostate cancer and may occur in other types of cancer, in non-malignant processes, and in normal tissues. Clinical correlation, which may include histopathological evaluation, is recommended.
Risk of Image Misinterpretation in Patients with Suspected Prostate Cancer Recurrence
The interpretation of POSLUMA PET may differ depending on imaging readers, particularly in the prostate/prostate bed region [see Clinical Studies ( 14.2)]. Because of the associated risk of false positive interpretation, consider multidisciplinary consultation and histopathological confirmation when clinical decision-making hinges on flotufolastat F18 uptake only in the prostate/prostate bed region or only on uptake interpreted as borderline.
5.2 Radiation Risks
POSLUMA use contributes to a patient's overall long-term cumulative radiation exposure. Long-term cumulative radiation exposure is associated with an increased risk for cancer. Advise patients to hydrate before and after administration and to void frequently after administration. Ensure safe handling to minimize radiation exposure to the patient and health care providers [see Dosage and Administration ( 2.1, 2.2)] .
-
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of POSLUMA was evaluated in 747 patients with prostate cancer [see Clinical Studies ( 14.1, 14.2)] . All patients received a single administration of POSLUMA with an administered radioactivity (mean ± SD) of 307 ± 23 MBq (8.3 ± 0.6 mCi). The mean age of patients was 67 years (range: 43 to 86 years); distribution by race was 78% White, 12% Black or African American, 2% other, and 7% unreported; and distribution by ethnicity was 5% Hispanic/Latino, 87% non-Hispanic/Latino, and 8% unreported.
The adverse reactions reported in ≥ 0.4% of patients are shown in Table 2.
Table 2: Adverse Reactions in ≥ 0.4% of Patients with Prostate Cancer Receiving POSLUMA Adverse Reaction POSLUMA
N = 747
n (%)Diarrhea 5 (0.7%) Blood pressure increase 4 (0.5%) Injection site pain 3 (0.4%) - 7 DRUG INTERACTIONS
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
POSLUMA is not indicated for use in females. There are no available data on the use of POSLUMA in pregnant women to evaluate for a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes. Animal reproduction studies have not been conducted with flotufolastat F 18. Radioactive drugs, including POSLUMA, have the potential to cause fetal harm depending on the fetal stage of development and the magnitude of the radiation dose.
8.4 Pediatric Use
The safety and effectiveness of POSLUMA have not been established in pediatric patients.
8.5 Geriatric Use
Among the total number of patients receiving POSLUMA in clinical studies of prostate cancer, 463 (62%) were 65 years of age and older, while 118 (16%) were 75 years of age and older [see Clinical Studies ( 14.1, 14.2)] . No overall differences in safety or effectiveness were observed between these patients and younger adult patients.
-
11 DESCRIPTION
11.1 Chemical Characteristics
POSLUMA (flotufolastat F 18) injection is a radioactive diagnostic agent for intravenous use. The active ingredient of POSLUMA is flotufolastat F 18 gallium, of which the molecular structure includes a DOTAGA complex with nonradioactive gallium. Radioactive fluorine-18 is covalently bound to silicon.
Chemically, flotufolastat F 18 gallium is gallate(6-), [(4 S,8 S,13 R,27 R,30 R,35 S)-35-[4,10-bis[(carboxy-k O)methyl]-7-(carboxymethyl)-1,4,7,10-tetraazacyclododec-1-yl-k N1,k N4,k N7,k N10]-30-[[[4-[bis(1,1-dimethylethyl)fluoro- 18F-silyl]benzoyl]amino]methyl]-1,36-dihydroxy-1,6,11,18,21,29,32,36-octaoxo-5,7,12,17,22,28,31-heptaazahexatriacontane-4,8,13,27-tetracarboxylato(9-)]-, hydrogen (1:6). The molecular weight is 1537.3 g/mol and the structural formula is:
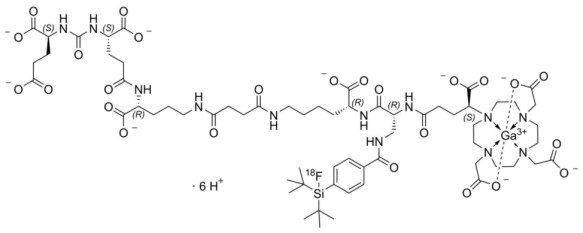
POSLUMA is a sterile, non-pyrogenic, clear, colorless, and isotonic solution. Each mL contains up to 20 mcg of flotufolastat gallium, up to 5,846 MBq (158 mCi) as flotufolastat F 18 gallium at end of synthesis, and the following inactive ingredients: 6.3% (v/v) alcohol, 1.9 mg anhydrous citric acid, 7.2 mg sodium chloride, and 0.75 mg sodium hydroxide to adjust pH between 4 and 6. POSLUMA contains no preservative.
11.2 Physical Characteristics
POSLUMA contains fluorine-18 (F 18) which is a cyclotron produced radionuclide that decays by positron emission (β+ decay, 96.7%) and orbital electron capture (3.3%) to stable oxygen-18 with a physical half-life of 109.8 minutes ( Table 3). The principal photons useful for diagnostic imaging are the coincident pair of 511 keV gamma photons, resulting from the interaction of the emitted positron with an electron ( Table 4).
Table 3: Physical Decay Chart for Fluorine-18 Minutes Fraction Remaining 0 1 15 0.909 30 0.826 60 0.683 110 0.5 220 0.25 Table 4: Principal Radiation Produced from Decay of Fluorine-18 Energy (keV) Abundance (%) Positron 249.8 96.7 Gamma 511 193.5 11.3 External Radiation
The point source air-kerma coefficient for F 18 is 3.75 × 10 -17 Gy m 2/(Bq s). The first half-value thickness of lead (Pb) for F 18 gamma rays is approximately 6 mm. The relative reduction of radiation emitted by F 18 that results from various thicknesses of lead shielding is shown in Table 5. The use of 8 cm of Pb will decrease the radiation transmission (i.e., exposure) by a factor of about 10,000.
Table 5: Radiation Attenuation of 511 keV Gamma Rays by Lead Shielding Shield Thickness cm of Lead (Pb) Coefficient of Attenuation 0.6 0.5 2 0.1 4 0.01 6 0.001 8 0.0001 - 10 OVERDOSAGE
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of action
Flotufolastat F 18 binds to PSMA (IC50 = 4.4 nM) expressed on cells, including prostate cancer cells, and is internalized. Prostate cancer cells usually overexpress PSMA. Fluorine-18 is a ß+ emitting radionuclide that can be detected using positron emission tomography.
12.2 Pharmacodynamics
The relationship between flotufolastat F 18 plasma concentrations and image interpretation has not been fully characterized.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Animal studies to assess the carcinogenicity or mutagenic potential of flotufolastat have not been conducted. However, flotufolastat F 18 has the potential to be mutagenic because of the F 18 radionuclide.
No studies in animals have been performed to evaluate potential impairment of fertility in males or females.
-
14 CLINICAL STUDIES
14.1 Imaging Prior to Initial Definitive Therapy of Prostate Cancer
The safety and efficacy of POSLUMA were evaluated in LIGHTHOUSE (NCT04186819), a prospective, multicenter, open-label, single-arm study in patients with prostate cancer who were candidates for initial definitive therapy.
The study enrolled 356 patients diagnosed with unfavorable intermediate-risk (32%) or high-/very high-risk prostate cancer (68%) who were candidates for radical prostatectomy and pelvic lymph node dissection (PLND). Unfavorable intermediate-risk was defined as having any ≥ 2 intermediate risk factors [T2b-T2c, Gleason score 7, PSA 10-20], Gleason pattern 4+3=7, or ≥ 50% of biopsy cores positive for prostate cancer. High or very high-risk was defined as having T3 or T4 disease, Gleason score ≥8, primary Gleason pattern 5, and/or PSA >20.
All patients received a single dose of POSLUMA with an administered radioactivity (mean ± SD) of 307 ± 23 MBq (8.3 ± 0.62 mCi), followed by PET/CT scan from mid-thigh to base of the skull. Three central readers blinded to clinical information independently interpreted each scan for lesions considered positive for prostate cancer in pelvic lymph nodes, categorized by subregion and left and right laterality [see Dosage and Administration ( 2.5)] . Positive lesions in the prostate gland, lymph nodes outside the pelvis, soft tissue/parenchyma, and bones were also recorded.
A total of 296 patients (83%) underwent standard-of-care prostatectomy and template PLND and had sufficient histopathology data for evaluation of the pelvic lymph nodes. The mean age was 65 years (range 46 to 82 years); distribution by race was 82% White, 8% Black or African American, 0.3% other, and 10% unreported; and distribution by ethnicity was 5% Hispanic/Latino, 86% non-Hispanic/Latino, and 9% unreported. The median serum PSA was 8.4 ng/mL. The total Gleason score was 7 for 45%, 8 for 26%, and 9 for 25% of the patients, with the remainder of the patients having Gleason scores of 6 or 10. Approximately 24% of patients had pelvic lymph node metastases based on histopathology.
POSLUMA performance was evaluated against histopathology after matching by hemipelvis. Table 6 shows the results, such that at least one true positive hemipelvis region defined a true positive patient.
Table 6: Patient-Level, Hemipelvis Region-Matched Performance of POSLUMA PET for Detection of Pelvic Lymph Node Metastasis (N1) in LIGHTHOUSE CI= confidence interval
N=296 Reader 1 Reader 2 Reader 3 True Positive 21 19 16 False Positive 16 14 7 True Negative 210 212 219 False Negative 49 51 54 Sensitivity, (%)
[95% CI]30%
[20, 42]27%
[17, 39]23%
[14, 35]Specificity, (%)
[95% CI]93%
[89, 96]94%
[90, 97]97%
[94, 99]Positive Predictive Value,
(%) [95% CI]57%
[40, 73]58%
[39, 75]70%
[47, 87]Negative Predictive Value,
(%) [95% CI]81%
[76, 86]81%
[75, 85]80%
[75, 85]In exploratory analyses, there were numerical trends towards higher sensitivity among patients with PSA greater than or equal to the median value (8.4 ng/mL) and among patients with high-risk or very high-risk categorization.
POSLUMA-positive lesions outside of the prostate gland and pelvic lymph nodes (M1) were also evaluated. As a percentage of the 352 patients with an evaluable POSLUMA scan and of the 61 patients with at least one POSLUMA positive M1 lesion, 10% (95% CI: 7% to 13%) and 56% (95% CI: 42% to 68%), respectively, had at least one matching positive M1 lesion between the POSLUMA majority read and a reference standard consisting of other imaging evaluated by a separate consensus panel or histopathology.
14.2 Imaging for Suspected Recurrence of Prostate Cancer
The safety and efficacy of POSLUMA were evaluated in SPOTLIGHT (NCT04186845), a prospective, multicenter, open-label, single-arm study in patients with biochemical evidence of recurrent prostate cancer.
The study enrolled 391 patients with suspected recurrence defined by either serum PSA of at least 0.2 ng/mL after radical prostatectomy (with confirmatory PSA level also at least 0.2 ng/mL) or by an increase in serum PSA of at least 2 ng/mL above the nadir after other therapies.
All patients received a single dose of POSLUMA with an administered radioactivity (mean ± SD) of 306 ± 22 MBq (8.27 ±0.61 mCi), followed by PET/CT scan from mid-thigh to base of the skull. Three central readers blinded to clinical information independently interpreted each scan by region for the presence and location of lesions considered positive for prostate cancer [see Dosage and Administration ( 2.5)] . The regions interpreted were grouped into three for primary analysis: prostate/prostate bed; pelvic lymph nodes; and other (including extra-pelvic lymph nodes, bone, and soft tissue/parenchyma).
A total of 389 patients had an evaluable POSLUMA PET scan. The mean age was 68 years (range: 43 to 86 years); distribution by race was 75% White, 16% Black or African American, 4% other, and 5% unreported; and distribution by ethnicity 5% was Hispanic/Latino, 87% non-Hispanic/Latino, and 8% unreported. The median baseline serum PSA level was 1.1 ng/mL with 60% of patients having a baseline PSA <2.0 ng/mL. Prior treatment included radical prostatectomy in 79% of the patients.
POSLUMA-positive interpretations were compared to a reference standard of either histopathology or other imaging (CT, MRI, Technetium 99m bone scan, or fluciclovine F 18 PET) obtained within 90 days of the POSLUMA scan using a lesion-to-lesion co-localization method and separate consensus panel. Reference standard information for negative interpretations was not collected.
At least one POSLUMA-positive lesion was detected by at least one reader in 366 patients (94%). Reference standard information consisted of imaging only (n=297) or histopathology (n=69). As a percentage of patients with an evaluable scan, 51% (95% CI: 46% to 56%) for reader 1, 48% (95% CI: 43% to 53%) for reader 2, and 49% (95% CI: 44% to 54%) for reader 3 had at least one matching positive region between the POSLUMA scan and the reference standard. Of all POSLUMA-positive regions, 46% (95% CI: 42% to 50%) for reader 1, 60% (95% CI: 55% to 66%) for reader 2, and 53% (95% CI: 48% to 58%) for reader 3 were categorized as positive by the reference standard.
Table 7 shows patient-level results from the majority read stratified by serum PSA level. Percent PET positivity was calculated as the percentage of patients with POSLUMA-positive lesions out of all patients with an evaluable PET scan. Percent PET positivity includes true and false positives and is not a measure of diagnostic performance.
Table 7: Patient-Level POSLUMA PET Results and Percent PET Positivity Stratified by Serum PSA Level in SPOTLIGHT by Majority Read (N=389) PSA = prostate-specific antigen, PA = positive agreement, NPA = no positive agreement, CI = confidence interval
aImaging comprised of one or more of the following: CT, MRI, 99mTc Bone Scan, fluciclovine F 18 PET
PSA (ng/mL) N PET Positive Patients PET Negative Patients Percent PET Positivity
[95% CI]Total Histopathology Imaging onlya PA NPA PA NPA < 0.5 121 77 6 4 27 40 44 64%
[54,72]≥ 0.5 and < 1 67 51 7 3 24 17 16 76%
[64,86]≥ 1 and < 2 45 42 10 2 18 12 3 93%
[82, 99]≥ 2 156 152 33 3 84 32 4 97%
[94, 99]Total 389 322 56 12 153 101 67 83%
[79, 86]Variable Interpretation in Patients with Suspected Prostate Cancer Recurrence
POSLUMA reader agreement was evaluated for the three central readers and 389 patients. Inter-reader Fleiss ϰ was 0.41 (95% CI: 0.39-0.43). The three readers agreed on the presence or absence of positive lesions across all five evaluated regions in 118 patients (30% unanimity) [see Warning and Precautions ( 5.1)].
Given the level of inter-reader agreement observed overall, POSLUMA reader agreement was further evaluated by regional subgroup. The Fleiss ϰ for was 0.40 (95% CI: 0.33-0.46) in the prostate/prostate bed, 0.73 (95% CI: 0.67-0.78) in the pelvic lymph nodes, and 0.62 (95% CI: 0.58-0.65) across the other regions.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
POSLUMA injection is supplied as a clear, colorless solution in a multiple-dose glass vial (NDC 69932-002-50) containing 296 MBq/mL to 5,846 MBq/mL (8 mCi/mL to 158 mCi/mL) as flotufolastat F 18 gallium in approximately 25 mL at end of synthesis.
Storage and Handling
Store POSLUMA at 20°C to 25°C (68°F to 77°F) [see USP Controlled Room Temperature]. Store POSLUMA in the original container in radiation shielding. The expiration date and time are provided on the container label. Use POSLUMA within 10 hours from end of synthesis.
Dispose of unused POSLUMA in compliance with applicable regulations.
This preparation is approved for use by persons under license by the Nuclear Regulatory Commission or the relevant regulatory authority of an Agreement State.
-
17 PATIENT COUNSELING INFORMATION
Adequate Hydration
Instruct patients to drink a sufficient amount of water to ensure adequate hydration before their PET study and urge them to drink and urinate as often as possible during the first hours following the administration of POSLUMA, in order to reduce radiation exposure [see Dosage and Administration ( 2.3) and Warnings and Precautions ( 5.2)].
Marketed by Blue Earth Diagnostics Ltd., Oxford, OX4 4GA, UK
POSLUMA ® is a registered trademark of Blue Earth Diagnostics Ltd.
© 2023 Blue Earth Diagnostics Ltd. – all rights reserved.
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
POSLUMA
flotufolastat f-18 injectionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:69932-002 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength FLOTUFOLASTAT F-18 (UNII: 811W19E3OL) (FLOTUFOLASTAT F-18 - UNII:811W19E3OL) FLOTUFOLASTAT F-18 158 mCi in 1 mL Inactive Ingredients Ingredient Name Strength SODIUM HYDROXIDE (UNII: 55X04QC32I) WATER (UNII: 059QF0KO0R) SODIUM CHLORIDE (UNII: 451W47IQ8X) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69932-002-50 25 mL in 1 VIAL, MULTI-DOSE; Type 0: Not a Combination Product 05/25/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA216023 05/25/2023 Labeler - Blue Earth Diagnostics (219742530) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Philadelphia) 004201823 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc (PETNET Corporate) 015223951 analysis(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Albany) 018537881 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Seattle) 026659644 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions Inc. (Culver City) 033883716 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Sacramento) 078575242 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Denver) 078575260 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Loma Linda) 079262099 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (San Francisco) 080547824 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Kansas City) 102326340 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Raleigh) 103781071 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Jacksonville) 111110727 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (South Florida) 117843428 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Chicago) 118120165 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Portland) 123562576 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (San Antonio) 125764907 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Columbia) 128523862 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Covington) 134146484 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Little Rock) 154173350 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations Wickham Micro Limited 228216353 analysis(69932-002) Establishment Name Address ID/FEI Business Operations Almac Sciences Limited 232665666 api manufacture(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Phoenix) 603833208 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Houston) 621380547 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Tampa) 788930480 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Hackensack) 796129646 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Spokane) 798775214 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Tulsa) 798884966 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Dallas) 799246256 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Nashville) 840517945 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Atlanta) 961592982 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Cincinnati) 961593220 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Louisville) 961593337 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Detroit) 961593386 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Boston) 961597122 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Cleveland) 961597213 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Las Vegas) 961597221 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Albuquerque) 961682551 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Minneapolis) 965557486 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations PETNET Solutions, Inc. (Charlottesville) 968897954 positron emission tomography drug production(69932-002) Establishment Name Address ID/FEI Business Operations Almac Sciences Ireland Limited 985822621 analysis(69932-002) Establishment Name Address ID/FEI Business Operations The University of Utah DBA Cyclotron Radiochemistry Lab Hubtsman Cancer Institute 018432646 positron emission tomography drug production(69932-002)


