Label: 4363 FIRST AID KIT- 4363 first aid kit
-
NDC Code(s):
0498-0100-02,
0498-0143-04,
0498-0733-00,
0498-0750-35, view more0498-4363-01
- Packager: Honeywell Safety Products USA, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated January 22, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
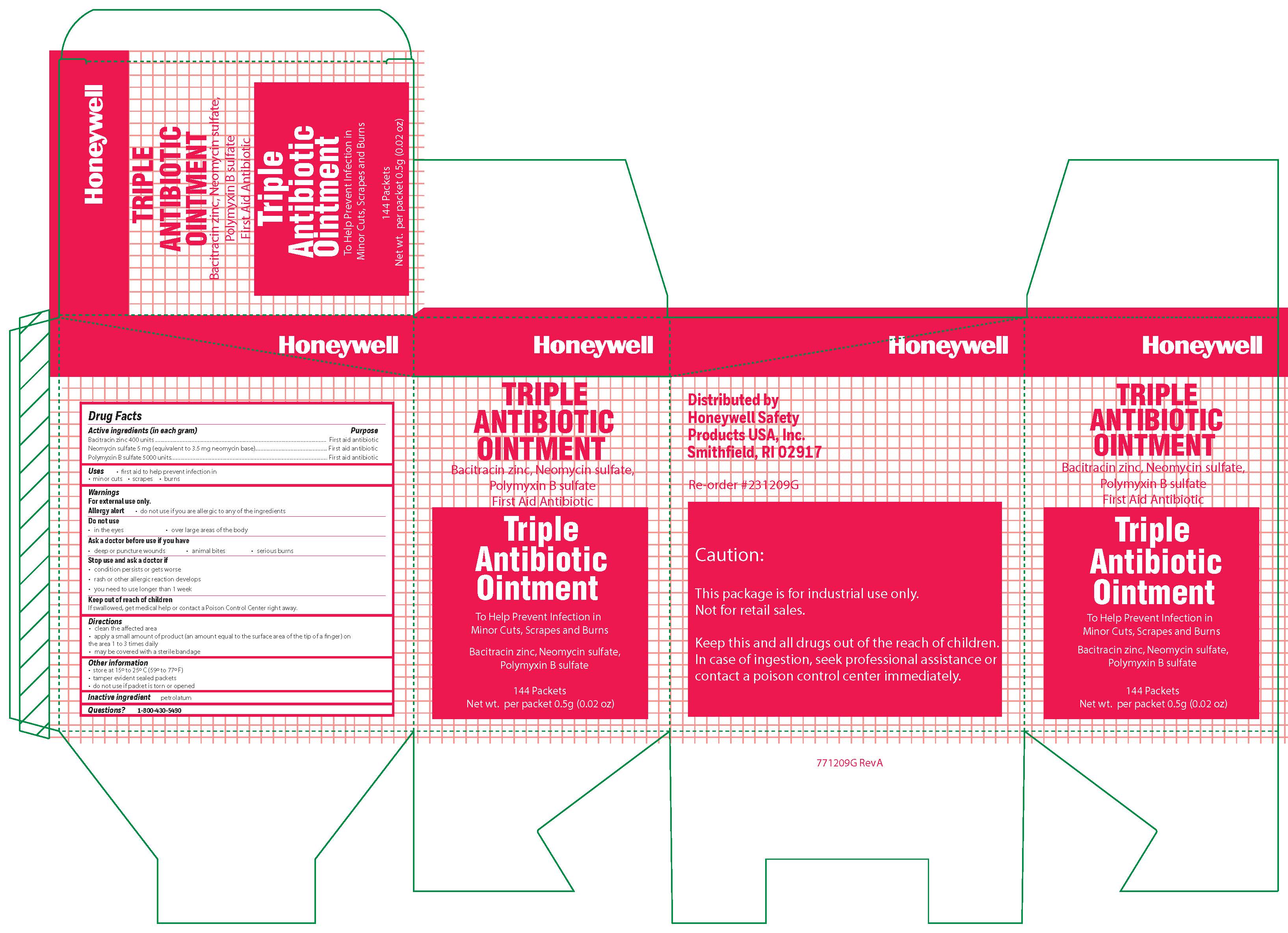
- Triple Active ingredient
- Triple Purpose
- Triple Uses
-
Triple
Warnings
For external use only
Allergy alert: do not use if you are allergic to any of the ingredients
Do not use
- in the eyes
- over large areas of the body
- Ask a doctor before use if you have
- a deep or puncture wounds
- animal bites
- serious burns
- Triple Directions
- Triple Other information
- Triple Inactive ingredient
- Triple Questions?
- Sting Relief Active ingredient (in each wipe)
- Sting Relief Purposse
- Sting Relief Uses
- Sting Relief Warnings
- Sting Relief Directions
- Sting Relief Inactive ingredients
- Sting Relief Questions or Comments?
- Alcohol Active ingredient
- Alcohol Purpose
- Alcohol Uses
- Alcohol Warnings
- Alcohol Directions
- Alcohol Other information
- Alcohol Questions
- Eyewash Active ingredient
- Eyewassh Purpose
- Eyewash Uses
-
Eyewash
Warnings
For external use only
- Obtain immediate medical treatment for all open wounds in or near eyes.
- To avoid contamination, do not touch tip of container to any surface.
- Do not reuse.
- Once opened, discard.
Do not use
- if solution changes color or becomes cloudy
- if you have open wounds in or near the eyes, get medical help right away.
- Eyeash Directions
- Eyewash Inactive ingredients
- Eyeash Questions
-
4363
SF00003199 kit contents
1 1 X 3 WOVEN 100/BOX
1 KNUCKLE BAND 8 PER
2 TRIPLE ANTIBIOTIC 10 PER
1 EYE DRESS PKT W/4 ADH STRIPS
1 TRIANGULAR BDG, NON-STERILE
4 GAUZE COMPRESS, 1728 SQ IN 1
1 GAUZE BANDAGE, 2" X 6 YD,2 PER
2 INSTANT COLD PACK 4" X 6"
2 PAD,NON-ADHR 2" X 3", 4 PER
1 ALCOHOL PREP PADS 10P
2 STING RELIEF WIPES 10 PER BOX
2 NITRILE GLOVES 2PR BBP
6 ADH BDG, CLOTH, 1"X3", 16 PER
1 FIRST AID GUIDE ASHI
1 NON-ADHERENT PADS 2"X3" 10'S
2 COHESIVE TAPE GRN 3/4X30
1 ELASTIC BANDAGE 3" X 4.5YD
1 CO-FLEX LATEX FREE 3"X5YD TAN
1 CTA 3" SINGLE TIP 100/PER
1 CPR MICROSHIELD W/2 PR LTX GLV
1 4OZ BFS EYEWASH TRILINGUAL BOTTLE
1 SCISSOR BDGE 4" RED PLS HDL
1 SPLINTER FORCEP 4 1/2"
1 F A KIT EMPTY BLANK 140
1 LBL STOCK 6-3/8"X4"
1 LBL STOCK 4"X2-7/8"
- Triple Principal Display Panel
- Sting Relief Principal Display Panel
- Alcohol Principal Display Panel
- Eyewash Principal Display Panel
- 4363 Kit Label SF00003199
-
INGREDIENTS AND APPEARANCE
4363 FIRST AID KIT
4363 first aid kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0498-4363 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0498-4363-01 1 in 1 KIT 09/13/2018 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 BOTTLE 118 mL Part 2 20 PACKET 18 g Part 3 10 POUCH 4 mL Part 4 20 POUCH 8 mL Part 1 of 4 EYESALINE EMERGENCY EYEWASH
purified water liquidProduct Information Item Code (Source) NDC:0498-0100 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength WATER (UNII: 059QF0KO0R) (WATER - UNII:059QF0KO0R) WATER 98.6 mL in 100 mL Inactive Ingredients Ingredient Name Strength SODIUM PHOSPHATE, DIBASIC (UNII: GR686LBA74) SODIUM PHOSPHATE, MONOBASIC, MONOHYDRATE (UNII: 593YOG76RN) SODIUM CHLORIDE (UNII: 451W47IQ8X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0498-0100-02 118 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M018 12/18/2018 Part 2 of 4 TRIPLE ANTIBIOTIC
bacitracin zinc, polymyxin b sulfate, neomycin sulfate ointmentProduct Information Item Code (Source) NDC:0498-0750 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POLYMYXIN B SULFATE (UNII: 19371312D4) (POLYMYXIN B - UNII:J2VZ07J96K) POLYMYXIN B 5000 [iU] in 1 g BACITRACIN ZINC (UNII: 89Y4M234ES) (BACITRACIN - UNII:58H6RWO52I) BACITRACIN 400 [iU] in 1 g NEOMYCIN SULFATE (UNII: 057Y626693) (NEOMYCIN - UNII:I16QD7X297) NEOMYCIN 3.5 mg in 1 g Inactive Ingredients Ingredient Name Strength PETROLATUM (UNII: 4T6H12BN9U) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0498-0750-35 0.9 g in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 09/19/2018 Part 3 of 4 ALCOHOL WIPE
isopropyl alcohol swabProduct Information Item Code (Source) NDC:0498-0143 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ISOPROPYL ALCOHOL (UNII: ND2M416302) (ISOPROPYL ALCOHOL - UNII:ND2M416302) ISOPROPYL ALCOHOL 0.7 mL in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0498-0143-04 0.4 mL in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 09/18/2018 Part 4 of 4 STING RELIEF PAD
ethyl alcohol, lidocaine swabProduct Information Item Code (Source) NDC:0498-0733 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE HYDROCHLORIDE (UNII: V13007Z41A) (LIDOCAINE - UNII:98PI200987) LIDOCAINE HYDROCHLORIDE ANHYDROUS 20 mg in 1 mL ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 0.5 mL in 1 mL Inactive Ingredients Ingredient Name Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) MENTHOL (UNII: L7T10EIP3A) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0498-0733-00 0.4 mL in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 12/23/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 09/13/2018 Labeler - Honeywell Safety Products USA, Inc. (118768815)