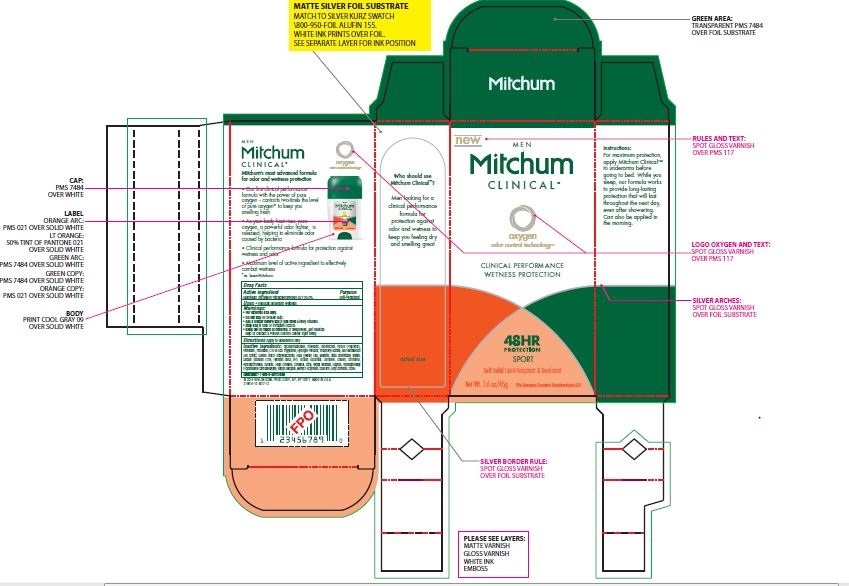
Label: MITCHUM CLINICAL SOFT SOLID- aluminum zirconium tetrachlorohydrex gly 20.0% gel
- NDC Code(s): 10967-658-16
- Packager: Revlon Consumer Products Corp.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 1, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients
- Keep out of reach of children
-
Inactive Ingredient section
Cyclopentasiloxane; Tribehenin; DIimethicone; Petrolatum; Parfume (Fragrance); Trisiloxane; C18-36 Acid Triglyceride; Aloe Barbadensis Leaf Extract; Tocopheryl Acetate; Sodium Starch Octenylsuccinate, Citric Acid, BHT, Sodium Ascorbate,
Calcium Disodium EDTA, Hydrated Silica, Hexyl Cinnamal, Limonene, Linalool, Mannitol,
Benzyl Salicylate, Alpha-Isomethyl Ionone, Citronellol, Hydroxycitronellal, Benzyl Benzoate,
Geraniol, Cinnamyl Alcohol.
- Uses
- DOSAGE & ADMINISTRATION
- WARNINGS
- INDICATIONS & USAGE
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
MITCHUM CLINICAL SOFT SOLID
aluminum zirconium tetrachlorohydrex gly 20.0% gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:10967-658 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALUMINUM ZIRCONIUM TETRACHLOROHYDREX GLY (UNII: 8O386558JE) (ALUMINUM ZIRCONIUM TETRACHLOROHYDREX GLY - UNII:8O386558JE) ALUMINUM ZIRCONIUM TETRACHLOROHYDREX GLY 0.2 g in 1 g Inactive Ingredients Ingredient Name Strength CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) TRISILOXANE (UNII: 9G1ZW13R0G) WATER (UNII: 059QF0KO0R) MANNITOL (UNII: 3OWL53L36A) SILICA DIMETHYL SILYLATE (UNII: EU2PSP0G0W) DIMETHICONE (UNII: 92RU3N3Y1O) PETROLATUM (UNII: 4T6H12BN9U) ALOE VERA LEAF (UNII: ZY81Z83H0X) ALPHA-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) EDETATE CALCIUM DISODIUM ANHYDROUS (UNII: 8U5D034955) COUMARIN (UNII: A4VZ22K1WT) SODIUM STARCH GLYCOLATE TYPE A CORN (UNII: AG9B65PV6B) HYDRATED SILICA (UNII: Y6O7T4G8P9) HYDROXYISOHEXYL 3-CYCLOHEXENE CARBOXALDEHYDE (UNII: QUE43B9Z2Q) BENZYL SALICYLATE (UNII: WAO5MNK9TU) GERANIOL (UNII: L837108USY) CINNAMALDEHYDE (UNII: SR60A3XG0F) METHYL HEPTINE CARBONATE (UNII: 0TTP6YT2T3) LIMONENE, (+)- (UNII: GFD7C86Q1W) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) BENZYL BENZOATE (UNII: N863NB338G) EUGENOL (UNII: 3T8H1794QW) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) SODIUM ASCORBATE (UNII: S033EH8359) LINALOOL, (+)- (UNII: F4VNO44C09) .ALPHA.-HEXYLCINNAMALDEHYDE (UNII: 7X6O37OK2I) CITRAL (UNII: T7EU0O9VPP) HYDROXYCITRONELLAL (UNII: 8SQ0VA4YUR) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) TRIBEHENIN (UNII: 8OC9U7TQZ0) C18-36 ACID TRIGLYCERIDE (UNII: ZRA72DR3R7) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10967-658-16 45 g in 1 CANISTER; Type 0: Not a Combination Product 10/01/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part350 10/01/2018 Labeler - Revlon Consumer Products Corp. (788820165) Establishment Name Address ID/FEI Business Operations REVLON, INC. 809725570 manufacture(10967-658)