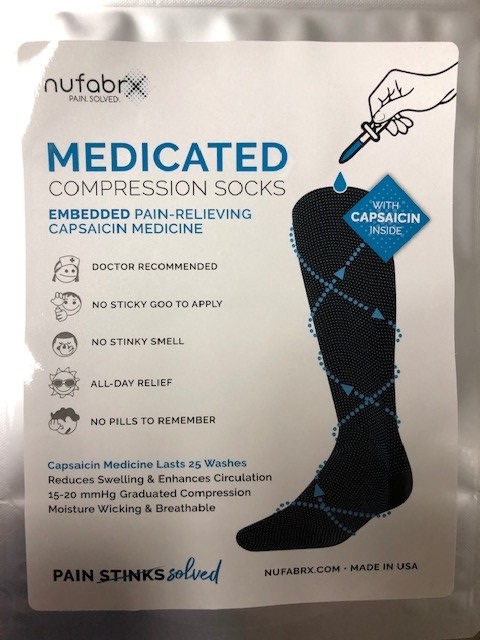
Label: MEDICATED COMPRESSION SOCK- capsaicin cloth
-
Contains inactivated NDC Code(s)
NDC Code(s): 72490-006-01, 72490-006-02, 72490-006-03 - Packager: Textile-Based Delivery, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated December 17, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Uses
-
Warnings
For external use only
Do not use
- Allergy Alert: if you are allergic to any ingredients of this product or chili peppers contact a doctor before use
- On wounds or damaged skin
- With, or at same time as, other external analgesic products
- With a heating pad
When using this product
- You may experience a burning sensation which is normal and related to the way the product works. With regular use, this sensation generally diminishes.
- Do not use immediately before or after activities such as swimming, showering, or sunbathing.
- Use only as directed.
- Avoid contact with eyes and mucous membranes or rashes.
- Wash hands after putting product on body and before touching your face or eyes.
- Directions
- Other Information
- Inactive Ingredients
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
MEDICATED COMPRESSION SOCK
capsaicin clothProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72490-006 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CAPSAICIN (UNII: S07O44R1ZM) (CAPSAICIN - UNII:S07O44R1ZM) CAPSAICIN 0.13 Inactive Ingredients Ingredient Name Strength BUTYL ACRYLATE/METHYL METHACRYLATE/METHACRYLIC ACID COPOLYMER (18000 MW) (UNII: JZ1374NL9E) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72490-006-01 2 in 1 POUCH; Type 0: Not a Combination Product 07/16/2019 2 NDC:72490-006-02 2 in 1 POUCH; Type 0: Not a Combination Product 07/16/2019 3 NDC:72490-006-03 2 in 1 POUCH; Type 0: Not a Combination Product 07/16/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 07/16/2019 Labeler - Textile-Based Delivery, Inc. (079309716)