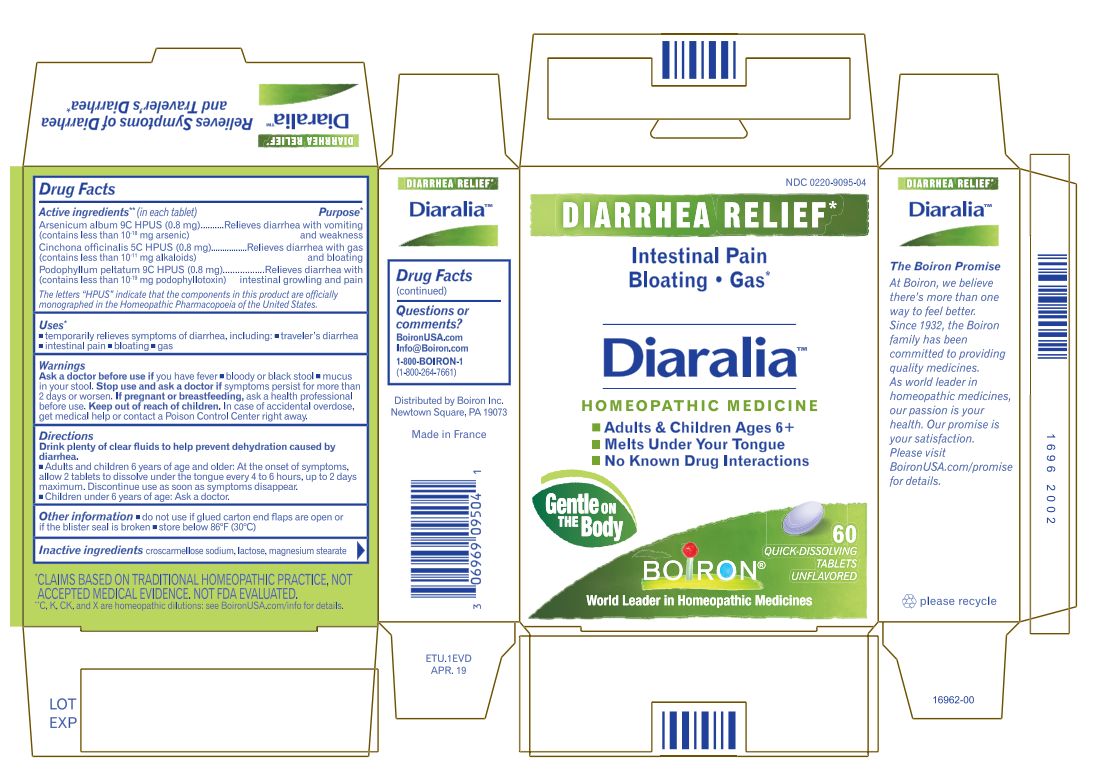
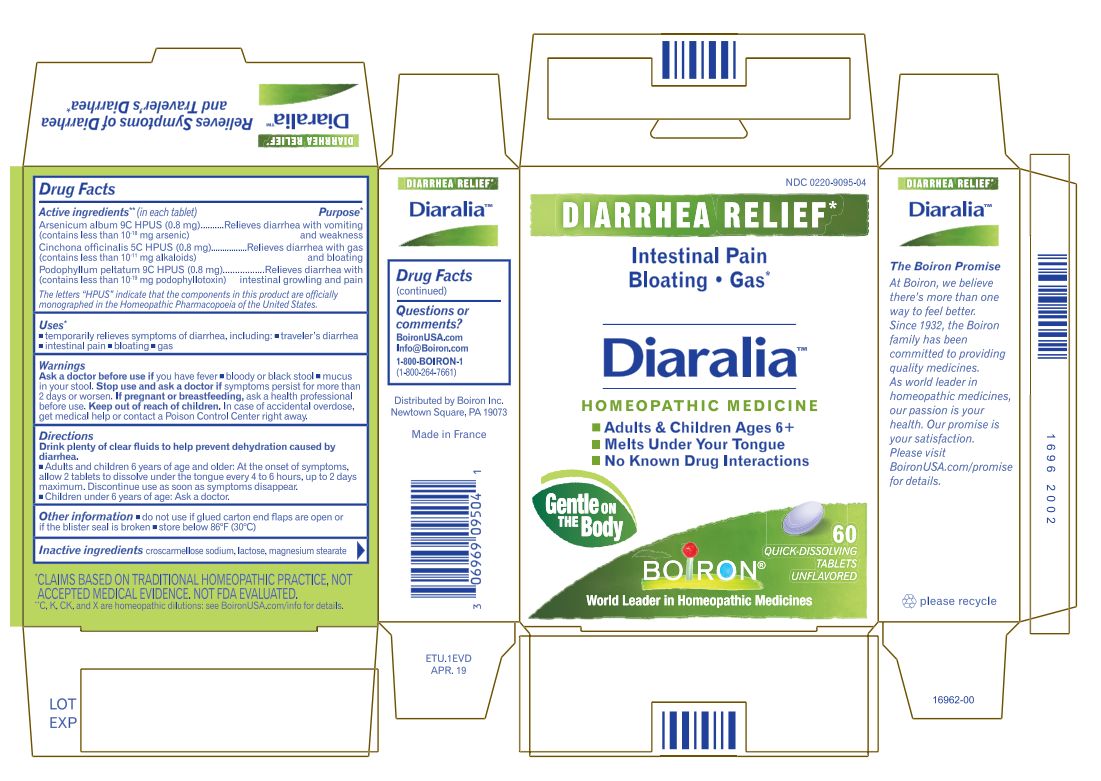
Label: DIARALIA- arsenic trioxide,cinchona officinalis bark, podophyllum tablet
- NDC Code(s): 0220-9095-04
- Packager: BOIRON
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated May 10, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- INDICATIONS & USAGE
-
PURPOSE
Purpose*
Arsenicum album 9C HPUS (0.83 mg) ... Relieves diarrhea with nausea, vomiting, and weakness. (contains less that 10 -18 mg arsenic)
Cinchona officinalis 5C HPUS (0.83 mg) ... Relieves diarrhea with gas and bloating
Podophyllum peltatum 9C HPUS (0.83 mg) ... Relieves diarrhea with intestinal rumbling and pain
- WARNINGS
- ASK DOCTOR
- STOP USE
- PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
-
DOSAGE & ADMINISTRATION
Drink plenty of clear fluids to help prevent dehydration caused by diarrhea.
- Adults and children 6 years of age and older: At the onset of symptoms, dissolve 2 tablets in the mouth every 4 to 6 hours, up to 2 days maximum. Discontinue use as soon as symptoms disappear.
- Children under 6 years of age: Ask a doctor.
-
SPL UNCLASSIFIED SECTION
do not use if glued carton end flaps are open or if the blister seal is broken.
store below 86° F (30° C)
60 Meltaway Tablets
No Known Drug Interactions
Diarrhea Relief*
Intestinal Pain Nausea Traveler's Diarrhea*
*CLAIMS BASED ON TRADITIONAL HOMEOPATHIC PRACTICE NOT ACCEPTED MEDICAL EVIDENCE. NOT FDA EVALUATED.
*C,K,CK, and X are homeopathic dilutions: see BoironUSA.com/info for details. - INACTIVE INGREDIENT
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
DIARALIA
arsenic trioxide,cinchona officinalis bark, podophyllum tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0220-9095 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PODOPHYLLUM (UNII: 2S713A4VP3) (PODOPHYLLUM - UNII:2S713A4VP3) PODOPHYLLUM 9 [hp_C] CINCHONA OFFICINALIS BARK (UNII: S003A158SB) (CINCHONA OFFICINALIS BARK - UNII:S003A158SB) CINCHONA OFFICINALIS BARK 5 [hp_C] ARSENIC TRIOXIDE (UNII: S7V92P67HO) (ARSENIC CATION (3+) - UNII:C96613F5AV) ARSENIC TRIOXIDE 9 [hp_C] Inactive Ingredients Ingredient Name Strength CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) MAGNESIUM STEARATE (UNII: 70097M6I30) LACTOSE, UNSPECIFIED FORM (UNII: J2B2A4N98G) Product Characteristics Color white Score no score Shape ROUND Size 9mm Flavor Imprint Code Boiron Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0220-9095-04 60 in 1 CARTON 08/01/2019 1 60 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 08/01/2019 Labeler - BOIRON (282560473) Registrant - BOIRON, INC. (014892269) Establishment Name Address ID/FEI Business Operations BOIRON 282560473 manufacture(0220-9095)