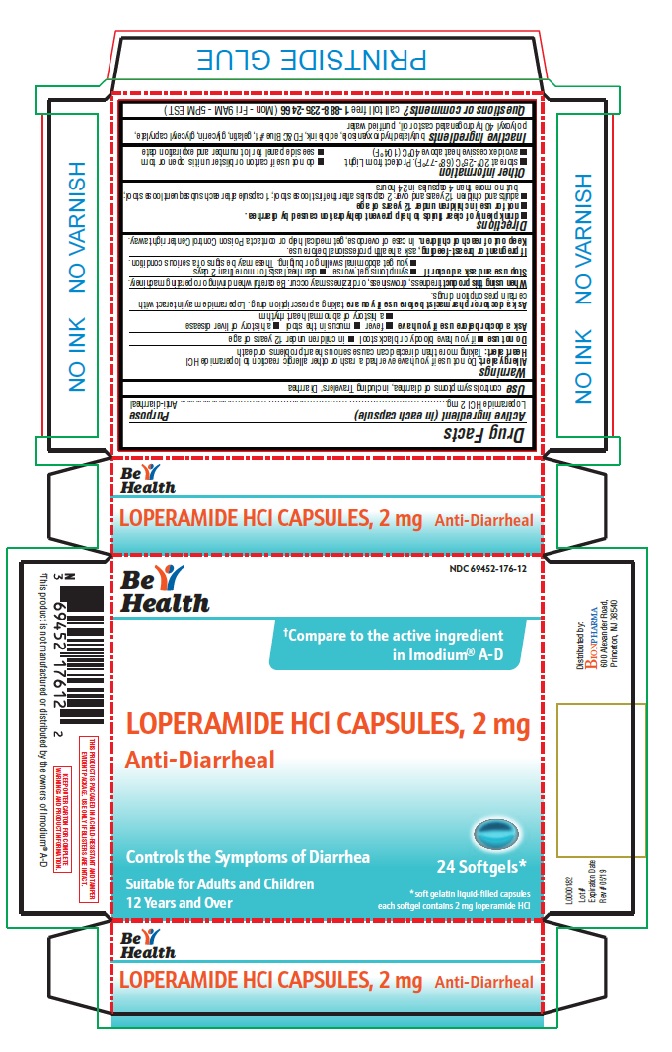
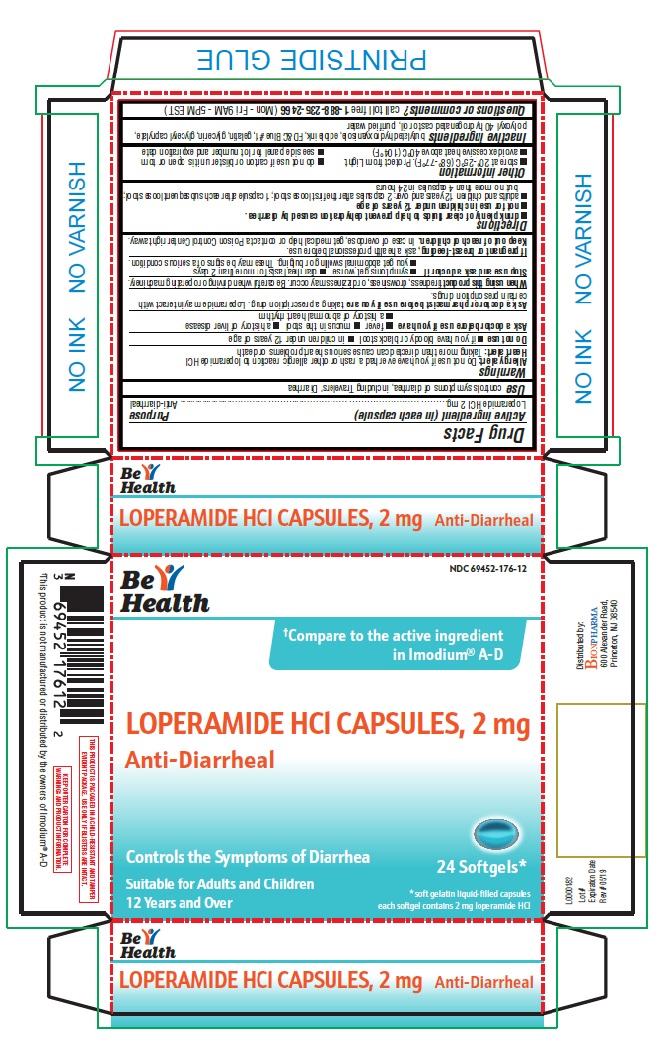
Label: ANTI-DIARRHEAL- loperamide hydrochloride capsule, liquid filled
- NDC Code(s): 69452-176-12
- Packager: Bionpharma Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated September 9, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each capsule)
- Purpose
- Use
- Warnings
- Do not use
- Ask a doctor before use if you have
- Ask a doctor or pharmacist before use if you are
- When using this product
- Stop use and ask a doctor if
- If pregnant or breast-feeding,
- Keep out of reach of children.
- Directions
- Other information
- Inactive ingredients
-
Questions or comments?
call toll free 1-888-235-2466 (Mon - Fri 9AM - 5PM EST)
†This product is not manufactured or distributed by the owners of Imodium® A-D
THIS PRODUCT IS PACKAGED IN A CHILD-RESISTANT AND TAMPER EVIDENT PACKAGE. USE ONLY IF BLISTERS ARE INTACT.
KEEP OUTER CARTON FOR COMPLETE WARNINGS AND PRODUCT INFORMATION.
Distributed by:
BIONPHARMA
600 Alexander Road,
Princeton, NJ 08540L0000182
Rev # 05/19
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
ANTI-DIARRHEAL
loperamide hydrochloride capsule, liquid filledProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69452-176 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LOPERAMIDE HYDROCHLORIDE (UNII: 77TI35393C) (LOPERAMIDE - UNII:6X9OC3H4II) LOPERAMIDE HYDROCHLORIDE 2 mg Inactive Ingredients Ingredient Name Strength BUTYLATED HYDROXYANISOLE (UNII: REK4960K2U) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) GELATIN (UNII: 2G86QN327L) GLYCERIN (UNII: PDC6A3C0OX) GLYCERYL CAPRYLATE (UNII: TM2TZD4G4A) POLYOXYL 40 HYDROGENATED CASTOR OIL (UNII: 7YC686GQ8F) WATER (UNII: 059QF0KO0R) Product Characteristics Color BLUE Score no score Shape CAPSULE Size 10mm Flavor Imprint Code LP2 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69452-176-12 1 in 1 CARTON 08/02/2019 1 24 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021855 08/02/2019 Labeler - Bionpharma Inc. (079637826) Registrant - Bionpharma Inc. (079637826) Establishment Name Address ID/FEI Business Operations Patheon Softgels Inc. 002193829 manufacture(69452-176)