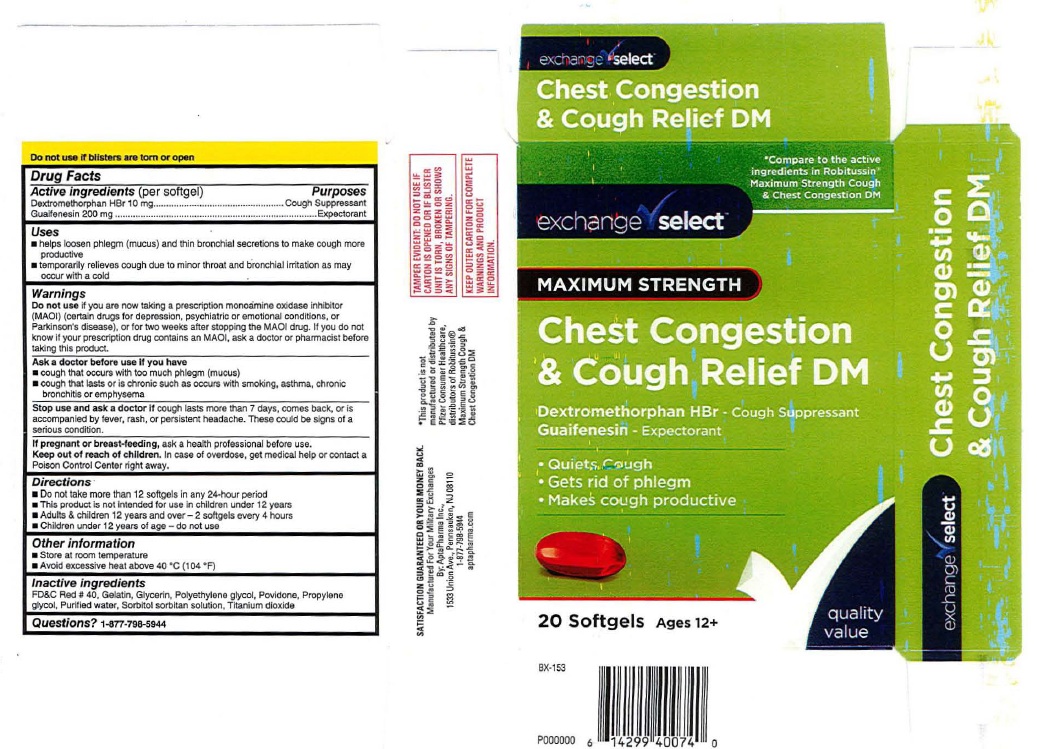
Label: CHEST CONGESTION AND COUGH RELIEF DM- dextromethorphan hbr, guaifenesin capsule, liquid filled
- NDC Code(s): 55301-601-02
- Packager: ARMY AND AIR FORCE EXCHANGE SERVICE
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 10, 2025
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
ACTIVE INGREDIENTDrug Facts - Active ingredients (per softgel) Dextromethorphan HBr 10 mg - Guaifenesin 200 mg
-
PURPOSEPurposes - Dextromethorphan HBr ..........................Cough Suppressant - Guaifenesin ....................................Expectorant
-
INDICATIONS & USAGEUses - ■ helps loosen phlegm (mucus) and thin broochial secretions to make cough more - productlve - ■ temporarily relieves cough due to minor throat and bronchial irritation as may - occur with a ...
-
WARNINGSWarnings - Do not use if you are now taking a prescription monoamine oxidase inhibitor - (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or - Parkinson's disease), or for two ...
-
ASK DOCTORAsk a doctor before use if you have - ■ cough that occurs with too much phlegm (mucus) ■ cough that lasts or is chronic such as occurs with smoking, asthma, chronic - bronchitis or emphysema
-
STOP USEStop use and ask a doctor if cough lasts more than 7 days, comes back, or is - accompanied by fever, rash, or persistent headache. These could be signs of a - serious condition.
-
PREGNANCY OR BREAST FEEDINGIf pregnant or breast-feeding, ask a health professional before use.
-
KEEP OUT OF REACH OF CHILDRENKeep out of reach of children. In case of overdose, get medical help or contact a - Poison Control Center right away.
-
DOSAGE & ADMINISTRATIONDirections - ■ Do not take more than 12 softgels in any 24·hour period - ■ This product is nol lntended for use in children under 12 years - ■ Adults & children 12 years and over - 2 softgels every 4 ...
-
OTHER SAFETY INFORMATIONOther information - ■ Store at room temperature - ■ Avoid excessive heat above 40'C (104'F)
-
INACTIVE INGREDIENTInactive ingredients - FD&C Red # 40, Gelatin, Glycerin, Polyethylene glycol, Povidone, Propylene - glycol, Purified water, Sorbitol sorbllan solulion, Titanium dioxide
-
QUESTIONSQuestions? 1-877-798-5944
-
Product Label: exchange select Chest Congestion & Cough Relief DM
exchange select - Chest Congestion - & Cough Relief DM - 20 Softgels Ages 12+ Manufactured For Your Military Exchange - By AptaPharma Inc., 1533 Union Avenue, Pennsauken, NJ ...
-
INGREDIENTS AND APPEARANCEProduct Information