Label: LEUPROLIDE ACETATE injection, solution
-
Contains inactivated NDC Code(s)
NDC Code(s): 0185-7400-14, 0185-7400-85 - Packager: Eon Labs, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated January 5, 2011
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
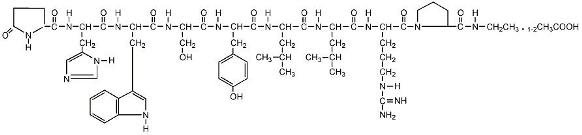
Leuprolide acetate is a synthetic nonapeptide analog of naturally occurring gonadotropin releasing hormone (GnRH or LH-RH). The analog possesses greater potency than the natural hormone. The chemical name is 5-oxo-L-prolyl-L-histidyl-L-tryptophyl-L-seryl-L-tyrosyl-D-leucyl-L-leucyl-L-arginyl-N-ethyl-L-prolinamide acetate (salt) with the following structural formula:
LEUPROLIDE ACETATE INJECTION is a sterile, aqueous solution intended for subcutaneous injection. It is available in a 2.8 mL multiple-dose vial containing leuprolide acetate (5 mg/mL), sodium chloride, USP (6.3 mg/mL) for tonicity adjustment, benzyl alcohol, NF as a preservative (9 mg/mL), and water for injection, USP. The pH may have been adjusted with sodium hydroxide, NF and/or acetic acid, NF. The pH range is 4.0 to 6.0.
-
CLINICAL PHARMACOLOGY
Leuprolide acetate, an LH-RH agonist, acts as a potent inhibitor of gonadotropin secretion when given continuously and in therapeutic doses. Animal and human studies indicate that following an initial stimulation of gonadotropins, chronic administration of leuprolide acetate results in suppression of ovarian and testicular steroidogenesis. This effect is reversible upon discontinuation of drug therapy. Administration of leuprolide acetate has resulted in inhibition of the growth of certain hormone dependent tumors (prostatic tumors in Noble and Dunning male rats and DMBA-induced mammary tumors in female rats) as well as atrophy of the reproductive organs.
In humans, subcutaneous administration of single daily doses of leuprolide acetate results in an initial increase in circulating levels of luteinizing hormone (LH) and follicle stimulating hormone (FSH) leading to a transient increase in levels of the gonadal steroids (testosterone and dihydrotestosterone in males, and estrone and estradiol in pre-menopausal females). However, continuous daily administration of leuprolide acetate results in decreased levels of LH and FSH. In males, testosterone is reduced to castrate levels. In pre-menopausal females, estrogens are reduced to post-menopausal levels. These decreases occur within two to four weeks after initiation of treatment, and castrate levels of testosterone in prostatic cancer patients have been demonstrated for periods of up to five years. Leuprolide acetate is not active when given orally.
Pharmacokinetics
Absorption
Bioavailability by subcutaneous administration is comparable to that by intravenous administration.
Distribution
The mean steady-state volume of distribution of leuprolide following intravenous bolus administration to healthy male volunteers was 27 L. In vitro binding to human plasma proteins ranged from 43% to 49%.
Metabolism
In healthy male volunteers, a 1 mg bolus of leuprolide administered intravenously revealed that the mean systemic clearance was 7.6 L/h, with a terminal elimination half-life of approximately 3 hours based on a two compartment model. In rats and dogs, administration of 14C-labeled leuprolide was shown to be metabolized to smaller inactive peptides, a pentapeptide (Metabolite I), tripeptides (Metabolites II and III) and a dipeptide (Metabolite IV). These fragments may be further catabolized. The major metabolite (M-I) plasma concentrations measured in 5 prostate cancer patients reached maximum concentration 2 to 6 hours after dosing and were approximately 6% of the peak parent drug concentration. One week after dosing, mean plasma M-I concentrations were approximately 20% of mean leuprolide concentrations.
Excretion
Following administration of LEUPROLIDE ACETATE FOR DEPOT SUSPENSION 3.75 mg to 3 patients, less than 5% of the dose was recovered as parent and M-I metabolite in the urine.
Special Populations
The pharmacokinetics of the drug in hepatically and renally impaired patients have not been determined.
Drug Interactions
No pharmacokinetic-based drug-drug interaction studies have been conducted with leuprolide acetate. However, because leuprolide acetate is a peptide that is primarily degraded by peptidase and not by cytochrome P-450 enzymes as noted in specific studies, and the drug is only about 46% bound to plasma proteins, drug interactions would not be expected to occur.
- CLINICAL STUDIES
- INDICATIONS AND USAGE
-
CONTRAINDICATIONS
- LEUPROLIDE ACETATE INJECTION is contraindicated in patients known to be hypersensitive to GnRH, GnRH agonist analogs, or any of the excipients in LEUPROLIDE ACETATE INJECTION: Reports of anaphylactic reactions to synthetic GnRH or GnRH agonist analogs have been reported in the medical literature.1
- LEUPROLIDE ACETATE is contraindicated in women who are or may become pregnant while receiving the drug. LEUPROLIDE ACETATE may cause fetal harm when administered to a pregnant woman. Therefore, the possibility exists that spontaneous abortion may occur if the drug is administered during pregnancy. If this drug is administered during pregnancy or if the patient becomes pregnant while taking any formulation of LEUPROLIDE ACETATE, the patient should be apprised of the potential hazard to the fetus.
-
WARNINGS
Initially, LEUPROLIDE ACETATE, like other LH-RH agonists, causes increases in serum levels of testosterone. Transient worsening of symptoms, or the occurrence of additional signs and symptoms of prostate cancer, may occasionally develop during the first few weeks of LEUPROLIDE ACETATE treatment. A small number of patients may experience a temporary increase in bone pain, which can be managed symptomatically. As with other LH-RH agonists, isolated cases of ureteral obstruction and spinal cord compression have been observed, which may contribute to paralysis with or without fatal complications. Safe use of leuprolide acetate in pregnancy has not been established clinically. Before starting treatment with LEUPROLIDE ACETATE, pregnancy must be excluded (see CONTRAINDICATIONS section). Periodic monitoring of serum testosterone and prostate-specific antigen (PSA) levels is recommended, especially if the anticipated clinical or biochemical response to treatment has not been achieved. It should be noted that results of testosterone determinations are dependent on assay methodology. It is advisable to be aware of the type and precision of the assay methodology to make appropriate clinical and therapeutic decisions.
-
PRECAUTIONS
Patients with metastatic vertebral lesions and/or with urinary tract obstruction should be closely observed during the first few weeks of therapy (see WARNINGS and PRECAUTIONS sections). Patients with known allergies to benzyl alcohol, an ingredient of the drug’s vehicle, may present symptoms of hypersensitivity, usually local, in the form of erythema and induration at the injection site.
Laboratory Tests
Response to leuprolide acetate should be monitored by measuring serum levels of testosterone and prostate-specific antigen (PSA). In the majority of patients, testosterone levels increased above baseline during the first week, declining thereafter to baseline levels or below by the end of the second week of treatment. Castrate levels were reached within two to four weeks and once attained were maintained for as long as drug administration continued.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Two-year carcinogenicity studies were conducted in rats and mice. In rats, a dose-related increase of benign pituitary hyperplasia and benign pituitary adenomas was noted at 24 months when the drug was administered subcutaneously at high daily doses (0.6 to 4 mg/kg). There was a significant but not dose-related increase of pancreatic islet cell adenomas in females and of testicular interstitial cell adenomas in males (highest incidence in the low dose group). In mice no pituitary abnormalities were observed at a dose as high as 60 mg/kg for two years. Patients have been treated with leuprolide acetate for up to three years with doses as high as 10 mg/day and for two years with doses as high as 20 mg/day without demonstrable pituitary abnormalities. Mutagenicity studies have been performed with leuprolide acetate using bacterial and mammalian systems. These studies provided no evidence of a mutagenic potential. Clinical and pharmacologic studies in adults (≥18 years) with leuprolide acetate and similar analogs have shown full reversibility of fertility suppression when the drug is discontinued after continuous administration for periods of up to 24 weeks. However, no clinical studies have been conducted with leuprolide acetate to assess the reversibility of fertility suppression.
Pregnancy
Teratogenic Effects
Pregnancy Category X
(See CONTRAINDICATIONS and WARNINGS sections.) When administered on day 6 of pregnancy at test dosages of 0.00024, 0.0024, and 0.024 mg/kg (1/600 to 1/6 the human dose) to rabbits, LEUPROLIDE ACETATE produced a dose-related increase in major fetal abnormalities. Similar studies in rats failed to demonstrate an increase in major fetal malformations throughout gestation. There was increased fetal mortality and decreased fetal weights with the two higher doses of LEUPROLIDE ACETATE in rabbits and with the highest dose in rats. The effects on fetal mortality are expected consequences of the alterations in hormonal levels brought about by this drug.
-
ADVERSE REACTIONS
Clinical Trials
In the majority of patients testosterone levels increased above baseline during the first week, declining thereafter to baseline levels or below by the end of the second week of treatment. This transient increase was occasionally associated with a temporary worsening of signs and symptoms, usually manifested by an increase in bone pain (see WARNINGS section). In a few cases a temporary worsening of existing hematuria and urinary tract obstruction occurred during the first week. Temporary weakness and paresthesia of the lower limbs have been reported in a few cases. Potential exacerbations of signs and symptoms during the first few weeks of treatment is a concern in patients with vertebral metastases and/or urinary obstruction which, if aggravated, may lead to neurological problems or increase the obstruction. In a comparative trial of LEUPROLIDE ACETATE INJECTION versus DES, in 5% or more of the patients receiving either drug, the following adverse reactions were reported to have a possible or probable relationship to drug as ascribed by the treating physician. Often, causality is difficult to assess in patients with metastatic prostate cancer. Reactions considered not drug related are excluded.
Leuprolide
Acetate
(N=98)DES
(N=101)Number of Reports - *
- Physiologic effect of decreased testosterone
Cardiovascular System Congestive heart failure 1 5 ECG changes/ischemia 19 22 High blood pressure 8 5 Murmur 3 8 Peripheral edema 12 30 Phlebitis/thrombosis 2 10 Gastrointestinal System Anorexia 6 5 Constipation 7 9 Nausea/vomiting 5 17 Endocrine System Decreased testicular size* 7 11 Gynecomastia/breast tenderness or pain* 7 63 Hot flashes* 55 12 Impotence* 4 12 Hemic and Lymphatic System Anemia 5 5 Musculoskeletal System Bone pain 5 2 Myalgia 3 9 Central/Peripheral Nervous System Dizziness/lightheadedness 5 7 General pain 13 13 Headache 7 4 Insomnia/sleep disorders 7 5 Respiratory System Dyspnea 2 8 Sinus congestion 5 6 Integumentary System Dermatitis 5 8 Urogenital System Frequency/urgency 6 8 Hematuria 6 4 Urinary tract infection 3 7 Miscellaneous Asthenia 10 10 In this same study, the following adverse reactions were reported in less than 5% of the patients on LEUPROLIDE ACETATE.
Cardiovascular System–Angina, Cardiac arrhythmias, Myocardial infarction, Pulmonary emboli;
Gastrointestinal System–Diarrhea, Dysphagia, Gastrointestinal bleeding, Gastrointestinal disturbance, Peptic ulcer, Rectal polyps;
Endocrine System–Libido decrease, Thyroid enlargement;
Musculoskeletal System–Joint pain;
Central/Peripheral Nervous System–Anxiety, Blurred vision, Lethargy, Memory disorder, Mood swings, Nervousness, Numbness, Paresthesia, Peripheral neuropathy, Syncope/blackouts, Taste disorders;
Respiratory System–Cough, Pleural rub, Pneumonia, Pulmonary fibrosis;
Integumentary System–Carcinoma of skin/ear, Dry skin, Ecchymosis, Hair loss, Itching, Local skin reactions, Pigmentation, Skin lesions;
Urogenital System–Bladder spasms, Dysuria, Incontinence, Testicular pain, Urinary obstruction;
Miscellaneous–Depression, Diabetes, Fatigue, Fever/chills, Hypoglycemia, Increased BUN, Increased calcium, Increased creatinine, Infection/inflammation, Ophthalmologic disorders, Swelling (temporal bone).
In an additional clinical trial and from long-term observation of both studies, the following additional adverse events (excluding those considered not drug related) were reported for patients receiving LEUPROLIDE ACETATE.
Cardiovascular System–Bradycardia, Carotid bruit, Extrasystole, Palpitations, Perivascular cuffing (eyes), Ruptured aortic aneurysm, Stroke, Tachycardia, Transient ischemic attack;
Gstrointestinal System–Flatus, Dryness of mouth and throat, Hepatitis, Hepatomegaly, Occult blood (rectal exam), Rectal fistula/erythema;
Endocrine System–Libido increase, Thyroid nodule;
Musculoskeletal System–Ankylosing spondylosis, Arthritis, Blurred disc margins, Bone fracture, Muscle stiffness, Muscle tenderness, Pelvic fibrosis, Spasms/cramps;
Central/Peripheral Nervous System–Auditory hallucinations/tinnitus, Decreased hearing, Decreased reflexes, Euphoria, Hyperreflexia, Loss of smell, Motor deficiency;
Respiratory System–Chest tightness, Decreased breathing sounds, Hemoptysis, Pleuritic chest pain, Pulmonary infiltrate, Rales/rhonchi, Rhinitis, Strep throat, Wheezing/bronchitis;
Integumentary System–Boil (pubic), Bruises, Hives, Keratosis, Mole, Shingles, Spiders;
Urogenital System–Blisters on penis, Inguinal hernia, Penile swelling, Post void residual, Prostatic pain, Pyuria;
Miscellaneous–Abdominal distention, Facial swelling/edema, Feet burning, Flu, Eyelid growth, Hypoproteinemia, Accidental injury, Knee effusion, Mass, Pallid, Sallow, Weakness.
Postmarketing
During postmarketing surveillance, which includes other dosage forms and other patient populations, the following adverse events were reported. Symptoms consistent with an anaphylactoid or asthmatic process have been rarely (incidence rate of about 0.002%) reported. Rash, urticaria, and photosensitivity reactions have also been reported. Localized reactions, including induration and abscess, have been reported at the site of injection. Symptoms consistent with fibromyalgia (e.g., joint and muscle pain, headaches, sleep disorders, gastrointestinal distress, and shortness of breath) have been reported individually and collectively.
Cardiovascular System–Hypotension; Myocardial infarction;
Endocrine System–Diabetes;
Gastrointestinal System–Hepatic dysfunction;
Hemic and Lymphatic System–Decreased WBC;
Integumentary System–Hair growth;
Central/Peripheral Nervous System–Spinal fracture/paralysis, Hearing disorder;
Miscellaneous–Hard nodule in throat, Weight gain, Increased uric acid;
Musculoskeletal System–Tenosynovitis-like symptoms;
Respiratory System–Respiratory disorders.
Changes in Bone Density
Decreased bone density has been reported in the medical literature in men who have had orchiectomy or who have been treated with an LH-RH agonist analog. In a clinical trial, 25 men with prostate cancer, 12 of whom had been treated previously with leuprolide acetate for at least six months, underwent bone density studies as a result of pain. The leuprolide-treated group had lower bone density scores than the nontreated control group. It can be anticipated that long periods of medical castration in men will have effects on bone density.
Pituitary Apoplexy
During post-marketing surveillance, rare cases of pituitary apoplexy (a clinical syndrome secondary to infarction of the pituitary gland) have been reported after the administration of gonadotropin-releasing hormone agonists. In a majority of these cases, a pituitary adenoma was diagnosed, with a majority of pituitary apoplexy cases occurring within 2 weeks of the first dose, and some within the first hour. In these cases, pituitary apoplexy has presented as sudden headache, vomiting, visual changes, ophthalmoplegia, altered mental status, and sometimes cardiovascular collapse. Immediate medical attention has been required.
See other LEUPROLIDE ACETATE FOR DEPOT SUSPENSION and LEUPROLIDE ACETATE INJECTION package inserts for other events reported in the same and different patient populations.
-
OVERDOSAGE
In rats subcutaneous administration of 250 to 500 times the recommended human dose, expressed on a per body weight basis, resulted in dyspnea, decreased activity, and local irritation at the injection site. There is no evidence at present that there is a clinical counterpart of this phenomenon. In early clinical trials with leuprolide acetate doses as high as 20 mg/day for up to two years caused no adverse effects differing from those observed with the 1 mg/day dose.
-
DOSAGE AND ADMINISTRATION
The recommended dose is 1 mg (0.2 mL or 20 unit mark) administered as a single daily subcutaneous injection. As with other drugs administered chronically by subcutaneous injection, the injection site should be varied periodically. Each 0.2 mL contains 1 mg of leuprolide acetate, sodium chloride for tonicity adjustment, 1.8 mg of benzyl alcohol as preservative and water for injection. The pH may have been adjusted with sodium hydroxide and/or acetic acid. Follow the pictorial directions on the Administering the Injection Insert. NOTE: As with all parenteral products, inspect the solution for discoloration and particulate matter before each use.
-
HOW SUPPLIED
LEUPROLIDE ACETATE INJECTION is a sterile solution supplied in a 2.8 mL multiple-dose vial. The vial is packaged as a 14 Day Patient Administration Kit with 14 disposable syringes and 28 alcohol swabs, NDC 0185-7400-85. Store below 77°F (25°C). Do not freeze. Protect from light; store vial in carton until use.
- REFERENCE
-
INFORMATION FOR PATIENTS
Be sure to consult your physician with any questions you may have or for information about LEUPROLIDE ACETATE INJECTION and its use.
WHAT IS LEUPROLIDE ACETATE?
LEUPROLIDE ACETATE INJECTION is chemically similar to gonadotropin releasing hormone (GnRH or LH-RH) a hormone which occurs naturally in your body. Normally, your body releases small amounts of LH-RH and this leads to events which stimulate the production of sex hormones. However, when you inject LEUPROLIDE ACETATE INJECTION, the normal events that lead to sex hormone production are interrupted and testosterone is no longer produced by the testes. LEUPROLIDE ACETATE must be injected because, like insulin which is injected by diabetics, LEUPROLIDE ACETATE is inactive when taken by mouth. If you were to discontinue the drug for any reason, your body would begin making testosterone again.
DIRECTIONS FOR USING LEUPROLIDE ACETATE
- Wash hands thoroughly with soap and water.
- If using a new bottle for the first time, flip off the plastic cover to expose the grey rubber stopper. Wipe metal ring and rubber stopper with an alcohol wipe each time you use LEUPROLIDE ACETATE. Check the liquid in the container. If it is not clear or has particles in it, DO NOT USE IT. Exchange it at your pharmacy for another container.
- Remove outer wrapping from one syringe. Pull plunger back until the tip of the plunger is at the 0.2 mL or 20-unit mark.
- Take cover off needle. Push the needle through the center of the rubber stopper on the LEUPROLIDE ACETATE bottle.
- Push the plunger all the way in to inject air into the bottle.
- Keep the needle in the bottle and turn the bottle upside down. Check to make sure the tip of the needle is in the liquid. Slowly pull back on the plunger, until the syringe fills to the 0.2 mL or 20-unit mark.
- Toward the end of a two-week period, the amount of LEUPROLIDE ACETATE left in the bottle will be small. Take special care to hold the bottle straight and to keep the needle tip in liquid while pulling back on the plunger.
- Keeping the needle in the bottle and the bottle upside down, check for air bubbles in the syringe. If you see any, push the plunger slowly in to push the air bubble back into the bottle. Keep the tip of the needle in the liquid and pull the plunger back again to fill to the 0.2 mL or 20-unit mark.
- Do this again if necessary to eliminate air bubbles.
- To protect your skin, inject each daily dose at a different body spot.
- Choose an injection spot. Cleanse the injection spot with another alcohol wipe.
- Hold the syringe in one hand. Hold the skin taut, or pull up a little flesh with the other hand, as you were instructed.
- Holding the syringe as you would a pencil, thrust the needle all the way into the skin at a 90° angle. Push the plunger to administer the injection.
- Hold an alcohol wipe down on your skin where the needle is inserted and withdraw the needle at the same angle it was inserted.
- Use the disposable syringe only once and dispose of it properly as you were instructed. Needles thrown into a garbage bag could accidentally stick someone. NEVER LEAVE SYRINGES, NEEDLES OR DRUGS WHERE CHILDREN CAN REACH THEM.
SOME SPECIAL ADVICE
- You may experience hot flashes when using LEUPROLIDE ACETATE INJECTION. During the first few weeks of treatment you may experience increased bone pain, increased difficulty in urinating, and less commonly but most importantly, you may experience the onset or aggravation of nerve symptoms. In any of these events, discuss the symptoms with your doctor. Like other treatment options, LEUPROLIDE ACETATE may cause impotence. Notify your doctor if you develop new or worsened symptoms after beginning LEUPROLIDE ACETATE treatment.
- You may experience some irritation at the injection site, such as burning, itching or swelling. These reactions are usually mild and go away. If they do not, tell your doctor.
- If you have experienced an allergic reaction to other drugs like LEUPROLIDE ACETATE, you should not use this drug.
- Do not stop taking your injections because you feel better. You need an injection every day to make sure LEUPROLIDE ACETATE keeps working for you.
- If you need to use an alternate to the syringe supplied with LEUPROLIDE ACETATE INJECTION, low-dose insulin syringes should be utilized.
- When the drug level gets low, take special care to hold the bottle straight up and down and to keep the needle tip in liquid while pulling back on the plunger.
- Do not try to get every last drop out of the bottle. This will increase the possibility of drawing air into the syringe and getting an incomplete dose. Some extra drug has been provided so that you can withdraw the recommended number of doses.
- Tell your pharmacist when you will need LEUPROLIDE ACETATE so it will be at the pharmacy when you need it.
- Store below 77°F (25°C). Do not store near a radiator or other very warm place. Do not freeze. Protect from light; store vial in carton until use.
- Do not leave your drug or hypodermic syringes where anyone can pick them up.
- Keep this and all other medications out of reach of children.
Manufactured by Ben Venue Laboratories, Inc., Bedford, OH 44146
Manufactured for Sandoz Inc., Princeton, NJ 08540 and Oakwood Laboratories, L.L.C. Oakwood Village, OH 44146
OS8247
Rev. 12/07
- 1 mg .2 mL Vial
-
INGREDIENTS AND APPEARANCE
LEUPROLIDE ACETATE
leuprolide acetate injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0185-7400 Route of Administration SUBCUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LEUPROLIDE ACETATE (UNII: 37JNS02E7V) (LEUPROLIDE - UNII:EFY6W0M8TG) LEUPROLIDE ACETATE 1 mg in .2 mL Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) BENZYL ALCOHOL (UNII: LKG8494WBH) ACETIC ACID (UNII: Q40Q9N063P) SODIUM HYDROXIDE (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0185-7400-85 1 in 1 CARTON 1 NDC:0185-7400-14 2.8 mL in 1 VIAL, MULTI-DOSE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA074728 08/04/1998 Labeler - Eon Labs, Inc. (012656273) Establishment Name Address ID/FEI Business Operations Ben Venue Laboratories Inc. 004327953 MANUFACTURE Establishment Name Address ID/FEI Business Operations Bachem, Inc. 077243640 API MANUFACTURE


