Label: NYSTATIN AND TRIAMCINOLONE ACETONIDE cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 71589-012-15, 71589-012-30, 71589-012-60 - Packager: Aleor Dermaceuticals Limited
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated December 1, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
Nystatin and triamcinolone acetonide cream, USP for dermatologic use contain the antifungal agent nystatin and the synthetic corticosteroid triamcinolone acetonide.
Nystatin is a polyene antimycotic obtained from Streptomyces noursei. It is a yellow to light tan powder with a cereal-like odor, practically insoluble or insoluble in water and in alcohol. Structural formula:
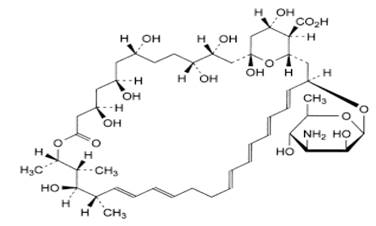
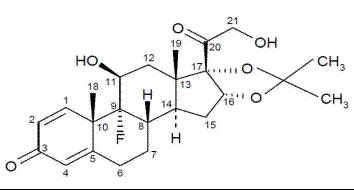
Triamcinolone acetonide is designated chemically as pregna-1,4-diene-3,20-dione, 9-fluoro-11,21-dihydroxy-16,17- [(1-methylethylidene)bis(oxy)]-, (11β,16α)-9-fluoro-11b,16a,17,21-tetrahydroxypregna-1,4-diene-3,20-dione cyclic 16,17- acetal with acetone.
The white to cream crystalline powder has a slight odor, is practically insoluble in water, and very soluble in alcohol. Structural formula:
Nystatin and triamcinolone acetonide cream, USP is a soft, smooth cream having a light yellow to buff color. Each gram contains 100,000 USP nystatin units and 1 mg triamcinolone acetonide USP in a cream base of polysorbate-60, aluminum hydroxide gel, titanium dioxide, mono-and di-glycerides, polyoxyl 8 stearate, simethicone emulsion, sorbic acid, propylene glycol, white petrolatum, polyoxyl 20- cetostearyl ether, methylparaben, propylparaben, noncrystallizing sorbitol solution and purified water.
-
CLINICAL PHARMACOLOGY
Nystatin exerts its antifungal activity against a variety of pathogenic and nonpathogenic yeasts and fungi by binding to sterols in the cell membrane. The binding process renders the cell membrane incapable of functioning as a selective barrier. Nystatin provides specific anticandidal activity to Candida (Monilia) albicans and other Candida species, but is not active against bacteria, protozoa, trichomonads, or viruses.
Nystatin is not absorbed from intact skin or mucous membranes.
Triamcinolone Acetonide
Triamcinolone acetonide is primarily effective because of its anti-inflammatory, antipruritic and vasoconstrictive actions, characteristic of the topical corticosteroid class of drugs. The pharmacologic effects of the topical corticosteroids are well known; however, the mechanisms of their dermatologic actions are unclear. Various laboratory methods, including vasoconstrictor assays, are used to compare and predict potencies and/or clinical efficacies of the topical corticosteroids. There is some evidence to suggest that a recognizable correlation exists between vasoconstrictor potency and therapeutic efficacy in man.
Pharmacokinetics
The extent of percutaneous absorption of topical corticosteroids is determined by many factors including the vehicle, the integrity of the epidermal barrier, and the use of occlusive dressings (see DOSAGE AND ADMINISTRATION).
Topical corticosteroids can be absorbed from normal intact skin. Inflammation and/or other disease processes in the skin increase percutaneous absorption. Occlusive dressings substantially increase the percutaneous absorption of topical corticosteroids (see DOSAGE AND ADMINISTRATION).
Once absorbed through the skin, topical corticosteroids are handled through pharmacokinetic pathways similar to systemically administered corticosteroids. Corticosteroids are bound to plasma proteins in varying degrees. Corticosteroids are metabolized primarily in the liver and are then excreted by the kidneys. Some of the topical corticosteroids and their metabolites are also excreted into the bile.
Nystatin and Triamcinolone Acetonide
During clinical studies of mild to severe manifestations of cutaneous candidiasis, patients treated with nystatin and triamcinolone acetonide showed a faster and more pronounced clearing of erythema and pruritus than patients treated with nystatin or triamcinolone acetonide alone.
- INDICATIONS AND USAGE
- CONTRAINDICATIONS
-
PRECAUTIONS
Systemic absorption of topical corticosteroids has produced reversible hypothalamic-pituitary-adrenal (HPA) axis suppression, manifestations of Cushing's syndrome, hyperglycemia, and glucosuria in some patients. Conditions that augment systemic absorption include application of the more potent steroids, use over large surface areas, prolonged use, and the addition of occlusive dressings (see DOSAGE AND ADMINISTRATION).
Therefore, patients receiving a large dose of any potent topical steroid applied to a large surface area should be evaluated periodically for evidence of HPA axis suppression by using the urinary free cortisol and ACTH stimulation tests, and for impairment of internal homeostasis. If HPA axis suppression or elevation of the body temperature occurs, an attempt should be made to withdraw the drug, to reduce the frequency of application, or substitute a less potent steroid.
Recovery of HPA axis function and thermal homeostasis are generally prompt and complete upon discontinuation of the drug. Infrequently, signs and symptoms of steroid withdrawal may occur, requiring supplemental systemic corticosteroids.
Children may absorb proportionally larger amounts of topical corticosteroids and thus be more susceptible to systemic toxicity (see PRECAUTIONS, Pediatric Use).
If irritation or hypersensitivity develops with the combination nystatin and triamcinolone acetonide, treatment should be discontinued and appropriate therapy instituted.
Information for the Patient
Patients using this medication should receive the following information and instructions:
- This medication is to be used as directed by the physician. It is for dermatological use only. Avoid contact with the eyes.
- Patients should be advised not to use this medication for any disorder other than for which it was prescribed.
- The treated skin area should not be bandaged or otherwise covered or wrapped as to be occluded (see DOSAGE AND ADMINISTRATION).
- Patients should report any signs of local adverse reactions.
- When using this medication in the inguinal area, patients should be advised to apply the cream sparingly and to wear loose fitting clothing.
- Parents of pediatric patients should be advised not to use tight-fitting diapers or plastic pants on a child being treated in the diaper area, as these garments may constitute occlusive dressings.
- Patients should be advised on preventive measures to avoid reinfection.
If there is a lack of therapeutic response, appropriate microbiological studies (e.g. KOH smears and/or cultures) should be repeated to confirm the diagnosis and rule out other pathogens, before instituting another course of therapy.
A urinary free cortisol test and ACTH stimulation test may be helpful in evaluating hypothalamic-pituitary-adrenal (HPA) axis suppression due to corticosteroids.
Carcinogenesis, Mutagenesis, and Impairment of Fertility
Long-term animal studies have not been performed to evaluate carcinogenic or mutagenic potential, or possible impairment of fertility in males or females.
Pregnancy
There are no teratogenic studies with combined nystatin and triamcinolone acetonide. Corticosteroids are generally teratogenic in laboratory animals when administered systemically at relatively low dosage levels. The more potent corticosteroids have been shown to be teratogenic after dermal application in laboratory animals. Therefore, any topical corticosteroid preparation should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Topical preparations containing corticosteroids should not be used extensively on pregnant patients, in large amounts, or for prolonged periods of time.
Nursing Mothers
It is not known whether any component of this preparation is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised during the use of this preparation by a nursing woman.
Pediatric Use
In clinical studies of a limited number of pediatric patients ranging from two months through 12 years, nystatin and triamcinolone acetonide cream formulation cleared or significantly ameliorated the disease state in most patients.
Pediatric patients may demonstrate greater susceptibility to topical corticosteroid-induced hypothalamic-pituitary-adrenal (HPA) axis suppression and Cushing's syndrome than mature patients because of a larger skin surface area to body weight ratio.
HPA axis suppression, Cushing's syndrome, and intracranial hypertension have been reported in children receiving topical corticosteroids. Manifestations of adrenal suppression in children include linear growth retardation, delayed weight gain, low plasma cortisol levels, and absence of response to ACTH stimulation. Manifestations of intracranial hypertension include bulging fontanelles, headaches, and bilateral papilledema.
Administration of topical corticosteroids to children should be limited to the least amount compatible with an effective therapeutic regimen. Chronic corticosteroid therapy may interfere with the growth and development of children.
-
ADVERSE REACTIONS
A single case (approximately one percent of patients studied) of acneiform eruption occurred with use of combined nystatin and triamcinolone acetonide in clinical studies.
Nystatin is virtually nontoxic and nonsensitizing and is well tolerated by all age groups, even during prolonged use. Rarely, irritation may occur.
The following local adverse reactions are reported infrequently with topical corticosteroids (reactions are listed in an approximate decreasing order of occurrence): burning, itching, irritation, dryness, folliculitis, hypertrichosis, acneiform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis, maceration of the skin, perioral secondary infection, skin atrophy, striae and miliaria.
To report SUSPECTED ADVERSE REACTIONS, contact FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
-
OVERDOSAGE
Topically applied corticosteroids can be absorbed in sufficient amounts to produce systemic effects (see PRECAUTIONS, General); however, acute overdosage and serious adverse effects with dermatologic use are unlikely.
-
DOSAGE AND ADMINISTRATION
Nystatin and triamcinolone acetonide cream is usually applied to the affected areas twice daily in the morning and evening by gently and thoroughly massaging the preparation into the skin. The cream should be discontinued if symptoms persist after 25 days of therapy (see PRECAUTIONS, Laboratory Tests).
Nystatin and triamcinolone acetonide cream should not be used with occlusive dressings.
-
HOW SUPPLIED
Nystatin and triamcinolone acetonide cream, USP is light yellow to buff color cream supplied as follows:
15 g tube (NDC 71589-012-15)
30 g tube (NDC 71589-012-30)
60 g tube (NDC 71589-012-60)
STORAGE
Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Avoid freezing.
Manufactured for:
Aleor Dermaceuticals Ltd.,
Karakhadi, Vadodara 391450, India.
Manufactured by:
Aleor Dermaceuticals Ltd.,
Karakhadi, Vadodara 391450, India.
Mfg. License. No.: G/25/2216
Revised: 12/2021
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
NYSTATIN AND TRIAMCINOLONE ACETONIDE
nystatin and triamcinolone acetonide creamProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:71589-012 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength NYSTATIN (UNII: BDF1O1C72E) (NYSTATIN - UNII:BDF1O1C72E) NYSTATIN 100000 [USP'U] in 1 g TRIAMCINOLONE ACETONIDE (UNII: F446C597KA) (TRIAMCINOLONE ACETONIDE - UNII:F446C597KA) TRIAMCINOLONE ACETONIDE 1 mg in 1 g Inactive Ingredients Ingredient Name Strength POLYSORBATE 60 (UNII: CAL22UVI4M) ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) GLYCERYL MONO AND DIPALMITOSTEARATE (UNII: KC98RO82HJ) PEG-8 STEARATE (UNII: 2P9L47VI5E) SORBIC ACID (UNII: X045WJ989B) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) PETROLATUM (UNII: 4T6H12BN9U) POLYOXYL 20 CETOSTEARYL ETHER (UNII: YRC528SWUY) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLPARABEN (UNII: Z8IX2SC1OH) SORBITOL (UNII: 506T60A25R) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71589-012-15 1 in 1 CARTON 04/08/2021 1 15 g in 1 TUBE; Type 0: Not a Combination Product 2 NDC:71589-012-30 1 in 1 CARTON 04/08/2021 2 30 g in 1 TUBE; Type 0: Not a Combination Product 3 NDC:71589-012-60 1 in 1 CARTON 04/08/2021 3 60 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA214090 04/08/2021 Labeler - Aleor Dermaceuticals Limited (871411532) Registrant - Aleor Dermaceuticals Limited (871411532) Establishment Name Address ID/FEI Business Operations Aleor Dermaceuticals Limited 871411532 MANUFACTURE(71589-012) , ANALYSIS(71589-012)




