Label: BACZOL EXPECTORANT- guaifenesin, phenylephrine hcl syrup
- NDC Code(s): 76864-209-02
- Packager: Procaps S.A. de C.V.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated August 28, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
-
WARNINGS
Warnings: • Do not exceed recommended dosage • If nervousness,
dizziness, or sleeplessness occurs, discontinue use and consult a
doctor.Do not use this product • for persistant or chronic cough, such as
occurs with smoking, asthma, chronic bronchitis, or emphysema, or if
cough is accompanied by excessive phlegm (mucus), unless directed
by a Doctor • if you are now taking a prescription Monoamineoxidase
Inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional
conditions, or Parkinson's disease), or for 2 weeks after stopping the
MAOI drug. If you do not know if you are taking a prescription drug that
contains an MAOI, ask a doctor or a pharmacist before taking this product.
• if you have heart disease, high blood pressure, thyroid disease, diabetes,
or difficulty in urination due to enlargement of the prostate gland unless
directed by a Doctor.Stop use and ask a Doctor • A persistent cough may be a sign of a
serious condition; • If cough persists for more than one week, tend to
recur, or is accompanied by fever, rash or persistent headache; • if
symptoms do not improve within 7 days or are accompanied by fever. - PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
- DOSAGE & ADMINISTRATION
- OTHER SAFETY INFORMATION
- INACTIVE INGREDIENT
- QUESTIONS
-
Product Labeling
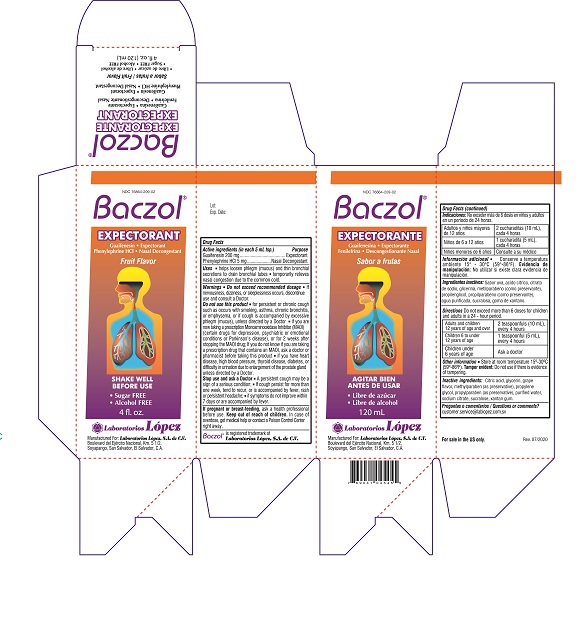
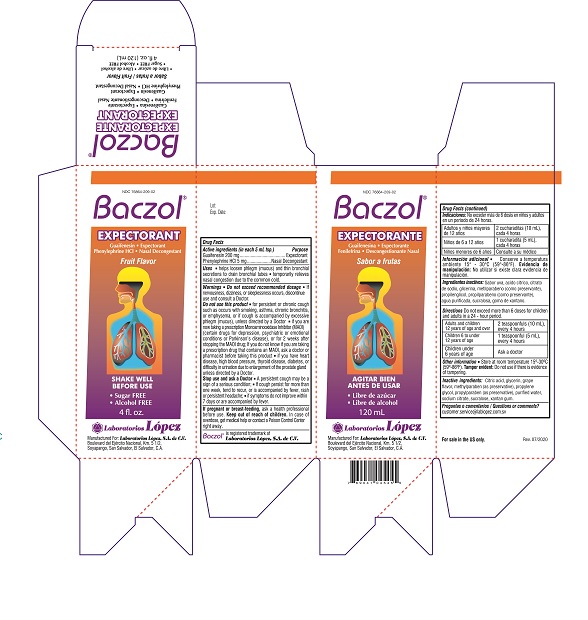
NDC 76864-209-02
Baczol ®
EXPECTORANTGuaifenesin • Expectorant
Phenylephrine HCl • Nasal DecongestantFruit Flavor
• Sugar FREE
• Alcohol FREE
4 fl. oz.
Manufactured For: Laboratorios López, S.A. de C.V.
Boulevard del Ejército Nacional, Km. 5 1/2,
Soyapango, San Salvador,, El Salvador, C.A.Lot: Rev. 07/2020
Exp. Date:
Carton
Bottle

res
-
INGREDIENTS AND APPEARANCE
BACZOL EXPECTORANT
guaifenesin, phenylephrine hcl syrupProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:76864-209 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GUAIFENESIN (UNII: 495W7451VQ) (GUAIFENESIN - UNII:495W7451VQ) GUAIFENESIN 200 mg in 5 mL PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE 5 mg in 5 mL Inactive Ingredients Ingredient Name Strength CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) GLYCERIN (UNII: PDC6A3C0OX) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) PROPYLPARABEN (UNII: Z8IX2SC1OH) WATER (UNII: 059QF0KO0R) SODIUM CITRATE (UNII: 1Q73Q2JULR) SUCRALOSE (UNII: 96K6UQ3ZD4) XANTHAN GUM (UNII: TTV12P4NEE) Product Characteristics Color Score Shape Size Flavor GRAPE Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:76864-209-02 1 in 1 CARTON 06/03/2021 1 120 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 06/03/2021 Labeler - Procaps S.A. de C.V. (851259341)