Label: CYCLAVANCE- cyclosporine oral solution solution
- NDC Code(s): 51311-994-15, 51311-994-50
- Packager: Virbac AH Inc
- Category: PRESCRIPTION ANIMAL DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Animal Drug Application
Drug Label Information
Updated April 29, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- CAUTION:
-
DESCRIPTION:
CYCLAVANCE® (cyclosporine oral solution) USP MODIFIED is an oral form of cyclosporine that immediately forms a microemulsion in an aqueous
environment. Cyclosporine, the active ingredient in CYCLAVANCE, is a cyclic polypeptide, immune modulating agent consisting of 11 amino acids. It is produced as a metabolite by the fungal species Beauveria nivea.
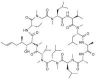
Chemically, cyclosporine A is designated Cyclo[[(E)-(2S,3R,4R)-3-hydroxy-4-methyl-2-(methylamino)-6-octenoyl]-L-2-aminobutyryl-N-methylglycyl-N-methyl-L-leucyl-Lvalyl-N-methyl-L-leucyl-L-alanyl-D-alanyl-N-methyl-L-leucyl-N-methyl-L-leucyl-N-methyl-L-valyl].The structural formula is:
- INDICATIONS:
-
DOSAGE AND ADMINISTRATION:
Always Provide the Instructions for Assembling the Dispensing System and Preparing a Dose of CYCLAVANCE and the Information
for Dog Owners with the prescription. The initial dose of CYCLAVANCE is 5 mg/kg/day as a single daily dose for 30 days. Following this initial daily treatment period, the dose of CYCLAVANCE may be tapered by decreasing the frequency of dosing to every other day or twice weekly, until a minimum frequency is reached which will maintain the desired therapeutic effect. CYCLAVANCE should be given at least one hour before or two hours after a meal. If a dose is missed, the next dose should be administered (without doubling) as soon as possible but dosing should be no more frequent than once daily. The dispensing system for the 5 and 15 mL vial sizes includes a 1 mL oral dosing syringe graduated in 0.05 mL increments. To dose the dog, administer 0.05 mL of CYCLAVANCE per 2.2 lbs of body weight. The dispensing system for the 30 and 50 mL vial sizes includes both a 1 mL oral dosing syringe graduated in 0.05 mL increments, and a 3 mL oral dosing syringe graduated in 0.1 mL increments. To dose the dog, administer 0.1 mL of CYCLAVANCE per 4.4 lbs of body weight. Do not rinse or clean the oral dosing syringe between uses. (See Instructions for Assembling the Dispensing System and Preparing a Dose of CYCLAVANCE.) - CONTRAINDICATIONS:
-
WARNINGS:
CYCLAVANCE (cyclosporine oral solution) is a systemic immunosuppressant that may increase the susceptibility to infection and the development of neoplasia.
HUMAN WARNINGS: Not for human use. Keep this and all drugs out of reach of children. For use only in dogs. Special precautions to be taken when administering CYCLAVANCE in dogs: Do not eat, drink, smoke, or use smokeless tobacco while handling CYCLAVANCE. Wear gloves during administration. Wash hands after administration. In case of accidental ingestion, seek medical advice immediately and provide the package insert or the label to the physician. People with known hypersensitivity to cyclosporine should avoid contact with CYCLAVANCE. People with known hypersensitivity to cyclosporine should avoid contact with CYCLAVANCE.
-
PRECAUTIONS:
The safety and effectiveness of cyclosporine has not been established in dogs less than 6 months of age or less than 4 lbs body weight. CYCLAVANCE is not for use in breeding dogs, pregnant or lactating bitches. As with any immunomodulation regimen, exacerbation of sub-clinical neoplastic and infectious conditions may occur. Gastrointestinal problems and gingival hyperplasia may occur at the initial recommended dose (See Animal Safety).
CYCLAVANCE may cause elevated levels of serum glucose, and should be used with caution in cases with diabetes mellitus. If signs of diabetes mellitus develop following the use of CYCLAVANCE, consideration should be given to tapering or discontinuing the dose.
CYCLAVANCE should be used with caution with drugs that affect the P-450 enzyme system. Simultaneous administration of CYCLAVANCE with drugs that suppress the P-450 enzyme system, such as azoles (e.g. ketoconazole), may lead to increased plasma levels of cyclosporine.
Since the effect of cyclosporine use on dogs with compromised renal function has not been studied, CYCLAVANCE should be used with caution in dogs with renal insufficiency.
There have been reports of convulsions in human adult and pediatric patients receiving cyclosporine, particularly in combination with high dose methylprednisolone (See Animal Safety).
Killed vaccines are recommended for dogs receiving CYCLAVANCE because the impact of cyclosporine on the immune response to modified live vaccines is unknown (See Animal Safety).
-
ADVERSE REACTIONS:
A total of 265 dogs were included in the field study safety analysis. One hundred and eleven (111) dogs were treated with placebo for the first 30
days. For the remainder of the study, all dogs received cyclosporine capsules.Fourteen dogs withdrew from the study due to adverse reactions. Four dogs withdrew from the study after vomiting. One dog each withdrew from the study after diarrhea; vomiting, diarrhea and pruritus; vomiting, depression and lethargy; lethargy, anorexia and hepatitis; gingival hyperplasia, lethargy, polyuria/polydipsia and soft stool; seizure; sebaceous cyst; pruritus; erythema; or otitis externa.
Vomiting and diarrhea were the most common adverse reactions occurring during the study. In most cases, signs spontaneously resolved with continued dosing. In other cases, temporary dose modifications (brief interruption in dosing, divided dosing, or administration with a small amount of food) were employed to resolve signs.
Persistent otitis externa, urinary tract infections, anorexia, gingival hyperplasia, lymphadenopathy and lethargy were the next most frequent adverse events observed. Gingival hyperplasia regressed with dose tapering. Owners of four dogs reported seizures while dogs were receiving cyclosporine. In one dog, seizures were the result of a brain tumor diagnosed one month into the study. Another dog experienced seizures before and after the study.
Otitis externa, allergic otitis, or pinna erythema, with or without exudates, commonly accompanies atopy. Many dogs entered the study with otitis externa, which did not resolve without otic treatment. New cases of otitis externa, allergic otitis, or pinna erythema developed while dogs were receiving cyclosporine. However, the incidence rate was lower with cyclosporine compared to placebo. A change in the dose frequency was not necessary when new cases occurred.
Number of Dogs Displaying Each Clinical Observation in the Field Study Clinical sign % out of 265 Vomiting 30.9% Diarrhea 20.0% Persistent Otitis Externa 6.8% Urinary Tract Infection 3.8% Anorexia 3.0% Lethargy 2.3% Gingival Hyperplasia 2.3% Lymphadenopathy 2.3% The following clinical signs were reported in less than 2% of dogs treated with cyclosporine in the field study: constipation, flatulence, Clostridial organisms in the feces, nausea, regurgitation, polyuria/polydipsia, strong urine odor, proteinuria, pruritus, erythema/flushed appearance, pyoderma, sebaceous adenitis, crusty dermatitis, excessive shedding, coarse coat, alopecia, papillomas, histiocytoma, granulomatous mass or lesion, cutaneous cyst, epulis, benign epithelial tumor, multiple hemangioma, raised nodule on pinna, seizure, shaking/trembling, hind limb twitch, panting, depression, irritability, hyperactivity, quieter, increased light sensitivity, reluctance to go outside, weight loss, hepatitis.
The following clinical signs were observed in 1.5-4.5% of dogs while receiving the placebo: vomiting, diarrhea and urinary tract infection. The following clinical signs were observed in less than 1% of dogs receiving the placebo: anorexia, otitis externa, cutaneous cysts, corneal opacity, lymphadenopathy, erythema/flushed appearance.
Clinical Pathology Changes: During the study, some dogs experienced changes in clinical chemistry parameters while receiving cyclosporine, as described in the following table:Clinical Chemistry % Affected (out of 265) Elevated Creatinine 7.8% Hyperglobulinemia 6.4% Hyperphosphatemia 5.3% Hyperproteinemia 3.4% Hypercholesterolemia 2.6% Hypoalbuminemia 2.3% Hypocalcemia 2.3% Elevated BUN 2.3% In addition, the following changes in clinical chemistry parameters were noted in less than 2% of dogs: hypernatremia; hyperkalemia, elevated ALT, elevated ALP, hypercalcemia and hyperchloremia. These clinical pathology changes were generally not associated with clinical signs.
POST-APPROVAL EXPERIENCE: The following adverse events are based on post-approval adverse drug experience reporting. Not all adverse reactions are reported to FDA CVM. It is not always possible to reliably estimate the adverse event frequency or establish a causal relationship to product exposure using this data. The following adverse events are grouped by body system and are presented in decreasing order of reporting frequency.
Gastrointestinal: Emesis, diarrhea, gingival hyperplasia, hemorrhagic diarrhea, abdominal pain, hematemesis, digestive tract hemorrhage, hypersalivation, retching, flatulence, tenesmus, intestinal stasis, digestive tract hypermotility, melena, pancreatitis, involuntary defecation
General: Lethargy, anorexia, weight loss, polydipsia, hyperthermia, pale mucous membrane, general pain, collapse, dehydration, edema
Dermatologic: Pruritus, dermatitis and eczema, alopecia, erythema, papilloma, bacterial skin infection, skin lesion, skin and/or appendage neoplasm, pigmentation disorder, hair change, hyperkeratosis, histiocytoma, fungal skin infection, dermal cyst(s), desquamation
Behavioral: Hyperactivity, behavioral changes, anxiety, vocalization, aggression, inappropriate urination, disorientation
Neurologic: Muscle tremor, convulsion, ataxia, paresis
Respiratory: Tachypnea, dyspnea, cough
Urologic: Polyuria, urine abnormalities (hematuria, urinary tract infection, proteinuria, glucosuria, decreased urine concentration) urinary incontinence, cystitis, renal failure, renal insufficiency
Immune: Urticaria, anaphylaxis, allergic edema
Blood and lymphatic: Lymphadenopathy, anemia, hypoalbuminemia, leukopenia
Hepatic: Elevated Liver Enzymes, hepatopathy, hepatomegaly, hepatitis
Musculoskeletal: Lameness, limb weakness, myositis
Ear and labyrinth: Otitis externa
Cardio-vascular: Tachycardia
Endocrine: Diabetes mellitus, hyperglycemia
In some cases, death/euthanasia has been reported as an outcome of the adverse events listed above.Neoplasms have been reported in dogs taking cyclosporine, including reports of lymphoma/lymphosarcoma and mast cell tumor. It is unknown if these were preexisting or developed de novo while on cyclosporine.
Diabetes mellitus has been reported; West Highland White Terriers are the most frequently reported breed.
To report suspected adverse drug events, for technical assistance or to obtain a copy of the Safety Data Sheet (SDS), contact Virbac AH, Inc. at 1-800-338-3659 or us.virbac.com. For additional information about adverse drug experience reporting for animal drugs, contact FDA at 1-888-FDA-VETS or http://www.fda.gov/reportanimalae.
-
CLINICAL PHARMACOLOGY:
Cyclosporine is a immunosuppressive agent that has been shown to work via suppression of T-helper and T-suppressor cells and inhibition of interleukin-2. It does not depress hematopoiesis or the function of phagocytic cells. A decrease in CD4 and CD8 cells was not seen in dogs receiving 20 mg/kg/day of cyclosporine for 56 days. Cyclosporine is not a corticosteroid or an antihistamine.
METABOLISM: Cyclosporine is extensively metabolized by the cytochrome P-450 enzyme system in the liver, and to a lesser degree in the gastrointestinal tract and the kidney. The metabolism of cyclosporine can be altered by the co-administration of a variety of agents (See Precautions).
-
EFFECTIVENESS FIELD STUDY:
A multisite, placebo controlled, double masked, field study was conducted in the United States and Canada using 16 investigators. Two hundred sixty five (265) dogs aged 1-10 years, weighing 4-121 lbs received either cyclosporine capsules at 5 mg/kg/day or placebo capsules. After 30 days, placebo dogs were switched to cyclosporine capsules.
Dogs were treated with cyclosporine capsules for a total of 4 months. No additional therapy with antihistamines, corticosteroids or medicated shampoos was permitted. Evaluations for pruritus and for skin lesions to derive a Canine Atopic Dermatitis Extent and Severity Index (CADESI) score occurred at enrollment and at monthly intervals. One hundred ninety-two (192) dogs were included in the statistical analysis of
effectiveness.At the end of the 30 day placebo controlled period, CADESI scores of dogs treated with cyclosporine capsules improved by 45% from enrollment, while CADESI scores of dogs treated with placebo worsened by 9%. Seventy-four percent (74%) of cyclosporine capsule treated dogs showed improvement in their pruritus scores over the first 30 day period, while only 24% of the placebo treated dogs showed an improvement.
Owner and Veterinary Global Assessment in response to treatment also demonstrated statistically significant (p<0.0001) improvement. After 4 weeks of therapy, Owner and Veterinary Global Assessments showed approximately twice as much improvement in the cyclosporine capsule treated dogs as compared to placebo treated dogs.Improvements in pruritus accompanied by 50% or 75% improvements in CADESI scores resulted in dose reductions to every other day or twice weekly respectively. Not all dogs were able to decrease to twice weekly dosing. Some animals required upward or downward dosage adjustments during the study. Such adjustments should be expected during therapy of this disease. Dogs unable to decrease from once daily dosing after
60 days were considered dose reduction failures for the purposes of the study.
The results of dose assignments, based on the study criteria, for each 4-week dosing period, are shown in the graph below.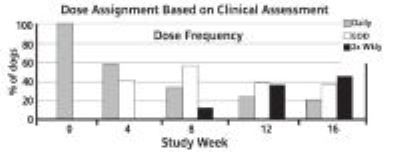
Analysis of blood levels of cyclosporine drawn during the study demonstrated no correlation between blood cyclosporine levels and CADESI scores or pruritus; therefore monitoring blood cyclosporine levels is not an appropriate predictor of effectiveness.
-
ANIMAL SAFETY:
In a 52-week oral study with dose levels of 0, 1, 3, and 9 times the target initial daily dose, emesis, diarrhea and weight loss were seen in all cyclosporine treated groups with increasing frequency as the dose increased.
Multilocular papilloma-like lesions of the skin were observed in 5 out of 8 high dose animals between weeks 20 and 40. These changes regressed spontaneously after drug was withdrawn.
Other findings in the mid and high dose animals included swollen gums due to chronic gingivitis and periodontitis, lower serum albumin and higher cholesterol, triglyceride, IgA and IgG. Hematological findings consisted of anemia and decreased leukocyte counts in a few high dose animals. Erythrocyte sedimentation rates were increased at all dose levels in a dose dependent fashion. Notable histopathological findings
were limited to lymphoid atrophy, hypertrophic gums (from gingivitis) and slight regenerative changes of the renal tubular epithelium in high dose animals. The findings were shown to be reversible during a 12-week recovery phase of the study.In a 90-day study with cyclosporine, dogs were dosed in one of two patterns: either 1, 3, or 5X the maximum recommended target initial daily dose for 90 days, or 1, 3, or 5X the maximum recommended target initial daily dose for 30 days followed by tapering to mimic the recommended clinical dosing pattern. The maximum recommended dose, when administered for 90 days causes callus-like lesions on the footpads, red/
swollen pinnae, mild to moderate gingival proliferation, hyperkeratotic areas on the integument, hair loss, salivation, vomiting, and diarrhea/ abnormal stools. These clinical signs lessened in severity or resolved as the drug was tapered to a lower dose. Increased erythrocyte sedimentation rate, hyperproteinemia, hyperglobulinemia, hypoalbuminemia, hypocalcemia, hypophosphatemia, and hypomagnesemia were observed at three and five times the maximum recommended dose. These resolved as the dose was tapered.When administered at higher than the maximum recommended dose, raised skin lesions, papilloma-like areas on the integument, popliteal lymph node enlargement, and weight loss were also seen. There were no cyclosporine related changes in urinalysis, ECG, blood pressure, or ophthalmologic exams.
Gross necropsy revealed epithelial changes consistent with those seen on physical examination. Proliferation of gingiva and toe pad epithelium was seen in all cyclosporine dosed groups, and was seen in a dose dependent fashion. The degree of the proliferation was greater in dogs in the non-tapered groups as compared to the tapered groups. Similar changes were noted on histopathologic examination of the cutaneous
changes seen on physical examination. These lesions were characterized by epidermal hyperplasia, chronic dermatitis and hyperkeratosis.Methylprednisolone combination: Twenty-four dogs were administered 1 mg/kg/day methylprednisolone alone for 14 days followed by 20 mg/kg/day cyclosporine either alone or in combination with methylprednisolone, or placebo for 14 days. There was no evidence of seizures/convulsions or neurological signs.
Vaccination effect: The effect of cyclosporine administration on the immunological response to vaccination was evaluated in a study in which 16 dogs were dosed with either cyclosporine at 20 mg/kg/day (4X the initial daily dose) or placebo for 56 days. All dogs were vaccinated on Day 27 with a killed commercial rabies virus and a multivalent vaccine (DHLPP) which included a modified live virus. Antibody titers for rabies,
canine distemper, canine adenovirus type 2, parainfluenza, parvovirus, Leptospira canicola, and Leptospira icterohaemmorrhagiae were examined on Days 0, 27 (prior to vaccination), 42 and 56. Quantification of CD4, CD8, and CD3 T-lymphocytes was analyzed.Clinical changes included soft stool and dermatologic changes consistent with those seen in previous studies. Antibody titers did not rise in dogs treated with cyclosporine or the placebo for any component of the multivalent vaccine which included a modified live virus while all animals demonstrated a significant increase in antibody rabies titer by Day 42 or 15 days post-revaccination. No effect was seen on T-lymphocytes.
- STORAGE INFORMATION:
- HOW SUPPLIED:
-
Instructions for Assembling the Dispensing System and Preparing a Dose of CYCLAVANCE™ (cyclosporine oral solution) USP MODIFIED.
Assembling the Dispensing System
The dispensing system consists of three parts:
1. A vial containing the medicine sealed with a rubber stopper
2. A plastic adapter (dispensing system) that you will push onto the top of the vial. The adapter must always remain on the vial after first use.
3. An oral dosing syringe that fits into the top of the plastic adapter to withdraw the prescribed dose of medicine from the vial.
(1 mL syringe with the 5 and 15 mL vial sizes; 1 and 3 mL syringes with the 30 and 50 mL vial sizes)Fitting the Plastic Adapter into the New Bottle of Medicine
1. Remove the plastic lid from the top of the vial.
2. Hold the vial upright on a table and align the stylet straight up and down over the center ring in the vial stopper. Push the plastic adapter firmly straight down onto the top of the vial until it is firmly and evenly seated.
Note: To prepare a dose, carefully follow the instructions for Preparing a Dose of Medicine.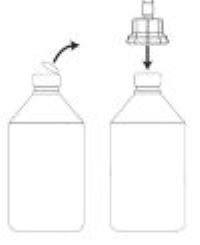
Preparing a Dose of Medicine
1. Check that the plunger of the oral dosing syringe is pushed all the way down.
2. Keep the vial upright and push the oral dosing syringe firmly into the plastic adapter while turning the syringe clockwise to secure the dispensing system.
3. Turn the vial with the attached dosing syringe upside down and slowly pull the plunger down so that the oral dosing syringe fills with the medicine.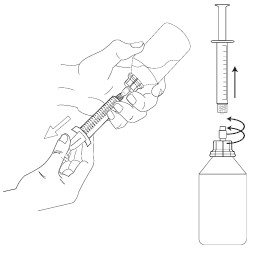
4. Expel any large bubbles by pushing and pulling the plunger a few times. The presence of a few tiny bubbles is not important for dosing accuracy.
5. Withdraw the dose of medicine prescribed by your veterinarian using the flange of the barrel to align with the marks on the plunger. These marks are in milliliters (mL).
Note: If the prescribed dose is more than the maximum volume marked on the oral dosing syringe, you will need to reload the syringe to withdraw the full dose.
6. Return the vial to its upright position and remove the oral dosing syringe by twisting it counterclockwise out of the plastic dispenser.
You can now introduce the syringe into the mouth of the dog according to your veterinarian’s instructions, and push the medicine out of the syringe.
See Information for Dog Owners for complete administration instructions. To view an instructional video on Assembling the Dispensing System and Preparing a Dose of CYCLAVANCE, please go to https://vet-us.virbac.com/cyclavance
Do not rinse or clean the oral dosing syringe between uses.
Store the medication and the dosing syringe securely.
CYCLAVANCE should be stored and dispensed in the original container at temperatures between 68-86˚F (20-30˚C).
Do not refrigerate because a precipitate may be observed below 68˚F (20˚C). Once opened, use contents within 12 weeks.
Keep out of reach of children -
SPL UNCLASSIFIED SECTION
Approved by FDA under ANADA # 200-692
Manufactured for:
Virbac AH, Inc.
P.O. Box 162059
Fort Worth, TX 76161
1-800-338-3659
us.virbac.com02026053
Rev. 08/2022© 2022 Virbac Corporation. All Rights Reserved.
CYCLAVANCE is a registered trademark of Virbac S.A.
Luer-Lok is a registered trademark of Becton, Dickinson and Company. -
Information for Dog Owners
CYCLAVANCE is indicated for the control of atopic dermatitis in dogs weighing at least 4 lbs. (1.8 kg) body weight. Dogs with atopic
dermatitis scratch, lick and chew their skin which can cause red, raised crusty bumps, open sores and/or hair loss.Atopic dermatitis is a common skin disease in dogs and is caused by allergens such as house dust mites or pollens which stimulate an
exaggerated immune response. The disease is chronic, recurrent, and requires lifelong management.This summary contains important information about CYCLAVANCE. You should read this information before starting your dog on
CYCLAVANCE. This sheet is provided only as a summary and does not take the place of instructions from your veterinarian. Talk to your
veterinarian if you do not understand any of this information or you want to know more about CYCLAVANCE.What is CYCLAVANCE?
CYCLAVANCE is an oral solution of cyclosporine that lowers the immune response.
CYCLAVANCE selectively acts on the immune cells involved in the allergic reaction.
CYCLAVANCE reduces the inflammation and itching associated with atopic dermatitis.What kind of results can I expect when my dog takes CYCLAVANCE for the control of atopic dermatitis? CYCLAVANCE should be
given daily until improvement is seen. This will generally be the case within 30 days. You should contact your veterinarian if you are
not satisfied with your dog’s response. Once the signs of atopic dermatitis are satisfactorily controlled, your veterinarian may reduce
the frequency of administration of the product. Dose adjustment should only be carried out in consultation with your veterinarian. Your
veterinarian will perform a clinical assessment at regular intervals and adjust the frequency of administration up or down according to the
clinical response obtained.What dogs should not take CYCLAVANCE?
Your dog should not be given CYCLAVANCE if s/he:
• Has a history of cancer or may possibly have cancer.
• Has a history of seizures, diabetes mellitus, and infections.
• Is hypersensitive to cyclosporine.What to discuss with your veterinarian before giving CYCLAVANCE to your dog.
Tell your veterinarian about:
• Any digestive upset (vomiting or diarrhea) your dog has had
• Any history of lack of appetite and/or weight loss your dog has had
• Any serious disease or health conditions your dog has had
• Any allergies that your dog has now or has had
• Any medications, specifically any azoles (e.g. ketoconazole) and/or steroids that you are giving your dog or plan to give your
dog, including those you can get without prescription (over the counter) and any dietary supplements
• If you plan to breed your dog, or if your dog is pregnant or nursingTalk to your veterinarian about:
• What tests might be done before CYCLAVANCE is prescribed
• The potential side effects your dog may experience while taking CYCLAVANCE
• How often your dog may need to be examined by your veterinarian
• The risks and benefits of using CYCLAVANCEWhat are the possible side effects that may occur in my dog during therapy with CYCLAVANCE® (cyclosporine oral solution)
USP MODIFIED?
CYCLAVANCE, like all other drugs, may cause some side effects in individual dogs. These are normally mild, but serious side effects
have been reported in dogs taking CYCLAVANCE. Serious side effects can, in rare situations, result in death. It is important to stop
the medication and contact your veterinarian immediately if you think your dog may have a medical problem or side effect while on
CYCLAVANCE. To report adverse effects, access medical information, or obtain additional product information call 1-800-338-3659.In clinical studies, the most commonly reported side effect for cyclosporine was vomiting and diarrhea. In most cases, the vomiting and
diarrhea stopped with continued use or dose modification. Persistent otitis externa, urinary tract infection, anorexia, lethargy, gingival
hyperplasia and lymphadenopathy were the next most frequent side effects observed. Persistent, progressive weight loss may be
associated with more serious side effects. You should monitor your dog’s appetite and body weight. If you think that your dog is losing
weight, you should contact your veterinarian. CYCLAVANCE may increase susceptibility to infection and to the development of tumors.CYCLAVANCE should only be given to dogs.
People should not take CYCLAVANCE. Keep CYCLAVANCE and all medication out of reach of children. Call your physician immediately if
you accidentally swallow CYCLAVANCE.How to give CYCLAVANCE to your dog.
CYCLAVANCE should be given according to your veterinarian’s instructions. Your veterinarian will tell you what amount of CYCLAVANCE
is right for your dog. CYCLAVANCE should be given at least one hour before or two hours after a meal. Do not change the way you
give CYCLAVANCE to your dog without first speaking with your veterinarian. If a dose is missed, the next dose should be administered
(without doubling) as soon as possible, but dosing should be no more frequent than once daily. Do not rinse or clean the oral dosing
syringe between uses.Advice on Correct Administration.
See Instructions for Assembling the Dispensing System and Preparing a Dose of CYCLAVANCE. If your veterinarian has already
assembled the dispensing system, skip the instructions for assembling the dispensing system and follow the instructions for preparing a
dose of medicine.How to Store CYCLAVANCE.
CYCLAVANCE should be stored and dispensed in the original container at temperatures between 68-86°F (20-30°C). Do not refrigerate
because a precipitate may be observed below 68°F (20°C). Once opened, use contents within 12 weeks.Special precautions to be taken when administering CYCLAVANCE.
Wear gloves during administration. Do not eat, drink, smoke, or use smokeless tobacco while handling CYCLAVANCE. Wash hands
after administration. In case of accidental ingestion, seek medical advice immediately and show the package insert or the label to the
physician.People with known hypersensitivity to cyclosporine should avoid contact with CYCLAVANCE.
Can CYCLAVANCE be given with other medications?
CYCLAVANCE should not be given with other drugs that may lower the immune response. Cyclosporine has been safely used in
conjunction with other common medications. However, interactions with certain medications are possible. Therefore, always tell your
veterinarian about all medications that you have given your dog in the past and all medications that you are planning to give with
CYCLAVANCE.What can I do in case my dog gets more than the prescribed amount of CYCLAVANCE?
Contact your veterinarian immediately if your dog gets more than the prescribed amount of CYCLAVANCE.What else should I know about CYCLAVANCE?
If your dog becomes seriously ill, consult your veterinarian who will recommend the appropriate treatment.This sheet provides a summary of information about CYCLAVANCE. If you have any questions or concerns about CYCLAVANCE or atopic
dermatitis in dogs, talk to your veterinarian. As with all prescribed medications, CYCLAVANCE should only be given to the dog for which it
was prescribed. It should be given to your dog only for the condition for which it was prescribed, at the prescribed dose, and as directed
by your veterinarian.Approved by FDA under ANADA # 200-692
- Principal Display 15 mL
- Principal Display 50 mL
-
INGREDIENTS AND APPEARANCE
CYCLAVANCE
cyclosporine oral solution solutionProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC:51311-994 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength cyclosporine (UNII: 83HN0GTJ6D) (CYCLOSPORINE - UNII:83HN0GTJ6D) cyclosporine 100 mg in 1 mL Product Characteristics Color white (colorless to yellow solution) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:51311-994-15 15 mL in 1 VIAL, MULTI-DOSE 2 NDC:51311-994-50 50 mL in 1 VIAL, MULTI-DOSE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANADA ANADA200692 12/18/2020 Labeler - Virbac AH Inc (131568396)