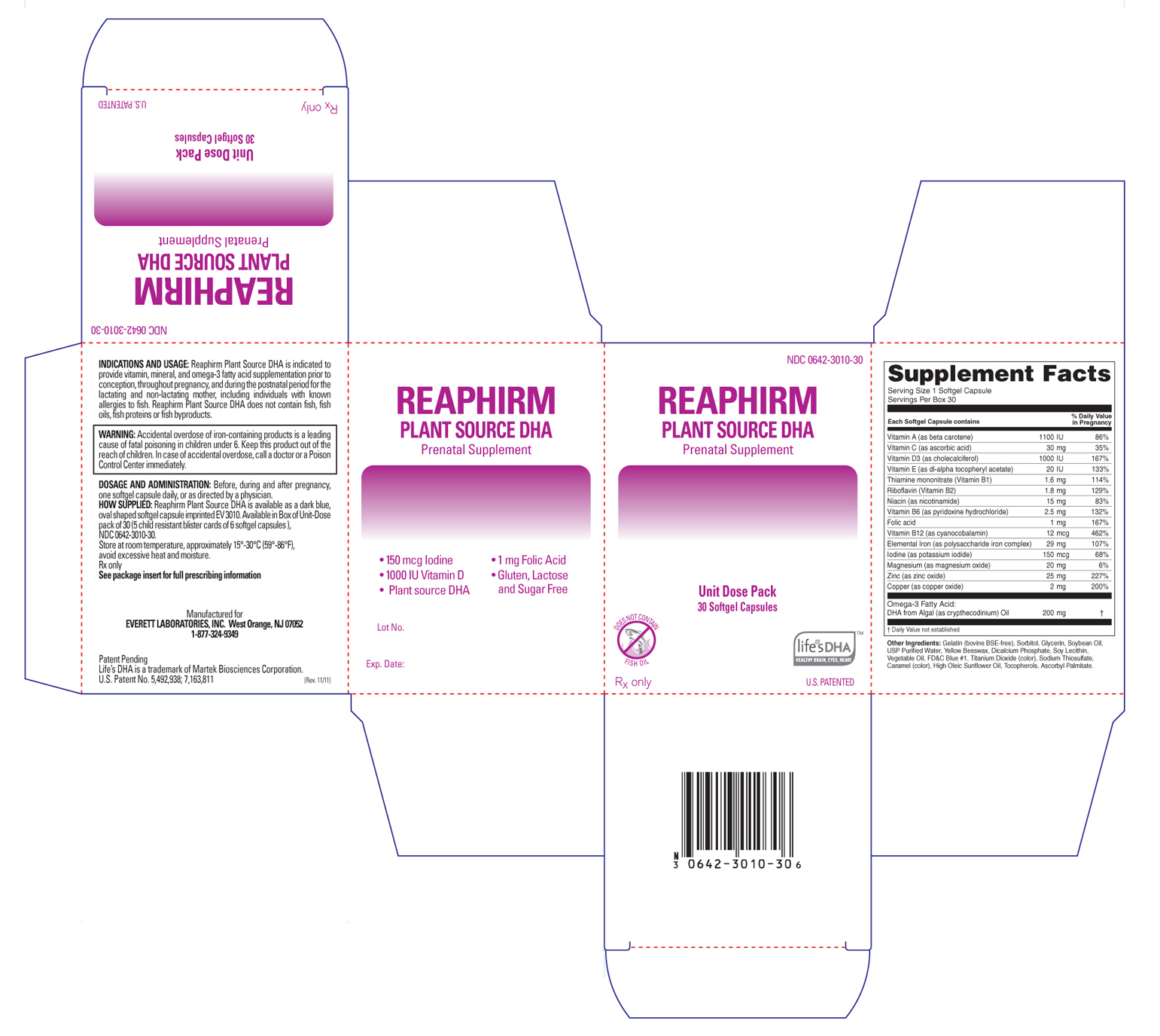
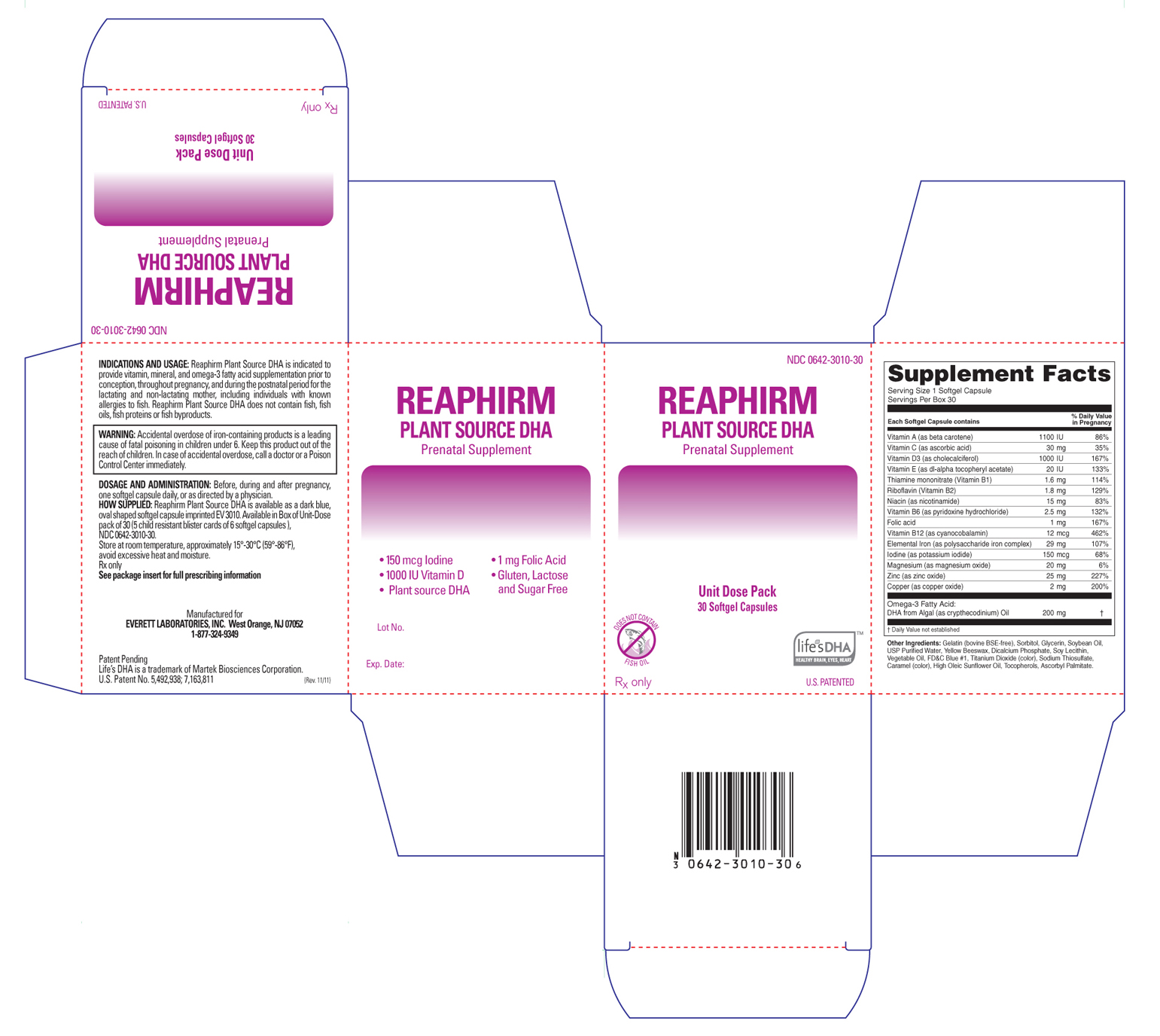
Label: REAPHIRM PLANT SOURCE DHA- prenatal supplement capsule, gelatin coated
-
Contains inactivated NDC Code(s)
NDC Code(s): 0642-3010-30 - Packager: Everett Laboratories, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated November 28, 2011
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
Contraindications
Reaphirm Plant Source DHA is contraindicated in patients with hypersensitivity to any of its components or color additives.
Folic acid is contraindicated in patients with untreated and uncomplicated pernicious anemia, and in those with anaphylactic sensitivity to folic acid.
Iron Therapy is contraindicated in patients with hemochromatosis and patients with iron storage disease or the potential for iron storage disease due to chronic hemolytic anemia (e.g., inherited anomalies of hemoglobin structure or synthesis and/or red cell enzyme deficiencies, etc.), pyridoxine responsive anemia, or cirrhosis of the liver.
Cyanocobalamin is contraindicated in patients with sensitivity to cobalt or to cyanocobalamin (Vitamin B12).
Refer to MM1carton label
- BOXED WARNING (What is this?)
-
WARNINGS/PRECAUTIONS:
Reaphirm Plant Source DHA should be used with caution in patients with known sensitivity or allergy to soy.
Vitamin D supplementation should be used with caution in those with hypercalcemia or conditions that may lead to hypercalcemia such as hyperparathyroidism and those who form calcium-containing kigney stones. High doses of vitamin D can lead to elevated levels of calcium that reside in the blood and soft tissues. Bone pain, high blood pressure, formation of kidney stones, renal failure, and increased risk of heart disease can occur.
Iodine should be used with caution in patients with an overactive thyroid.
Prolonged use of iron salts may produce iron storage disease.
Folic acid, especially in doses above 0.1 mg daily, may obscure pernicious anemia, in that hematologic remission may occur while neurological manifestations remain progressive.
The use of folic acid doses above 1 mg daily may precipitate or exacerbate the neurological damage of vitamin B12 deficiency.
Consumption of more than 3 grams of omega-3 fatty acids per day from all sources may lead to excessive bleeding. Supplemental intake of omega-3 fatty acids such as DHA exceeding 2 grams per day is not recommended.
Avoid Overdosage. Keep out of the reach of children.
Drug Interactions:
Medications for an overactive thyroid (anti-thyroid drugs) used in conjunction with iodine supplementation may lead to hypothyroidism.
Medications for hypertension used in conjunction with iodine supplementation may increase potassium.
High doses of folic acid may result in decreased serum levels of the anticonvulsant drugs.
Vitamin D supplementation should not be given with large amounts of calcium in those with hypercalcemia or conditions that may lead to hypercalcemia such as hyperparathyroidism and those who form calcium-containing kidney stones.
Zinc can inhibit the absorption of certain antibiotics; take at least 2 hours apart to minimize interactions.
Consult appropriate references for additional specific vitamin-drug interactions.
Information for Patients: Patients should be counseled to disclose all medical conditions, including use of all medications, vitamins and supplements, pregnancy, and breast-feeding.
Pediatric Use: Not for pediatric use.
- Dosage and Administration
-
How Supplied
Reaphirm Plant Source DHA is available as a dark blue, oval shaped softgel capsule imprinted "EV3010". Available in Box of Unit-Dose pack of 30 (5 child resistant blister cards of 6 softgel capsules).
Store at room temperature, approximately 15-30C (59-86F), avoid excessive heat.
Rx Only
Manufactured for
EVERETT LABORATORIES, INC.
West Orange, NJ 07052
1-877-324-9349
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
REAPHIRM PLANT SOURCE DHA
prenatal supplement capsule, gelatin coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0642-3010 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Vitamin A (UNII: 81G40H8B0T) (Beta Carotene - UNII:01YAE03M7J) Beta Carotene 1100 [iU] Ascorbic Acid (UNII: PQ6CK8PD0R) (Ascorbic Acid - UNII:PQ6CK8PD0R) Ascorbic Acid 30 mg Thiamine Mononitrate (UNII: 8K0I04919X) (Thiamine - UNII:X66NSO3N35) Thiamine Mononitrate 1.6 mg Riboflavin (UNII: TLM2976OFR) (Riboflavin - UNII:TLM2976OFR) Riboflavin 1.8 mg Niacin (UNII: 2679MF687A) (Niacinamide - UNII:25X51I8RD4) Niacinamide 15 mg Pyridoxine Hydrochloride (UNII: 68Y4CF58BV) (Pyridoxine - UNII:KV2JZ1BI6Z) Pyridoxine 2.5 mg Cyanocobalamin (UNII: P6YC3EG204) (Cyanocobalamin - UNII:P6YC3EG204) Cyanocobalamin .012 mg Folic Acid (UNII: 935E97BOY8) (Folic Acid - UNII:935E97BOY8) Folic Acid 1 mg Iodine (UNII: 9679TC07X4) (Iodine - UNII:9679TC07X4) Iodine 0.150 mg Magnesium (UNII: I38ZP9992A) (Magnesium Oxide - UNII:3A3U0GI71G) Magnesium Oxide 20 mg Zinc (UNII: J41CSQ7QDS) (Zinc Oxide - UNII:SOI2LOH54Z) Zinc Oxide 25 mg Copper (UNII: 789U1901C5) (Cupric Oxide - UNII:V1XJQ704R4) Cupric Oxide 2 mg Vitamin D (UNII: 9VU1KI44GP) (Cholecalciferol - UNII:1C6V77QF41) Cholecalciferol 1000 [iU] Omega-3 Fatty Acids (UNII: 71M78END5S) (Doconexent - UNII:ZAD9OKH9JC) Doconexent 200 mg Vitamin E (UNII: H4N855PNZ1) (Alpha-Tocopherol - UNII:H4N855PNZ1) Alpha-Tocopherol 20 [iU] Iron (UNII: E1UOL152H7) (Iron - UNII:E1UOL152H7) Iron 29 mg Inactive Ingredients Ingredient Name Strength Gelatin (UNII: 2G86QN327L) Sorbitol (UNII: 506T60A25R) Glycerin (UNII: PDC6A3C0OX) Soybean Oil (UNII: 241ATL177A) Water (UNII: 059QF0KO0R) Yellow Wax (UNII: 2ZA36H0S2V) Anhydrous Dibasic Calcium Phosphate (UNII: L11K75P92J) Lecithin, Soybean (UNII: 1DI56QDM62) Titanium Dioxide (UNII: 15FIX9V2JP) Sodium Thiosulfate (UNII: HX1032V43M) Sunflower Oil (UNII: 3W1JG795YI) Tocopherol (UNII: R0ZB2556P8) Ascorbyl palmitate (UNII: QN83US2B0N) Caramel (UNII: T9D99G2B1R) FD&C Blue NO. 1 (UNII: H3R47K3TBD) Corn Oil (UNII: 8470G57WFM) Product Characteristics Color blue (Dark Blue) Score no score Shape OVAL (size 12 Oval Capsule) Size 10mm Flavor Imprint Code EV3010 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0642-3010-30 5 in 1 BOX 1 6 in 1 BLISTER PACK Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 12/05/2011 Labeler - Everett Laboratories, Inc. (071170534) Registrant - Everett Laboratories, Inc. (071170534)