Label: TOPCARE COLD SORE TREATMENT- camphor, menthol, phenol, white petrolatum gel
- NDC Code(s): 36800-418-01
- Packager: Topco Associates LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated April 1, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
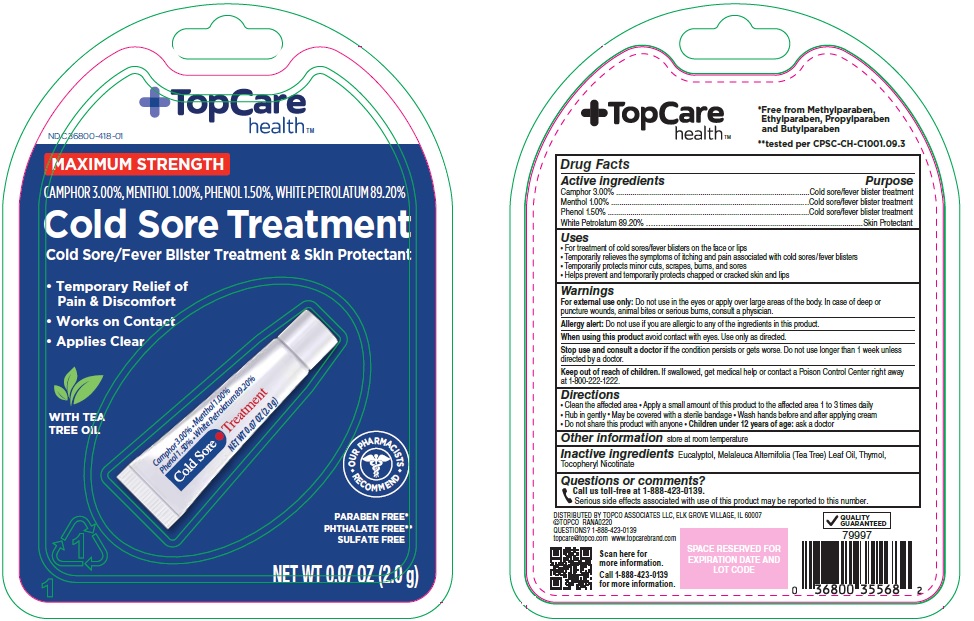
- Active ingredients
- Purpose
- Uses
-
Warnings
For external use only: Do not use in the eyes or apply over large areas of the body. In case of deep or puncture wounds, animal bites or serious burns, consult a physician.
Allergy alert: Do not use if you are allergic to any of the ingredients in this product.
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
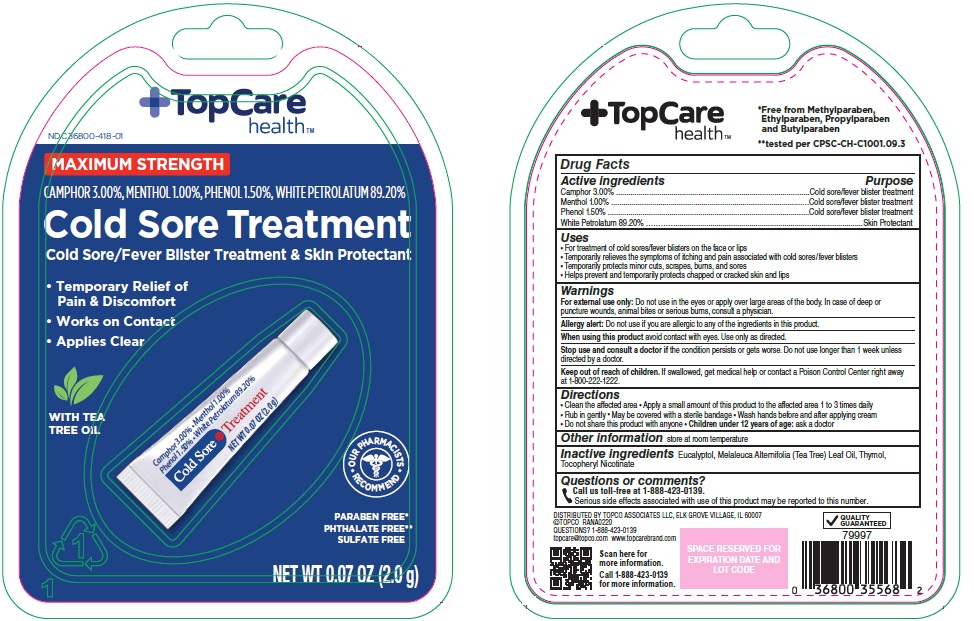
Package/Label Principal Display Panel
MAXIMUM STRENGTH
CAMPHOR 3.00%, MENTHOL 1.00%, PHENOL 1.50%, WHITE PETROLATUM 89.20%
Cold Sore Treatment
Cold Sore/Fever Blister Treatment & Skin Protectant
Temporary Relief of Pain & Discomfort
Works on Contact
Applies Clear
WITH TEA TREE OIL
OUR PHARMACISTS RECOMMEND
PARABEN FREE
PHTHALATE FREE
SULFATE FREE
NET WT 0.07 FL OZ (2.0 g)
-
INGREDIENTS AND APPEARANCE
TOPCARE COLD SORE TREATMENT
camphor, menthol, phenol, white petrolatum gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:36800-418 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CAMPHOR (SYNTHETIC) (UNII: 5TJD82A1ET) (CAMPHOR (SYNTHETIC) - UNII:5TJD82A1ET) CAMPHOR (SYNTHETIC) 30 mg in 1 g MENTHOL, UNSPECIFIED FORM (UNII: L7T10EIP3A) (MENTHOL, UNSPECIFIED FORM - UNII:L7T10EIP3A) MENTHOL, UNSPECIFIED FORM 10 mg in 1 g PHENOL (UNII: 339NCG44TV) (PHENOL - UNII:339NCG44TV) PHENOL 15 mg in 1 g PETROLATUM (UNII: 4T6H12BN9U) (PETROLATUM - UNII:4T6H12BN9U) PETROLATUM 892 mg in 1 g Inactive Ingredients Ingredient Name Strength EUCALYPTOL (UNII: RV6J6604TK) MELALEUCA ALTERNIFOLIA LEAF (UNII: G43C57162K) THYMOL (UNII: 3J50XA376E) TOCOPHERYL NICOTINATE, D-.ALPHA. (UNII: WI1J5UCY5C) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:36800-418-01 1 in 1 PACKAGE 03/09/2020 1 2 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 03/09/2020 Labeler - Topco Associates LLC (006935977)