Label: DUAKLIR PRESSAIR- aclidinium bromide and formoterol fumarate powder, metered
- NDC Code(s): 0310-0900-30, 0310-0900-60
- Packager: AstraZeneca Pharmaceuticals LP
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated January 25, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use DUAKLIR PRESSAIR safely and effectively. See full prescribing information for DUAKLIR PRESSAIR.
DUAKLIR® PRESSAIR® (aclidinium bromide and formoterol fumarate) inhalation powder, for oral inhalation
Initial U.S. Approval: 2019INDICATIONS AND USAGE
DUAKLIR PRESSAIR is a combination of aclidinium bromide an anticholinergic, and formoterol fumarate, a long-acting beta2-adrenergic agonist (LABA) indicated for the maintenance treatment of patients with chronic obstructive pulmonary disease (COPD). (1)
Limitations of Use: Not indicated for the relief of acute bronchospasm or for the treatment of asthma. (1, 5.4)
DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHS
CONTRAINDICATIONS
- •
- Use of a long-acting beta2-adrenergic agonist (LABA), including formoterol fumarate, one of the active ingredients in DUAKLIR PRESSAIR, without an inhaled corticosteroid is contraindicated in patients with asthma. (1, 4)
- •
- Hypersensitivity to aclidinium bromide or formoterol fumarate or to any component of this product. (4, 5.5)
WARNINGS AND PRECAUTIONS
- •
- Asthma-related death: Long-acting beta2-adrenergic agonists as monotherapy (without an inhaled corticosteroid) for asthma increase the risk of serious asthma-related events. (5.1)
- •
- Do not initiate in acutely deteriorating COPD or to treat acute symptoms. (5.2)
- •
- Do not use in combination with an additional medicine containing a LABA because of risk of overdose. (5.3)
- •
- If paradoxical bronchospasm occurs, discontinue DUAKLIR PRESSAIR and institute alternative therapy. (5.4)
- •
- Use with caution in patients with cardiovascular disorders. (5.6)
- •
- Use with caution in patients with convulsive disorders, thyrotoxicosis, diabetes mellitus and ketoacidosis. (5.7)
- •
- Be alert to hypokalemia and hyperglycemia. (5.8)
- •
- Worsening of narrow-angle glaucoma may occur. Use with caution in patients with narrow-angle glaucoma and instruct patients to contact a physician immediately if symptoms occur. (5.9)
- •
- Worsening urinary retention may occur. Use with caution in patients with prostatic hyperplasia or bladder-neck obstruction and instruct patients to consult a physician immediately if symptoms occur. (5.10)
ADVERSE REACTIONS
Most common adverse reactions (incidence ≥ 3% and more common than with placebo) include: upper respiratory tract infection, headache and, back pain. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact AstraZeneca at 1-800-236-9933 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- •
- Use of other adrenergic by any route may potentiate the effect of DUAKLIR PRESSAIR. Use with caution. (5.3, 7.1)
- •
- Xanthine derivatives, steroids, diuretics or non-potassium sparing diuretics may potentiate hypokalemia or ECG changes. Use with caution. (7.2, 7.3)
- •
- Diuretics: Use with caution. Electrocardiographic changes and/or hypokalemia associated with non-potassium sparing diuretics may worsen with concomitant beta2-agonists. (7.3)
- •
- Monoamine oxidase inhibitors and tricyclic antidepressants: Use with extreme caution. May potentiate effect of formoterol fumarate on cardiovascular system. (7.4)
- •
- Beta-blockers: Use with caution and only when medically necessary. (7.5)
- •
- Anticholinergics: May interact additively with concomitantly used anticholinergic medications. Avoid administrations of DUAKLIR PRESSAIR with other anticholinergic-containing drugs. (7.6)
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 1/2022
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Serious Asthma-Related Events-Hospitalizations, Intubations, Death
5.2 Deterioration of Disease and Acute Episodes
5.3 Excessive Use of DUAKLIR PRESSAIR and Use with Other Long-Acting Beta2 Agonists
5.4 Paradoxical Bronchospasm
5.5 Immediate Hypersensitivity Reactions
5.6 Cardiovascular Effects
5.7 Coexisting Conditions
5.8 Hypokalemia and Hyperglycemia
5.9 Worsening of Narrow-Angle Glaucoma
5.10 Worsening of Urinary Retention
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
7 DRUG INTERACTIONS
7.1 Adrenergic Drugs
7.2 Xanthine Derivatives, Steroids
7.3 Non-Potassium Sparing Diuretics
7.4 Monoamine Oxidase Inhibitors and Tricyclic Antidepressants, QTc Prolonging Drugs
7.5 Beta-Blockers
7.6 Anticholinergics
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Hepatic Impairment
8.7 Renal Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Dose-Ranging Trials
14.2 Confirmatory Trials
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
DUAKLIR PRESSAIR is a combination of aclidinium bromide (an anticholinergic) and formoterol fumarate (a LABA) indicated for the maintenance treatment of patients with chronic obstructive pulmonary disease (COPD).
Limitations of Use: DUAKLIR PRESSAIR is not indicated for the relief of acute bronchospasm or for the treatment of asthma [see Warnings and Precautions (5.1, 5.4)].
- 2 DOSAGE AND ADMINISTRATION
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
Use of a long-acting beta2-adrenergic agonist (LABA), including formoterol fumarate, one of the active ingredients in DUAKLIR PRESSAIR, without an inhaled corticosteroid is contraindicated in patients with asthma [see Warnings and Precautions (5.1)]. DUAKLIR PRESSAIR is not indicated for the treatment of asthma.
DUAKLIR PRESSAIR is contraindicated in patients with:
- •
- Severe hypersensitivity to milk proteins [see Warnings and Precautions (5.5)].
- •
- Hypersensitivity to aclidinium bromide, formoterol fumarate, or to any component of the product [see Warnings and Precautions (5.5)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Serious Asthma-Related Events-Hospitalizations, Intubations, Death
- •
- The safety and efficacy of DUAKLIR PRESSAIR in patients with asthma have not been established. DUAKLIR PRESSAIR is not indicated for the treatment of asthma. [see Contraindications (4)].
- •
- Use of LABA as monotherapy [without inhaled corticosteroids (ICS)] for asthma is associated with an increased risk of asthma-related death. Available data from controlled clinical trials also suggest that use of LABA as monotherapy increases the risk of asthma-related hospitalization in pediatric and adolescent patients. These findings are considered a class effect of LABA monotherapy. When LABA are used in fixed-dose combination with ICS, data from large clinical trials do not show a significant increase in the risk of serious asthma-related events (hospitalizations, intubations, death) compared with ICS alone.
- •
- A 28-week, placebo-controlled, U.S. trial comparing the safety of another LABA (salmeterol) with placebo, each added to usual asthma therapy, showed an increase in asthma-related deaths in subjects receiving salmeterol (13/13,176 in subjects treated with salmeterol vs 3/13,179 in subjects treated with placebo; relative risk: 4.37 [95% CI: 1.25, 15.34]). The increased risk of asthma-related death is considered a class effect of LABAs, including formoterol fumarate, one of the active ingredients in DUAKLIR PRESSAIR.
- •
- No trial adequate to determine whether the rate of asthma-related deaths is increased in subjects treated with DUAKLIR PRESSAIR has been conducted.
- •
- Available data do not suggest an increased risk of death with use of LABA in patients with COPD.
5.2 Deterioration of Disease and Acute Episodes
DUAKLIR PRESSAIR should not be initiated in patients with acutely deteriorating COPD, which may be a life-threatening condition. DUAKLIR PRESSAIR has not been studied in patients with acutely deteriorating COPD. The use of DUAKLIR PRESSAIR in this setting is inappropriate.
DUAKLIR PRESSAIR is intended as twice daily maintenance treatment for COPD and should not be used for the relief of acute symptoms, i.e., as rescue therapy for treatment of acute episodes of bronchospasm. DUAKLIR PRESSAIR has not been studied in the relief of acute symptoms and extra doses should not be used for that purpose. Acute symptoms should be treated with an inhaled short-acting beta2-agonist.
When beginning treatment with DUAKLIR PRESSAIR, patients who have been taking oral or inhaled, short-acting beta2-agonists on a regular basis (e.g., four times a day) should be instructed to discontinue the regular use of these drugs and use them only for symptomatic relief of acute respiratory symptoms. When prescribing DUAKLIR PRESSAIR, the healthcare provider should also prescribe an inhaled, short-acting beta2-agonist and instruct the patient on how it should be used. Increasing inhaled beta2-agonist use is a signal of deteriorating disease for which prompt medical attention is indicated.
COPD may deteriorate acutely over a period of hours or chronically over several days or longer. If DUAKLIR PRESSAIR no longer controls symptoms of bronchoconstriction; the patient’s inhaled, short-acting beta2-agonist becomes less effective; or the patient needs more short-acting beta2-agonist than usual, these may be markers of deterioration of disease. In this setting, a re-evaluation of the patient and the COPD treatment regimen should be undertaken at once. Increasing the daily dose of DUAKLIR PRESSAIR beyond the recommended dose is not appropriate in this situation.
5.3 Excessive Use of DUAKLIR PRESSAIR and Use with Other Long-Acting Beta2 Agonists
As with other inhaled drugs containing beta-agonists, DUAKLIR PRESSAIR should not be used more often than recommended, at higher doses than recommended, or in conjunction with other medications containing LABAs, as an overdose may result. Clinically significant cardiovascular effects and fatalities have been reported in association with excessive use of inhaled sympathomimetic drugs. Patients using DUAKLIR PRESSAIR should not use another medicine containing a LABA for any reason [see Drug Interactions (7.1)].
5.4 Paradoxical Bronchospasm
Inhaled medicines, including DUAKLIR PRESSAIR, may cause paradoxical bronchospasm, which may be life threatening. If paradoxical bronchospasm occurs following dosing with DUAKLIR PRESSAIR, it should be treated immediately with an inhaled, short acting bronchodilator. DUAKLIR PRESSAIR should be discontinued immediately, and alternative therapies should be instituted.
5.5 Immediate Hypersensitivity Reactions
Immediate hypersensitivity reactions including anaphylaxis, angioedema (including swelling of the lips, tongue, or throat), urticaria, rash, bronchospasm, or itching, have occurred after administration of DUAKLIR PRESSAIR. If such a reaction occurs, therapy with DUAKLIR PRESSAIR should be stopped at once and alternative treatments should be considered.
5.6 Cardiovascular Effects
Formoterol fumarate, like other beta agonists, can produce a clinically significant cardiovascular effect in some patients as measured by increases in pulse rate, systolic or diastolic or blood pressure, or symptoms [see Clinical Pharmacology (12.2)]. If such effects occur, DUAKLIR PRESSAIR may need to be discontinued.
In addition, beta-agonists have been reported to produce ECG changes, such as flattening of the T wave, prolongation of the QTc interval, and ST segment depression, although the clinical significance of these findings is unknown. Therefore, DUAKLIR PRESSAIR should be used with caution in patients with severe cardiovascular disorders, especially coronary insufficiency, cardiac arrhythmias, and hypertension.
5.7 Coexisting Conditions
DUAKLIR PRESSAIR, like other medications containing sympathomimetic amines, should be used with caution in patients with convulsive disorders, thyrotoxicosis, and in those who are unusually responsive to sympathomimetic amines. Doses of the related beta-agonist albuterol, when administered intravenously, have been reported to aggravate pre-existing diabetes mellitus and ketoacidosis.
5.8 Hypokalemia and Hyperglycemia
Beta-agonist medications may produce significant hypokalemia in some patients, possibly through intracellular shunting, which has the potential to produce adverse cardiovascular effects [see Clinical Pharmacology (12.2)]. The decrease in serum potassium is usually transient, not requiring supplementation. Beta-agonist medications may produce transient hyperglycemia in some patients. In 4 clinical trials of 24 to 52 weeks duration evaluating DUAKLIR PRESSAIR in patients with COPD, there was no evidence of a treatment effect on serum glucose or potassium.
5.9 Worsening of Narrow-Angle Glaucoma
DUAKLIR PRESSAIR should be used with caution in patients with narrow-angle glaucoma. Prescribers and patients should be alert for signs and symptoms of acute narrow-angle glaucoma (e.g., eye pain or discomfort, blurred vision, visual halos, or colored images in association with red eyes from conjunctival congestion and corneal edema). Instruct patients to consult a physician immediately should any of these signs or symptoms develop.
5.10 Worsening of Urinary Retention
DUAKLIR PRESSAIR should be used with caution in patients with urinary retention or bladder neck obstruction. Prescribers and patients should be alert for signs and symptoms of urinary retention (e.g., difficulty passing urine, painful urination), especially in patients with prostatic hyperplasia or bladder neck obstruction. Instruct patients to consult a physician immediately should any of these signs or symptoms develop.
-
6 ADVERSE REACTIONS
LABAs, such as formoterol fumarate, one of the active ingredients in DUAKLIR PRESSAIR, increase the risk of asthma-related death. DUAKLIR PRESSAIR is not indicated for the treatment of asthma [see Warnings and Precautions (5.1)].
The following adverse reactions are described in greater detail elsewhere in the labeling:
- •
- Paradoxical bronchospasm [see Warnings and Precautions (5.4)]
- •
- Immediate hypersensitivity reactions [see Contraindications (4), Warnings and Precautions (5.5)]
- •
- Cardiovascular effects [see Warnings and Precautions (5.6)]
- •
- Worsening of narrow-angle glaucoma [see Warnings and Precautions (5.9)]
- •
- Worsening of urinary retention [see Warnings and Precautions (5.10)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The clinical program for DUAKLIR PRESSAIR included 6501 subjects with COPD in 2 placebo-controlled and 1 active-controlled 24-week lung function trials, one long-term safety extension study of 28 weeks and 2 other clinical trials. A total of 1893 subjects have received at least 1 dose of DUAKLIR PRESSAIR.
24-Week Trials
The frequency of common adverse reactions in Table 1 below is based upon pooled data from two, double-blind, placebo-controlled parallel group clinical trials (Trials 1 and 2, n=1729 and n=1669) in 3398 adult patients with moderate to severe COPD. Of these, 60% were male and 94% were Caucasian. They had a mean age of 64 years and an average smoking history of 46 pack-years, with 49% identified as current smokers. At screening, the mean post-bronchodilator percent predicted forced expiratory volume in 1 second (FEV1) was 54% (range: 28% to 80%) and the mean percent reversibility was 15% (range: -19% to 69%).
Table 1 shows all adverse reactions that occurred with a frequency of greater than or equal to 3% in the DUAKLIR PRESSAIR group in the two 24-week placebo-controlled trials where the rates in the DUAKLIR PRESSAIR group exceeded placebo.
Table 1: Adverse Reactions with DUAKLIR PRESSAIR ≥3% Incidence and More Common than with Placebo in Subjects with COPD - *
- Includes Viral Upper Respiratory Tract Infection and Upper Respiratory Tract Infection
Treatment
Adverse Reactions
Preferred Term
DUAKLIR PRESSAIR
(N=720)
%
Aclidinium
(N=722)
%
Formoterol
(N=716)
%
Placebo
(N=526)
%
Upper respiratory tract infection*
8.9
7.6
8.9
6.3
Headache
6.3
6.6
7.7
5.1
Back pain
3.8
3.3
3.5
3.4
Other adverse reactions reported in clinical studies with an incidence of >1% but less than 3% with DUAKLIR PRESSAIR but more common than with placebo were cough, sinusitis, influenza, tooth abscess, insomnia, dizziness, dry mouth, oropharyngeal pain, muscle spasms, musculoskeletal pain, arthralgia, pain in extremity, urinary tract infection, and blood creatine phosphokinase increased.
The adverse events reported in the 24-week active-controlled trial were consistent with those observed in 24-week placebo-controlled trials.
Long-Term Safety Extension Trial
In a 28-week safety extension trial, 918 subjects who successfully completed Trial 2 were treated for up to an additional 28 weeks for a total treatment period of up to 52 weeks with DUAKLIR PRESSAIR, aclidinium 400 mcg, formoterol fumarate 12 mcg administered twice daily or placebo. Because the subjects continued from Trial 2 into the safety extension trial, the demographic and baseline characteristics of the long-term safety extension trial were similar to those of the placebo-controlled efficacy trials described above. The adverse reactions reported in the long-term safety trial were consistent with those observed in the 24-week placebo-controlled trials.
-
7 DRUG INTERACTIONS
No formal drug interaction studies have been performed with DUAKLIR PRESSAIR.
7.1 Adrenergic Drugs
If additional adrenergic drugs are to be administered by any route, they should be used with caution because the sympathetic effects of formoterol, a component of DUAKLIR PRESSAIR, may be potentiated [see Warnings and Precautions (5.7)].
7.2 Xanthine Derivatives, Steroids
Concomitant treatment with xanthine derivatives, or steroids may potentiate any hypokalemic effect of beta-adrenergic agonists such as formoterol, a component of DUAKLIR PRESSAIR.
7.3 Non-Potassium Sparing Diuretics
The electrocardiographic changes and/or hypokalemia that may result from the administration of non-potassium-sparing diuretics (such as loop or thiazide diuretics) can be acutely worsened by beta-agonists, especially when the recommended dose of the beta-agonist is exceeded. Although the clinical significance of these effects is not known, caution is advised in the coadministration of beta-agonists with non–potassium-sparing diuretics.
7.4 Monoamine Oxidase Inhibitors and Tricyclic Antidepressants, QTc Prolonging Drugs
DUAKLIR PRESSAIR, as with other drugs containing beta2-agonists, should be administered with caution to patients being treated with monoamine oxidase inhibitors, tricyclic antidepressants, or other drugs known to prolong the QTc interval, because the action of adrenergic agonists on the cardiovascular system may be potentiated by these agents. Drugs that are known to prolong the QTc interval have an increased risk of ventricular arrhythmias.
7.5 Beta-Blockers
Beta-adrenergic receptor antagonists (beta-blockers) and DUAKLIR PRESSAIR may inhibit the effect of each other when administered concurrently. Beta-blockers not only block the therapeutic effects of beta2-agonists, such as formoterol, a component of DUAKLIR PRESSAIR, but may produce severe bronchospasm in COPD patients. Therefore, patients with COPD should not normally be treated with beta-blockers. However, under certain circumstances, e.g., as prophylaxis after myocardial infarction, there may be no acceptable alternatives to the use of beta-blockers in patients with COPD. In this setting, cardioselective beta-blockers could be considered, although they should be administered with caution.
7.6 Anticholinergics
There is a potential for an additive interaction with concomitantly used anticholinergic medications. Therefore, avoid coadministration of DUAKLIR PRESSAIR with other anticholinergic-containing drugs as this may lead to an increase in anticholinergic effects [see Warnings and Precautions (5.3, 5.4) and Adverse Reactions (6)].
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no adequate and well-controlled studies of DUAKLIR PRESSAIR or its individual components, formoterol fumarate or aclidinium bromide, in pregnant women to inform drug-associated risks.
No adverse developmental effects were seen with inhalation administration of aclidinium bromide to pregnant rats and rabbits during organogenesis at 15 or 20 times, respectively, the maximum recommended human daily inhaled dose (MRHDID). However, reduced pup weights were seen when pregnant rats continued inhalation administration through lactation at 5 times the MRHDID of aclidinium bromide. Adverse developmental effects occurred when rabbits were orally dosed with aclidinium bromide at approximately 1,400 times the MRHDID [see Data].
Formoterol fumarate alone, administered by the oral route, was teratogenic in rats and rabbits at 1,200 and 49,000 times the MRHDID, respectively. Formoterol fumarate was also embryocidal, increased pup loss at birth and during lactation, and decreased pup weight in rats at 85 times the MRHDID. These adverse effects generally occurred at large multiples of the MRHDID when formoterol fumarate was administered by the oral route to achieve high systemic exposures. No teratogenic, embryocidal, or developmental effects were seen in rats that received inhalation doses up to 280 times the MRHDID [see Data].
The estimated background risk of major birth defects and miscarriage of the indicated populations is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Clinical Considerations
Labor or Delivery
There are no well-controlled human studies that have investigated the effects of DUAKLIR PRESSAIR during labor and delivery. Because of the potential for beta-agonist interference with uterine contractility, use of DUAKLIR PRESSAIR during labor should be restricted to those patients in whom the benefits clearly outweigh the risk.
Data
Animal Data
Aclidinium bromide
In an embryo-fetal development study in pregnant rats dosed during the period of organogenesis from gestation days 6-17, no evidence of structural alterations was observed at approximately 15 times the maximum recommended human daily inhaled dose (MRHDID) [based on summed AUCs of aclidinium bromide and its metabolites at inhaled doses less than or equal to 5.0 mg/kg/day]. However, in a pre- and post-natal development study, decreased pup weights were observed when pregnant rats were exposed from gestation day 6 and continuing during the lactation period at approximately 5 times the MRHDID [based on summed AUCs of aclidinium bromide and its metabolites at inhaled doses greater than or equal to 0.2 mg/kg/day]. Maternal toxicity was also observed at inhaled doses greater than or equal to 0.2 mg/kg/day.
In an embryo-fetal development study in pregnant Himalayan rabbits administered inhaled doses of aclidinium bromide during the period of organogenesis from gestation days 6-19, no evidence of structural alterations was observed at approximately 20 times the MRHDID [based on summed AUCs of aclidinium bromide and its metabolites at inhaled doses less than or equal to 3.6 mg/kg/day]. However, in another embryo-fetal development study in pregnant Himalayan rabbits dosed orally from gestation days 6-19, increased incidences of additional liver lobes (3-5%), as compared to 0% in the control group, were observed at approximately 1,400 times the MRHDID [based on summed AUCs of aclidinium bromide and its metabolites at oral doses greater than or equal to 150 mg/kg/day], and decreased fetal body weights were observed at approximately 2,300 times the MRHDID [based on summed AUCs of aclidinium bromide and its metabolites at oral doses greater than or equal to 300 mg/kg/day]. These fetal findings were observed in the presence of maternal toxicity.
Formoterol fumarate
In a fertility and reproduction study, male rats were orally dosed for 9 weeks and females for 2 weeks prior to pairing and throughout the mating period. Females were either dosed up to gestation day 19 or up until weaning of their offspring. Males were dosed up to 25 weeks. Umbilical hernia was observed in rat fetuses at oral doses 1,200 times and greater than the MRHDID (on a mg/m2 basis at maternal oral doses of 3 mg/kg/day and higher). Brachygnathia, abnormal shortness of the mandible, was observed in rat fetuses at a dose 6,000 times the MRHDID (on a mg/m2 basis at a maternal oral dose of 15 mg/kg/day). Pregnancy was prolonged at a dose 6,000 times the MRHDID (on a mg/m2 basis at a maternal oral dose of 15 mg/kg/day). Fetal and pup deaths occurred at doses approximately 1,200 times the MRHDID and higher (on a mg/m2 basis at oral doses of 3 mg/kg/day and higher) during gestation.
In an embryo-fetal development study in pregnant rats dosed during the period of organogenesis from gestation days 6-15, no teratogenic, embryocidal or developmental effects were seen at doses up to 280 times the MRHDID (on a mg/m2 basis with maternal inhalation doses up to 0.69 mg/kg/day).
In an embryo-fetal development study in pregnant rabbits dosed during the period of organogenesis from gestation days 6-18, subcapsular cysts on the liver were observed in the fetuses at a dose 49,000 times the MRHDID (on a mg/m2 basis with a maternal oral dose of 60 mg/kg/day). No teratogenic effects were observed at doses up to 2,800 times the MRHDID (on a mg/m2 basis at maternal oral doses up to 3.5 mg/kg/day).
In a pre- and post-natal development study, pregnant female rats received formoterol at oral doses of 0, 0.21, 0.84, and 3.4 mg/kg/day from gestation day 6 through the lactation period. Pup survival was decreased from birth to postpartum day 26 at doses 85 times the MRHDID and higher (on a mg/m2 basis at maternal oral doses of 0.21 mg/kg/day and higher), although there was no evidence of a dose-response relationship. There were no treatment-related effects on the physical, functional, and behavioral development of rat pups.
8.2 Lactation
Risk Summary
There are no available data on the effects of DUAKLIR PRESSAIR, aclidinium bromide, or formoterol fumarate on the breastfed child or on milk production or presence in human milk. Both aclidinium bromide and formoterol fumarate are present in rat milk [see Data]. When a drug is present in animal milk, it is likely that the drug will be present in human milk. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for DUAKLIR PRESSAIR and any potential adverse effects on the breastfed child from DUAKLIR PRESSAIR or from the underlying maternal condition.
Data
In a pharmacokinetic study, levels of radioactivity in milk and plasma in rats were measured after a single intravenous dose of 1 mg/kg of radiolabeled aclidinium bromide on approximately post-natal day 14 [see Use in Specific Populations (8.1)]. The maximum concentration of radioactivity [14C aclidinium] in milk was measured at 6 hours post-dose and was found to be 10-14 times higher than in plasma.
In the fertility and reproduction study in rats, plasma levels of formoterol were measured in pups on post-natal day 15 [see Use in Specific Populations (8.1)]. It was estimated that the maximum plasma concentration that the pups received from the maternal animal, at the highest dose of 15 mg/kg, after nursing was 4.4% (0.24 nmol/L for a litter vs. 5.5 nmol/L for the mother).
8.4 Pediatric Use
DUAKLIR PRESSAIR is not indicated for use in children. The safety and effectiveness of DUAKLIR PRESSAIR in the pediatric population have not been established.
8.5 Geriatric Use
Based on available data for DUAKLIR PRESSAIR or its active components, no adjustment of dosage in geriatric patients is warranted [see Clinical Pharmacology (12.3)].
Of the 720 COPD patients exposed to DUAKLIR PRESSAIR for 24 weeks in two placebo-controlled clinical trials, 238 were less than 60 years, 301 were greater than or equal to 60 to less than 70 years, and 181 were greater than or equal to 70 years of age.
No overall differences in safety or effectiveness were observed between these subjects and younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
-
10 OVERDOSAGE
DUAKLIR PRESSAIR contains both aclidinium and formoterol fumarate; therefore, the risks associated with overdosage for the individual components described below apply to DUAKLIR PRESSAIR. The most common symptoms are blurred vision, dry mouth, nausea, muscle spasms, tremor, headache, palpitations, and systolic hypertension.
Treatment of overdosage consists of discontinuation of DUAKLIR PRESSAIR together with institution of appropriate symptomatic and/or supportive therapy.
-
11 DESCRIPTION
DUAKLIR PRESSAIR consists of a dry powder formulation of aclidinium bromide and formoterol fumarate for oral inhalation only.
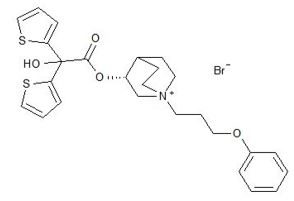
Aclidinium bromide, is an anticholinergic with specificity for muscarinic receptors. Aclidinium bromide is a synthetic, quaternary ammonium compound, chemically described as 1-azoniabicyclo[2.2.2]octane, 3-[(hydroxydi-2-thienylacetyl)oxy]-1-(3-phenoxypropyl)-, bromide, (3R)-. The structural formula is:
Aclidinium bromide is a white powder with a molecular formula of C26H30 NO4S2Br and a molecular mass of 564.56. It is very slightly soluble in water and ethanol and sparingly soluble in methanol.
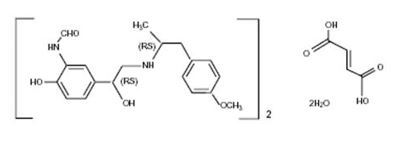
Formoterol fumarate, a racemate, is a selective beta2-adrenergic agonist. Its chemical name is (±)-2-hydroxy-5-[(1RS)-1-hydroxy-2-[[(1RS)-2-(4-methoxyphenyl)-1methylethyl]-amino]ethyl]formanilide fumarate dihydrate. The structural formula is:
Formoterol fumarate (as a dihydrate) has a molecular weight of 840.91, and its molecular formula is (C19H24N2O4)2• C4H4O4•2H2O. Formoterol fumarate is a white to yellowish crystalline powder, which is freely soluble in glacial acetic acid, soluble in methanol, sparingly soluble in ethanol and isopropanol, slightly soluble in water, and practically insoluble in acetone, ethyl acetate, and diethyl ether.
DUAKLIR PRESSAIR is a breath-actuated multi-dose dry powder inhaler. Each actuation of DUAKLIR PRESSAIR provides a metered dose of 12 mg of the formulation which contains lactose monohydrate (which may contain milk proteins) as the carrier, 400 mcg of aclidinium bromide (equivalent to 343 mcg of aclidinium), and 12.0 mcg of formoterol fumarate (as a dihydrate, equivalent to 11.5 mcg of formoterol fumarate anhydrous and 9.8 mcg of formoterol). This results in delivery of 396 mcg of aclidinium bromide (equivalent to 340 mcg of aclidinium) and 11.8 mcg of formoterol fumarate (as a dihydrate, equivalent to 11.3 mcg of formoterol fumarate anhydrous and 9.7 mcg of formoterol) from the mouthpiece, based on in vitro testing at an average flow rate of approximately 63 L/min with constant volume of 2 L. The amount of drug delivered to the lungs will vary depending on patient factors such as inspiratory flow rate and inspiratory time.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
DUAKLIR PRESSAIR
DUAKLIR PRESSAIR contains two bronchodilators: aclidinium a long-acting muscarinic antagonist (also known as an anticholinergic) and formoterol a long-acting beta2-adrenergic agonist. Further information regarding these two substances is provided below. The mechanism of action described below for the individual components apply to DUAKLIR PRESSAIR. These drugs represent two different classes of medications (a long-acting antimuscarinic agent and a selective long-acting beta2-adrenergic receptor agonist) that have different effects on clinical and physiological indices of COPD.
Aclidinium bromide
Aclidinium bromide is a long-acting antimuscarinic agent, which is often referred to as an anticholinergic. It has similar affinity to the subtypes of muscarinic receptors M1 to M5. In the airways, it exhibits pharmacological effects through inhibition of M3 receptors at the smooth muscle leading to bronchodilation. The competitive and reversible nature of antagonism was shown with human and animal origin receptors and isolated organ preparations. In preclinical in vitro as well as in vivo studies, prevention of acetylcholine-induced bronchoconstriction effects was dose-dependent and lasted longer than 24 hours. The clinical relevance of these findings is unknown. The bronchodilation following inhalation of aclidinium bromide is predominantly a site-specific effect.
Formoterol fumarate
Formoterol fumarate is a long-acting selective beta2-adrenergic receptor agonist (LABA) (beta2-agonist). Inhaled formoterol fumarate acts locally in the lung as a bronchodilator. In-vitro studies have shown that formoterol has more than 200-fold greater agonist activity at beta2-receptors than at beta1-receptors. The in-vitro binding selectivity to beta2-over beta1-adrenoceptors is higher for formoterol than for albuterol (5 times), whereas salmeterol has a higher (3 times) beta‑selectivity ratio than formoterol.
Although beta2-receptors are the predominant adrenergic receptors in bronchial smooth muscle and beta1-receptors are the predominant receptors in the heart, there are also beta2-receptors in the human heart comprising 10% to 50% of the total beta-adrenergic receptors. The precise function of these receptors has not been established, but they raise the possibility that even highly selective beta2-agonists may have cardiac effects.
The pharmacologic effects of beta2-adrenoceptor agonist drugs, including formoterol, are at least in part attributable to stimulation of intracellular adenyl cyclase, the enzyme that catalyzes the conversion of adenosine triphosphate (ATP) to cyclic-3', 5'-adenosine monophosphate (cyclic AMP). Increased cyclic AMP levels cause relaxation of bronchial smooth muscle and inhibition of release of mediators of immediate hypersensitivity from cells, especially from mast cells.
In vitro tests show that formoterol is an inhibitor of the release of mast cell mediators, such as histamine and leukotrienes, from the human lung. Formoterol also inhibits histamine-induced plasma albumin extravasation in anesthetized guinea pigs and inhibits allergen-induced eosinophil influx in dogs with airway hyper responsiveness. The relevance of these in vitro and animal findings to humans is unknown.
12.2 Pharmacodynamics
Cardiovascular effects
DUAKLIR PRESSAIR
The effect of DUAKLIR PRESSAIR on cardiac rhythm was assessed in a subset of subjects using 24-hour Holter monitoring in the 24-week placebo-controlled trials. The Holter monitoring population included 114 subjects on DUAKLIR PRESSAIR, 118 subjects on aclidinium bromide 400 mcg, 121 subjects on formoterol fumarate 12 mcg, and 81 subjects on placebo. No clinically meaningful effects on cardiac rhythm were observed.
12.3 Pharmacokinetics
Absorption
Following twice-daily oral inhalation administration of DUAKLIR PRESSAIR in COPD patients, mean maximum aclidinium and formoterol concentrations of 128 pg/mL and 17 pg/mL, respectively, occurred within 5 minutes after inhalation. Steady state was achieved within 5 days.
Distribution
Aclidinium: Aclidinium shows a volume of distribution of approximately 300 L following intravenous administration of 400 mcg in humans.
Formoterol: Over the concentration range of 10-500 nmol/L, plasma protein binding for the RR and SS enantiomers of formoterol was 46% and 58%, respectively. The concentrations of formoterol used to assess the plasma protein binding were higher than those achieved in plasma following inhalation of a single 54 mcg dose.
Elimination
Metabolism
Aclidinium: Clinical pharmacokinetics studies, including a mass balance study, indicate that the major route of metabolism of aclidinium is hydrolysis, which occurs both chemically and enzymatically by esterases.
Aclidinium is rapidly and extensively hydrolyzed in plasma to its alcohol and dithienylglycolic acid derivatives, neither of which binds to muscarinic receptors and are devoid of pharmacologic activity.
Therefore, due to the low plasma levels achieved at the clinically relevant doses, aclidinium and its metabolites are not expected to alter the disposition of drugs metabolized by the human CYP450 enzymes.
Formoterol:
Metabolism of formoterol is primarily by direct glucuronidation and by O-demethylation followed by conjugation to inactive metabolites. Secondary metabolic pathways include deformylation and sulfate conjugation. CYP2D6 and CYP2C have been identified as being primarily responsible for O-demethylation.
Excretion
Aclidinium: Total clearance was approximately 170 L/h after an intravenous dose of aclidinium bromide in young healthy volunteers with an inter-individual variability of 36%. Intravenously administered radiolabeled aclidinium bromide was administered to healthy volunteers and was extensively metabolized with 1% excreted as unchanged aclidinium. Approximately 54% to 65% of the radioactivity was excreted in urine and 20% to 33% of the dose was excreted in feces. The combined results indicated that almost the entire aclidinium bromide dose was eliminated by hydrolysis. After dry powder inhalation, urinary excretion of aclidinium is about 0.09% of the dose and the estimated effective half-life approximately 12 hours.
Formoterol: The excretion of formoterol was studied in four healthy subjects following simultaneous administration of radiolabeled formoterol via the oral and IV routes. In that study, 62% of the radiolabeled formoterol was excreted in the urine while 24% was eliminated in the feces.
Specific Populations
Geriatric Patients
The pharmacokinetic profile of aclidinium bromide and its main metabolites was assessed in 12 elderly COPD patients (aged 70 years or older) compared to a younger cohort of 12 COPD patients (40-59 years) that were administered 400 mcg aclidinium bromide once daily for 3 days via inhalation. No clinically significant differences in systemic exposure (AUC and Cmax) were observed when the two groups were compared. No dosage adjustment is necessary in elderly patients [see Use in Specific Populations (8.5)].
The pharmacokinetics of formoterol have not specifically been studied in geriatric patients.
Patients with Renal Impairment
The impact of renal disease upon the pharmacokinetics of aclidinium bromide was studied in 18 subjects with mild, moderate, or severe renal impairment. Systemic exposure (AUC and Cmax) to aclidinium and its main metabolites following single doses of 400 mcg aclidinium bromide was similar in renally impaired patients compared with 6 matched healthy control subjects. No dose adjustment is necessary in renally impaired patients [see Use in Specific Populations (8.6)]. Renal excretion of unchanged formoterol is low.
Patients with Hepatic Impairment
The effects of hepatic impairment on the pharmacokinetics of aclidinium bromide were not studied. However, hepatic insufficiency is not expected to have relevant influence on aclidinium bromide pharmacokinetics, since it is predominantly metabolized by chemical and enzymatic hydrolysis to products that do not bind to muscarinic receptors [see Use in Specific Populations (8.7)]. Specific data with formoterol is not available, but because formoterol is primarily eliminated via hepatic metabolism, an increased exposure can be expected in patients with severe liver impairment.
Drug Interactions Studies
Aclidinium: Formal drug interaction studies were not performed. In vitro studies using human liver microsomes indicated that aclidinium and its major metabolites do not inhibit CYP450, 1A2, 2A6, 2B6, 2C8, 2C9, 2C19, 2D6, 2E1, 3A4/5, or 4A9/11 at concentrations up to 1,000-fold higher than the maximum plasma concentration that would be expected to be achieved at the therapeutic dose. Therefore, it is unlikely that aclidinium causes CYP450 related drug interactions [see Drug Interactions (7)].
Formoterol: Specific drug-drug interaction studies with formoterol have not been performed.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies in animals with the combination of aclidinium bromide and formoterol fumarate, to evaluate the potential for carcinogenesis, mutagenesis, or impairment of fertility, have not been performed. The data described below is from studies conducted on the individual components.
Aclidinium Bromide
Two-year inhalation studies were conducted in mice and rats to assess the carcinogenic potential of aclidinium bromide. No evidence of tumorigenicity was observed in rats and mice at aclidinium doses up to 0.20 and 2.4 mg/kg/day, respectively [approximately 10 and 80 times the Maximum Recommended Human Daily Inhalation Dose (MRHDID), respectively, based on summed AUCs of aclidinium bromide and its metabolites].
Aclidinium bromide was positive in the in vitro bacterial gene mutation assay and the in vitro thymidine locus mouse lymphoma assay. However, aclidinium bromide was negative in the in vivo mouse micronucleus assay and the in vivo/in vitro unscheduled DNA synthesis assay with rat liver.
Aclidinium bromide impaired several fertility and reproductive performance indices (increased number of days to mate, decreased conception rate, decreased number of corpora lutea, increased pre-implantation loss with consequent decreased number of implantations and live embryos) in both male and female rats administered inhaled doses greater than or equal to 0.8 mg/kg/day [approximately 15 times the MRHDID based on summed AUCs of aclidinium bromide and its metabolites]. These adverse fertility effects were observed in the presence of paternal toxicity as evidenced by mortality and decreased body weight gain. However, there were no effects on mating index and sperm number and morphology. In the separate fertility assessments (treated males mated with untreated females; treated females mated with untreated males), no effect was observed in male and female rats at inhaled doses of 1.9 and 0.8 mg/kg/day, respectively [approximately 30 and 15 times the MRHDID, respectively, based on summed AUCs of aclidinium bromide and its metabolites].
Formoterol Fumarate
Long-term studies were conducted in mice using oral administration and rats using inhalation administration to evaluate the carcinogenic potential of formoterol fumarate. In a 24-month carcinogenicity study in CD-1 mice, formoterol fumarate at oral doses of 0.1 mg/kg and above [approximately 20 times the maximum recommended human daily inhalation dose (MRHDID) on a mg/m2 basis] caused a dose-related increase in the incidence of uterine leiomyomas.
In a 24-month carcinogenicity study in Sprague-Dawley rats, an increased incidence of mesovarian leiomyoma and uterine leiomyosarcoma were observed at the inhaled dose of 0.13 mg/kg (approximately 55 times the MRHDID on a mg/m2 basis). No tumors were seen at 0.022 mg/kg (approximately 9 times the MRHDID on a mg/m2 basis). Other beta-agonist drugs have similarly demonstrated increases in leiomyomas of the genital tract in female rodents. The relevance of these findings to human use is unknown.
Formoterol fumarate was not mutagenic or clastogenic in Ames Salmonella/microsome plate test, mouse lymphoma test, chromosome aberration test in human lymphocytes, and rat micronucleus test.
A reduction in fertility and/or reproductive performance was identified in male rats treated with formoterol at an oral dose of 15 mg/kg (approximately 6,000 times the MRHDID on a mg/m2 basis). In a separate study with male rats treated with an oral dose of 15 mg/kg (approximately 6,000 times the MRHDID on a mg/m2 basis), there were findings of testicular tubular atrophy and spermatic debris in the testes and oligospermia in the epididymides. No such effect was seen at 3 mg/kg (approximately 1,200 times the MRHDID on a mg/m2 basis). No effect on fertility was detected in female rats at doses up to 15 mg/kg (approximately 6,000 times the MRHDID on a mg/m2 basis).
-
14 CLINICAL STUDIES
The safety and efficacy of DUAKLIR PRESSAIR was evaluated in a clinical development program that included three dose ranging trials, one active and two placebo-controlled lung function trials of 24 weeks duration; and one 28-week long-term safety extension study. The efficacy of DUAKLIR was primarily based on one dose ranging trial in 128 subjects with COPD and the three 6-month duration confirmatory trials in 5015 subjects with COPD, including chronic bronchitis and emphysema.
14.1 Dose-Ranging Trials
Dose selection for DUAKLIR PRESSAIR for COPD was primarily based on data for the individual components, aclidinium bromide and formoterol fumarate, in COPD patients.
Aclidinium
Dose selection for aclidinium was supported by a 7-day, randomized, double-blind, active and placebo-controlled, crossover trial evaluating 3 doses of aclidinium bromide (400, 200, and 100 mcg) administered twice daily and an active control in 79 subjects with COPD. A dose ordering was observed, with the aclidinium bromide 400 mcg demonstrating larger improvements in FEV1 over 24 hours compared with aclidinium bromide 200 mcg and 100 mcg.
The change from baseline in FEV1 AUC0-12 from placebo after 7 days for the 100, 200, and 400 mcg doses were 154 mL (95% CI: 116, 192), 176 mL (95% CI: 137, 215), and 208 mL (95% CI: 170, 247), respectively. The results supported the selection of 400 mcg of aclidinium bromide twice daily in the confirmatory COPD trials.
Formoterol
Dose selection for formoterol was supported by a randomized, double-blind, placebo and active comparator (open-label) controlled, 5-period incomplete unbalanced crossover trial evaluating 3 doses of formoterol fumarate (24, 12, and 6 mcg) administered twice daily and an open-label active comparator in 132 subjects with COPD.
The change from baseline in FEV1 AUC0-12 from placebo after 7 days for the 6, 12, and 24 mcg doses were 108 mL (95% CI: 55, 161), 117 mL (95% CI: 64, 171), and 162 mL (95% CI: 107, 216), respectively. The results supported the evaluation of 12 mcg of formoterol twice daily in the confirmatory COPD trials.
14.2 Confirmatory Trials
The clinical development program for DUAKLIR PRESSAIR included two placebo-controlled [Trial 1 (NCT 01462942) and Trial 2 (NCT 01437397)] and one active-controlled [Trial 3 (NCT 02796677)] randomized, double-blind, parallel-group 24-week trials in subjects with moderate to very severe COPD, including chronic bronchitis and emphysema, designed to evaluate the efficacy of DUAKLIR PRESSAIR on lung function. The 24-week trials included 4,977 subjects >40 years of age that had a clinical diagnosis of COPD, with a smoking history greater than or equal to 10 pack-years, a post-albuterol FEV1 less than 80% of predicted normal values, and a ratio of FEV1/FVC of less than 0.7. The majority of these patients were male (61%) and Caucasian (94%) with a mean age of 64 years and an average smoking history of 46 pack-years (50% current smokers). During screening, mean post-bronchodilator percent predicted FEV1 was 53% (range: 10% to 86%) and mean percent reversibility was 15% (range: -33% to 121%).
Trials 1, 2, and 3 evaluated DUAKLIR PRESSAIR (aclidinium/formoterol fumarate) 400 mcg/12 mcg, aclidinium 400 mcg, and formoterol fumarate 12 mcg. Trials 1 and 2 included a placebo arm, and Trial 3 included an active, blinded, control arm.
The co-primary endpoints were change from baseline in trough FEV1 and change from baseline in one-hour post-dose FEV1 at Week 24 compared with formoterol fumarate 12 mcg and aclidinium 400 mcg, respectively.
In the three trials, DUAKLIR PRESSAIR demonstrated a statistically significant increase in mean change from baseline in trough FEV1 and change from baseline in one-hour post-dose FEV1 at Week 24 relative to formoterol fumarate 12 mcg and aclidinium 400 mcg, respectively (Table 2).
Table 2: Least Squares (LS) Mean Change from Baseline in 1-hour Morning Post dose FEV1 and Trough FEV1 at 24 weeks - *
- The placebo, aclidinium and formoterol fumarate comparators used the same inhaler and excipients as DUAKLIR PRESSAIR.
1-hour Post-dose FEV1 (L)
Trough FEV1 (L)
Difference from
Difference from
Treatment
Placebo*
(95% CI)
Aclidinium Bromide*
400 mcg
(95% CI)
Placebo*
(95% CI)
Formoterol Fumarate*
12 mcg
(95% CI)
Trial 1
DUAKLIR PRESSAIR
400 mcg/12 mcg
n=385
n=194
0.299 L
(0.255, 0.343)
n=383
0.125 L
(0.090, 0.160)
n=194
0.143 L
(0.101, 0.185)
n=383
0.085 L
(0.051, 0.119)
Trial 2
DUAKLIR PRESSAIR
400 mcg/12 mcg
n=335
n=331
0.284 L
(0.247, 0.320)
n=337
0.108 L
(0.073, 0.144)
n=331
0.130 L
(0.095, 0.165)
n=332
0.045 L
(0.011, 0.079)
Trial 3
DUAKLIR PRESSAIR
400 mcg/12 mcg
n=314
n=475
0.084 L
(0.051, 0.117)
n=319
0.055 L
(0.023, 0.088)
n = Number in the intent to treat population
With the limited data available, there were consistent improvements in trough FEV1 and one-hour post-dose FEV1 with respect to age, sex, degree of airflow limitation, reversibility, GOLD stage, smoking status, or inhaled corticosteroid use.
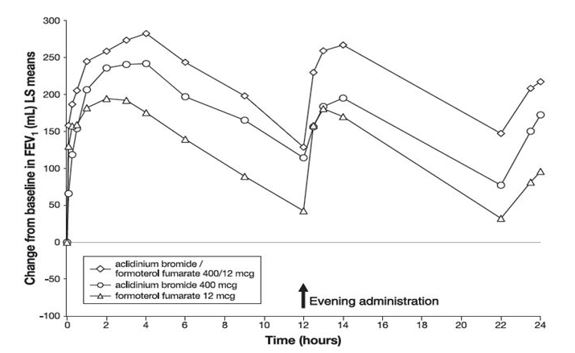
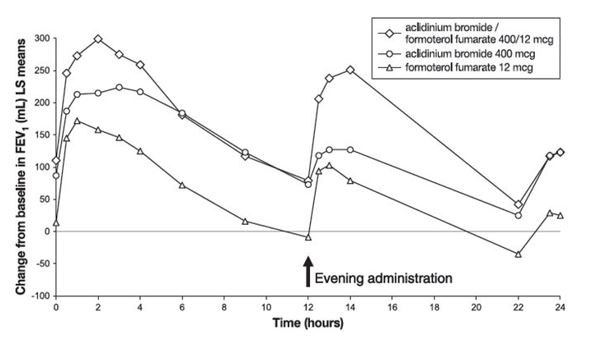
In Trial 3, serial spirometric evaluations were performed throughout the 24-hour dosing interval in a subset of subjects (n=563) at Day 1 and Week 24. Results from Trial 3 are shown in Figures 1 and 2.
Figure 1: FEV1 profile for DUAKLIR PRESSAIR (AB/FF 400/12 mcg), aclidinium bromide 400 mcg, formoterol fumarate 12 mcg, over a 24-hour dosing interval at Day 1 (active control not shown)
Figure 2: FEV1 profile for DUAKLIR PRESSAIR (AB/FF 400/12 mcg), aclidinium bromide 400 mcg, formoterol fumarate 12 mcg, over a 24-hour dosing interval at Week 24 (active control not shown)
In Trials 1 and 2, DUAKLIR PRESSAIR displayed an increase in FEV1 compared to placebo of 0.108 L (95% CI; 0.089, 0.127) and 0.128 L (95% CI; 0.111, 0.145) within 5 minutes after the first dose, respectively.
In Trials 1 and 2, patients treated with DUAKLIR PRESSAIR used less rescue medication compared to patients treated with placebo.
The St. George’s Respiratory Questionnaire (SGRQ) was assessed in Trials 1, 2, and 3. In Trial 1, the SGRQ responder rate (defined as an improvement in score of 4 or more as threshold) was 55.3%, 53.5%, 52.1%, and 53.2% for DUAKLIR PRESSAIR, aclidinium, formoterol fumarate, and placebo, respectively, with odds ratios of 1.12 (95% CI: 0.76, 1.67) for DUAKLIR PRESSAIR vs. aclidinium, 1.16 (95% CI: 0.78, 1.73) for DUAKLIR PRESSAIR vs. formoterol fumarate, and 1.12 (95% CI: 0.68, 1.84) for DUAKLIR PRESSAIR vs. placebo. In Trial 2, the SGRQ responder rate was 58.2%, 54.5%, 52.4%, and 38.7% for DUAKLIR PRESSAIR, aclidinium, formoterol fumarate, and placebo, respectively, with odds ratios of 1.03 (95% CI: 0.66, 1.62), 1.21 (95% CI: 0.77, 1.90), and 2.26 (95% CI: 1.41, 3.61), for DUAKLIR PRESSAIR vs. aclidinium, DUAKLIR PRESSAIR vs. formoterol fumarate, and DUAKLIR PRESSAIR vs. placebo, respectively. In Trial 3, the odds ratios were 0.96 (95% CI: 0.61, 1.51) and 0.97 (95% CI: 0.59, 1.58), for DUAKLIR PRESSAIR vs. aclidinium, DUAKLIR PRESSAIR vs. formoterol fumarate, respectively.
Exacerbations
A randomized, double-blind, placebo-controlled study of up to 36 months evaluated the efficacy of aclidinium bromide 400 mcg on COPD exacerbations in patients with moderate to very severe COPD with and without a history of exacerbations. The trial enrolled 3630 COPD patients, aged between 40 and 91 years, 58.7% were male and 90.6% were Caucasian, with a mean post-bronchodilator FEV1 of 47.7% of predicted value. The majority of patients had moderate (45.1%) or severe (40.2%) airflow obstruction.
The primary efficacy endpoint was the rate of moderate to severe exacerbations during the first year of treatment, defined as worsening of COPD symptoms (dyspnea, cough, sputum) for at least 2 consecutive days that required treatment with antibiotics and/or systemic corticosteroids or resulted in hospitalization or lead to death. Aclidinium bromide demonstrated a statistically significant reduction in the rate of on-study moderate to severe COPD exacerbations by 17% compared to placebo (rate ratio [RR] 0.83; 95% CI 0.73 to 0.94; p=0.003).
-
16 HOW SUPPLIED/STORAGE AND HANDLING
DUAKLIR PRESSAIR 400 mcg/12 mcg (aclidinium bromide and formoterol fumarate inhalation powder) is supplied in a sealed bag with a desiccant sachet and is available in 60 metered doses (NDC 0310-0900-60) and 30 metered doses (NDC 0310-0900-30).
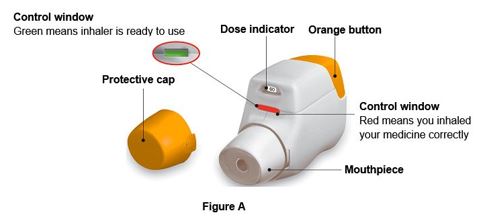
The active ingredients are administered using a multi-dose dry powder inhaler, PRESSAIR®, which delivers 60 doses or 30 doses of aclidinium bromide and formoterol fumarate powder for oral inhalation. The PRESSAIR inhaler is a white and orange colored device and is comprised of an assembled plastic dosing mechanism with a dose indicator, a drug-product storage unit containing the drug-product formulation, and a mouthpiece covered by an orange protective cap.
Store DUAKLIR PRESSAIR in a dry place at 20°C-25°C (68°F-77°F); excursions permitted to 15-30°C (59-86°F) [see USP Controlled Room Temperature]. Do not store the inhaler on a vibrating surface.
The PRESSAIR inhaler should be stored inside the sealed bag and only be opened immediately before use. Throw away the bag and the desiccant sachet.
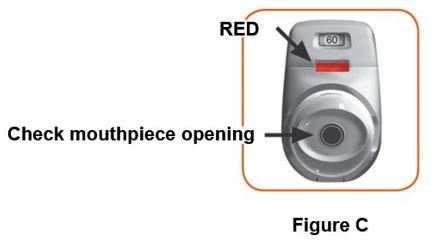
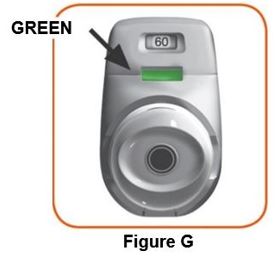
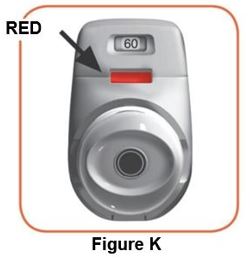
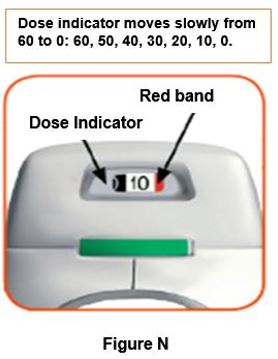
Throw away (dispose of) the PRESSAIR inhaler after the marking “0” with a red background shows in the middle of the dose indicator, when the device is empty and locks out, or 2 months after the date you opened the sealed bag that the inhaler comes in, whichever comes first.
Keep out of reach of children.
-
17 PATIENT COUNSELING INFORMATION
See FDA-approved Patient Labeling (Patient Information and Instructions for Use)
- •
- Serious Asthma-Related Events
Inform patients that LABAs, such as formoterol fumarate, one of the active ingredients in DUAKLIR PRESSAIR, when used alone (without an inhaled corticosteroid), increase the risk of serious asthma-related events, including asthma-related death. DUAKLIR PRESSAIR is not indicated for the treatment of asthma.
- •
- Not for Acute Symptoms
DUAKLIR PRESSAIR is not meant to relieve acute symptoms of COPD and extra doses should not be used for that purpose.
Instruct patients to seek medical attention immediately if they experience a worsening of symptoms or a need for more inhalations of their rescue inhaler than usual.
Instruct patients not to stop therapy with DUAKLIR PRESSAIR without physician/provider guidance since symptoms may recur after discontinuation.
- •
- Do Not Use Additional Long-Acting Beta2-Agonists
Instruct patients to not use other medicines containing a LABA. Patients should not use more than the recommended dose of DUAKLIR PRESSAIR.
Instruct patients who have been taking inhaled, short-acting beta-agonists on a regular basis to discontinue the regular use of these products and use them only for the symptomatic relief of acute symptoms.
- •
- Paradoxical Bronchospasm
Inform patients that DUAKLIR PRESSAIR can cause paradoxical bronchospasm. Advise patients that if paradoxical bronchospasm occurs, patients should discontinue DUAKLIR PRESSAIR.
- •
- Visual Effects
Eye pain or discomfort, blurred vision, visual halos, or colored images in association with red eyes from conjunctival congestion and corneal edema may be signs of acute narrow-angle glaucoma. Inform patients to consult a physician immediately should any of these signs and symptoms develop.
Inform patients that care must be taken not to allow the powder to enter into the eyes as this may cause blurring of vision and pupil dilation.
- •
- Urinary Retention
Difficulty passing urine and dysuria may be symptoms of new or worsening prostatic hyperplasia or bladder outlet obstruction. Patients should be instructed to consult a physician immediately should any of these signs or symptoms develop.
- •
- Immediate Hypersensitivity Reactions
Inform patients that anaphylaxis, angioedema (including swelling of the lips, tongue, or throat), urticaria, rash, bronchospasm, or itching, may occur after administration of DUAKLIR PRESSAIR. Advise patient to immediately discontinue treatment and consult a physician should any of these signs or symptoms develop.
- •
- Risks Associated with Beta2-Agonist Therapy
Inform patients of adverse effects associated with beta2-agonists, such as palpitations, chest pain, rapid heart rate, tremor, or nervousness.
- •
- Instructions for Administering DUAKLIR PRESSAIR
It is important for patients to understand how to correctly use DUAKLIR PRESSAIR. Instruct patients not to use more than the recommended dose of DUAKLIR PRESSAIR or with other medicines containing LABAs as excessive use may lead to significant cardiovascular effects including fatalities.
Inform patients that if they miss a dose, they should take their next dose at the usual time; they should not take 2 doses at one time.
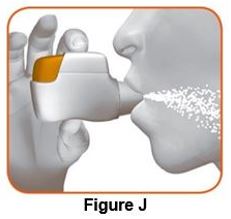
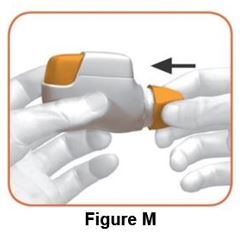
Instruct patients to thoroughly read complete Instructions for Use before using DUAKLIR PRESSAIR.
Distributed by:
AstraZeneca Pharmaceuticals LP,
Wilmington, DE 19850
Under license of ALMIRALL, S.A.
DUAKLIR® is a registered trademark of ALMIRALL and PRESSAIR® is a registered trademark of the AstraZeneca group of companies.
© AstraZeneca 2019
Patient Information
DUAKLIR™ PRESSAIR® (DU-aakl-ir PRESS-air)
(aclidinium bromide and formoterol fumarate)
inhalation powder, for oral Inhalation use
Important: Do not get DUAKLIR PRESSAIR in your eyes.
What is DUAKLIR PRESSAIR?
DUAKLIR PRESSAIR combines an anticholinergic, aclidinium bromide, and a long-acting beta2-adrenergic agonist (LABA medicine) formoterol fumarate.
- •
- Anticholinergic and LABA medicines help the muscles around the airways in your lungs stay relaxed to prevent symptoms such as wheezing, cough, chest tightness, and shortness of breath. These symptoms can happen when the muscles around the airways tighten. This makes it hard to breathe.
- •
- DUAKLIR PRESSAIR is a prescription medicine that is used to treat COPD. COPD is a chronic lung disease that includes chronic bronchitis, emphysema, or both.
- •
- DUAKLIR PRESSAIR is used long term as 1 inhalation 2 times each day to improve symptoms of COPD for better breathing and to reduce the number of flare-ups (the worsening of your COPD symptoms for several days).
- •
- DUAKLIR PRESSAIR is not for use to treat sudden symptoms of COPD. Always have a rescue inhaler (short-acting beta2 agonist medicine) with you to treat sudden symptoms of COPD. If you do not have a rescue inhaler, contact your healthcare provider to have one prescribed for you.
- •
- DUAKLIR PRESSAIR is not for the treatment of asthma. It is not known if DUAKLIR PRESSAIR is safe and effective in people with asthma.
- •
- DUAKLIR PRESSAIR should not be used in children. It is not known if DUAKLIR PRESSAIR is safe and effective in children.
Do not use DUAKLIR PRESSAIR if you:
- •
- have asthma and you use DUAKLIR PRESSAIR without also using a medicine called an inhaled corticosteroid.
- •
- have a severe allergy to milk proteins. Ask your healthcare provider if you are not sure.
- •
- are allergic to aclidinium bromide, formoterol fumarate, or any of the ingredients in DUAKLIR PRESSAIR. See “What are the ingredients in DUAKLIR PRESSAIR?” below for a complete list of ingredients.
Before you use DUAKLIR PRESSAIR tell your healthcare provider about all of your medical conditions, including if you:
- •
- have heart problems
- •
- have high blood pressure
- •
- have seizures
- •
- have thyroid problems
- •
- have diabetes
- •
- have eye problems especially glaucoma. DUAKLIR PRESSAIR may make your glaucoma worse.
- •
- have prostate or bladder problems, or problems passing urine. DUAKLIR PRESSAIR may make these problems worse.
- •
- are pregnant or plan to become pregnant. It is not known if DUAKLIR PRESSAIR can harm your unborn baby.
- •
- are breastfeeding or plan to breastfeed. It is not known if the medicines, aclidinium bromide and formoterol fumarate in DUAKLIR PRESSAIR pass into your breast milk and if they can harm your baby.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
DUAKLIR PRESSAIR and certain other medicines may interact with each other. This may cause serious side effects.
Especially tell your healthcare provider if you take:
- •
- anticholinergics (including umeclidinium, tiotropium, ipratropium, aclidinium, glycopyrrolate).
- •
- other LABA medicines (including formoterol, arformoterol, salmeterol, vilanterol, indacaterol, olodaterol).
- •
- atropine
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine.
How should I use DUAKLIR PRESSAIR?
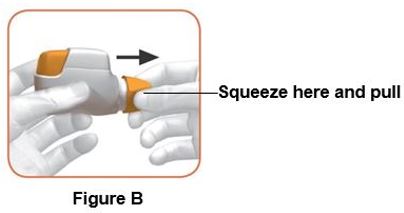
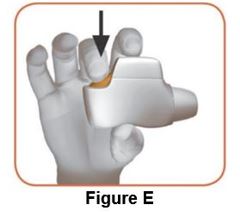
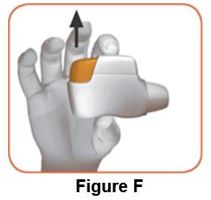
Read the step-by-step instructions for using DUAKLIR PRESSAIR at the end of this Patient Information.
- •
- Do not use DUAKLIR PRESSAIR unless your healthcare provider has taught you how to use the inhaler and you understand how to use it correctly.
- •
- Use DUAKLIR PRESSAIR exactly as prescribed. Do not use DUAKLIR PRESSAIR more often than prescribed.
- •
- Use 1 inhalation 2 times each day. Use DUAKLIR PRESSAIR 1 time in the morning and 1 time in the evening.
- •
- If you miss a dose of DUAKLIR PRESSAIR skip the missed dose. Take your next dose at your usual time. Do not take more than 1 inhalation 2 times each day. Do not take 2 doses at one time.
- •
- If you take too much DUAKLIR PRESSAIR, call your healthcare provider and get medical help right away if you have any unusual symptoms, such as worsening shortness of breath, chest pain, increased heart rate, or shakiness.
- •
- Do not use other medicines that contain a LABA for any reason. Ask your healthcare provider or pharmacist if any of your other medicines are LABA medicines.
- •
- Do not stop using DUAKLIR PRESSAIR unless told to do so by your healthcare provider because your symptoms might get worse. Your healthcare provider will change your medicines as needed.
- •
- DUAKLIR PRESSAIR does not relieve sudden symptoms. Always have a rescue inhaler with you to treat sudden symptoms. If you do not have a rescue inhaler, call your healthcare provider to have one prescribed for you.
- •
- Call your healthcare provider or get medical care right away if your breathing problems get worse, you need to use your rescue inhaler more often than usual, your rescue inhaler does not work as well to relieve your symptoms.
What are the possible side effects of DUAKLIR PRESSAIR?
DUAKLIR PRESSAIR can cause serious side effects including:
- •
-
people with asthma who take LABA medicines, such as formoterol fumarate (one of the medicines in DUAKLIR PRESSAIR), without also using a medicine called an inhaled corticosteroid, have an increased risk of serious problems from asthma, including being hospitalized, needing a tube placed in their airway to help them breathe or death.
- ∘
-
Call your healthcare provider if breathing problems worsen over time with using DUAKLIR PRESSAIR.
You may need a different treatment. - ∘
-
Get emergency medical care if:
- •
- your breathing problems worsen quickly
- •
- you use your rescue inhaler, but it does not relieve your breathing problems
- •
- COPD symptoms that get worse over time. If your COPD symptoms worsen over time, do not increase your dose of DUAKLIR PRESSAIR, call your healthcare provider.
- •
-
using too much of a LABA medicine may cause:
- ∘
- chest pain
- ∘
- fast and irregular heartbeat
- ∘
- tremor
-
- ∘
- increased blood pressure
- ∘
- headache
- ∘
- nervousness
- •
- sudden breathing problems immediately after use of DUAKLIR PRESSAIR. These sudden breathing problems may be life-threatening. If you have sudden breathing problems immediately after inhaling DUAKLIR PRESSAIR, stop using DUAKLIR PRESSAIR and call your healthcare provider right away or go to the nearest hospital emergency room.
- •
- serious allergic reactions. Call your doctor or get emergency medical care if you get any symptoms of a serious allergic reaction.
- ∘
- rash
- ∘
- hives
- ∘
- swelling of the face, mouth and tongue
- ∘
- breathing problems
- •
- effects on your heart:
- ∘
- chest pain
- ∘
- increased blood pressure
- ∘
- fast or irregular heartbeat
- •
- changes in laboratory blood levels: high blood sugar (hyperglycemia) and low blood potassium (hypokalemia), this last may cause symptoms of muscle spasms, muscle weakness, or abnormal heart rhythm.
- •
- new or worsened increased pressure in your eyes (acute narrow-angle glaucoma). Acute narrow-angle glaucoma can lead to permanent loss of vision if not treated. Symptoms of acute narrow-angle glaucoma may include:
- ∘
- eye pain or discomfort
- ∘
- seeing halos or bright colors around lights
- ∘
- nausea or vomiting
- ∘
- red eyes
- ∘
- blurred vision
If you have these symptoms, stop using DUAKLIR PRESSAIR and call your healthcare provider right away.
- •
- new or worsened urinary retention. Urinary retention can be caused by blockage in your bladder or, if you are a male, a larger than normal prostate. Symptoms of urinary retention may include:
- ∘
- difficulty urinating
- ∘
- painful urination
- ∘
- urinating frequently
- ∘
- urination in a weak stream or drips
If you have these symptoms of urinary retention, stop using DUAKLIR PRESSAIR and call your doctor right away.
The most common side effects of DUAKLIR PRESSAIR include: upper respiratory tract infection, headache and, back pain.
Tell your healthcare provider about any side effect that bothers you or does not go away.
These are not all the possible side effects of DUAKLIR PRESSAIR. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store DUAKLIR PRESSAIR?
- •
- Store DUAKLIR PRESSAIR at room temperature between 68°F to 77°F (20°C to 25°C) in the sealed bag that it comes in until you are ready to use DUAKLIR PRESSAIR. Do not open the sealed bag until you are ready to use a dose of DUAKLIR PRESSAIR.
- •
- Keep DUAKLIR PRESSAIR in a dry place.
- •
- Do not store the inhaler on a vibrating surface.
- •
- Throw away (dispose of) DUAKLIR PRESSAIR® and use a new one:
- ∘
- when the marking “0” with a red background shows in the middle of the dose indicator,
- ∘
- if your inhaler is empty and locks out, or
- ∘
- 2 months after the date you opened the sealed bag that the inhaler comes in, whichever comes first.
- Keep DUAKLIR PRESSAIR and all medicines out of the reach of children.
General information about the safe and effective use of DUAKLIR PRESSAIR
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use DUAKLIR PRESSAIR for a condition for which it was not prescribed. Do not give your DUAKLIR PRESSAIR to other people, even if they have the same symptoms that you have. It may harm them.
You can ask your doctor or pharmacist for information about DUAKLIR PRESSAIR that is written for healthcare professionals.
What are the ingredients in DUAKLIR PRESSAIR?
Active ingredient: aclidinium bromide and formoterol fumarate
Inactive ingredient: lactose monohydrate
Distributed by: AstraZeneca Pharmaceuticals LP, Wilmington, DE 19850
©AstraZeneca 2019
For more information go to www.DUAKLIR.com or call 1-800-236-9933
This Patient Information has been approved by the U.S. Food and Drug Administration 03/2019
-
PATIENT PACKAGE INSERT
-
PACKAGE/LABEL PRINCIPAL DISPLAY PANEL
NDC 0310-0900-60
Rx Only
Duaklir® Pressair®
(aclidinium bromide and formoterol fumarate) inhalation powder
400 mcg/12 mcg per actuation
For Oral Inhalation Only
*Each metered dose contains 400 mcg of aclidinium bromide
(equivalent to 343 mcg of aclidinium), and 12.0 mcg of formoterol
fumarate (as a dihydrate, equivalent to 9.8 mcg of formoterol)
from the device reservoir.
60 Metered Doses
Manufactured for: AstraZeneca Pharmaceuticals LP.
Wilmington, DE 19850
Manufactured by: AstraZeneca AB,
SE-151 85 Södertälje, Sweden
-
INGREDIENTS AND APPEARANCE
DUAKLIR PRESSAIR
aclidinium bromide and formoterol fumarate powder, meteredProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0310-0900 Route of Administration RESPIRATORY (INHALATION) Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACLIDINIUM BROMIDE (UNII: UQW7UF9N91) (ACLIDINIUM - UNII:K17VY42F6C) ACLIDINIUM BROMIDE 400 ug FORMOTEROL FUMARATE (UNII: W34SHF8J2K) (FORMOTEROL - UNII:5ZZ84GCW8B) FORMOTEROL FUMARATE 12 ug Inactive Ingredients Ingredient Name Strength LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0310-0900-60 1 in 1 CARTON 08/17/2022 04/30/2025 1 1 in 1 POUCH 1 60 in 1 INHALER; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) 2 NDC:0310-0900-30 1 in 1 CARTON 08/17/2022 09/30/2024 2 1 in 1 POUCH 2 30 in 1 INHALER; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA210595 08/17/2022 04/30/2025 Labeler - AstraZeneca Pharmaceuticals LP (054743190) Registrant - AstraZeneca PLC (230790719)