Label: THERAFLU SEVERE COLD RELIEF CHEST CONGESTION DAYTIME- acetaminophen, dextromethorphan hbr, guaifenesin syrup
- NDC Code(s): 0067-0104-08
- Packager: Haleon US Holdings LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated April 10, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients (in each 30 mL)
- Purposes
-
Uses
- temporarily relieves these symptoms due to a cold:
- minor aches and pains
- minor sore throat pain
- headache
- cough due to minor throat and bronchial irritation
- temporarily reduces fever
- helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive
- temporarily relieves these symptoms due to a cold:
-
Warnings
Liver warning:This product contains acetaminophen. Severe liver damage may occur if you take
- more than 4,000 mg of acetaminophen in 24 hours
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Allergy alert:Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Sore throat warning:If sore throat is severe, persists for more than 2 days, is accompanied or followed by a fever, headache, rash, nausea, or vomiting consult a doctor promptly.
Do not use
- in a child under 12 years of age
- if you are allergic to acetaminophen
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- liver disease
- cough that occurs with too much phlegm (mucus)
- a breathing problem such as emphysema or chronic bronchitis
- cough that lasts or is chronic such as occurs with smoking, asthma, chronic bronchitis or emphysema
-
Directions
- do not use more than directed
- measure the dose correctly using the enclosed dosing cup
- take every 4 hours in dosing cup provided, while symptoms persist
- do not take more than 5 doses (150 mL) in 24 hours unless directed by a doctor
Age
Dose
Adults and children 12 years of age and over
30 mL
Children under 12 years of age
do not use
- Other information
- Inactive ingredients
- Questions or comments?
-
Generic Section
Distributed by: Haleon, Warren, NJ 07059. Made in Canada
Trademarks are owned by or licensed to the Haleon group of companies.
©2023 Haleon group of companies or its licensor. Pat. Info www.productpats.com62000000207727
PEEL BACK HERE
DO NOT USE IF NECKBAND PRINTED WITH “SEALED FOR SAFETY” IS TORN OR MISSING.
-
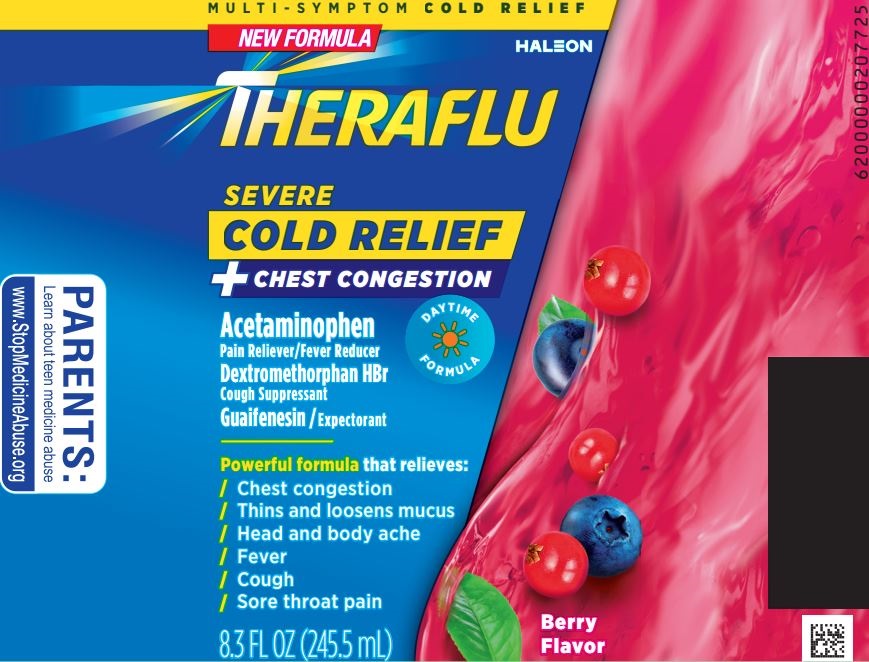
Package/Label Principal Display Panel
MULTI-SYMPTOM COLD RELIEF
NEW FORMULA
THERAFLU
SEVERE
COLD RELIEF
+ CHEST CONFESTION
Acetaminophen
Pain reliever/Fever Reducer
Dextromethorphan HBr
Cough Suppressant
Guaifenesin/ Expectorant
DAYTIME FORMULA
Powerful formula that relieves:
/ Chest congestion
/ Thins and loosens mucus
/ Head and body ache
/ Fever
/ Cough
/ Sore throat pain
8.3 FL OZ (245.5 mL)
Berry Flavor
PARENTS:
Learn about teen medicine abuse
62000000207725
-
INGREDIENTS AND APPEARANCE
THERAFLU SEVERE COLD RELIEF CHEST CONGESTION DAYTIME
acetaminophen, dextromethorphan hbr, guaifenesin syrupProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0067-0104 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 650 mg in 30 mL DEXTROMETHORPHAN HYDROBROMIDE (UNII: 9D2RTI9KYH) (DEXTROMETHORPHAN - UNII:7355X3ROTS) DEXTROMETHORPHAN HYDROBROMIDE 20 mg in 30 mL GUAIFENESIN (UNII: 495W7451VQ) (GUAIFENESIN - UNII:495W7451VQ) GUAIFENESIN 400 mg in 30 mL Inactive Ingredients Ingredient Name Strength ACESULFAME POTASSIUM (UNII: 23OV73Q5G9) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) EDETATE DISODIUM (UNII: 7FLD91C86K) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C RED NO. 40 (UNII: WZB9127XOA) GLYCERIN (UNII: PDC6A3C0OX) MALTITOL (UNII: D65DG142WK) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) SODIUM BENZOATE (UNII: OJ245FE5EU) SODIUM CITRATE, UNSPECIFIED FORM (UNII: 1Q73Q2JULR) Product Characteristics Color red Score Shape Size Flavor BERRY Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0067-0104-08 245.5 mL in 1 BOTTLE; Type 0: Not a Combination Product 12/01/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M012 12/01/2023 Labeler - Haleon US Holdings LLC (079944263)