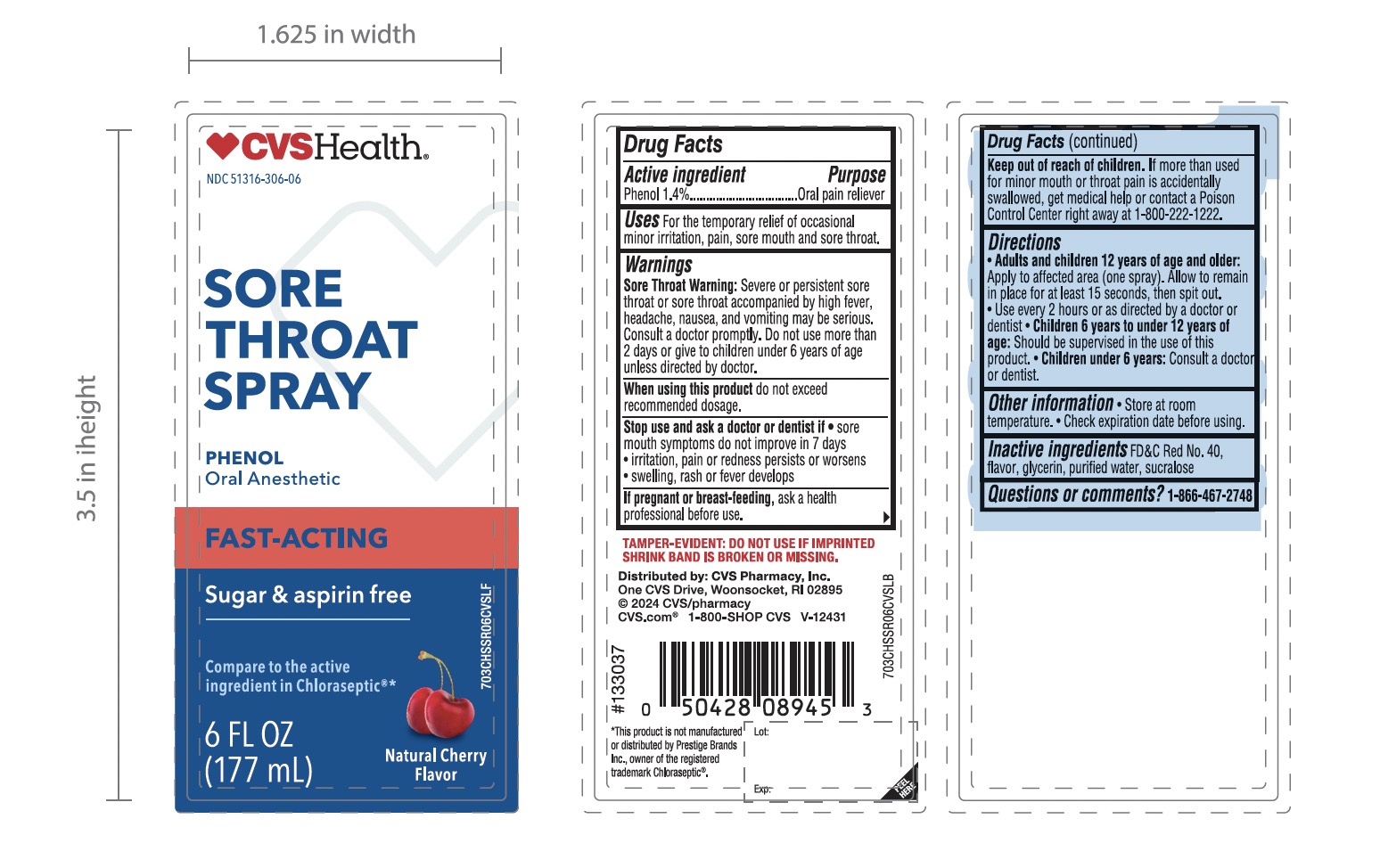
Label: CVS SORE THROAT CHERRY- phenol spray
- NDC Code(s): 51316-306-06
- Packager: CVS Pharmacy,Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated July 4, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
-
Warnings
Sore Throat Warning:Severe or persistent sore throat or sore throat accompanied by high fever, headache, nausea, and vomiting may be serious.
Consult doctor promptly. Do not use more than 2 days or administer to children under 6 years of age unless directed by doctor.
-
Directions
- Adults and children 12 years of age and older:
- Apply to the affected area (one spray)
- Allow to remain in place for at least 15 seconds, then spit out
- Use every 2 hours or as directed by a doctor or dentist
- Children 6 years to under 12 years of ageshould be supervised in the use of this product
- Children under 6 years:Consult a doctor or dentist.
- Other information
- Inactive ingredients
-
Principal Display Panel
*Compare to active ingredient in Chloraseptic ®
NDC 51316-306-06
SORE THROAT SPRAY
Phenol / oral Pain Reliever
- Fast acting
- Numbs on Contact
SUGAR-FREE
ASPIRIN-FREE
NATURAL CHERRY FLAVOR
6 FL OZ (177 mL)
*This product is not manufactured or distributed by Prestige Brands Inc, owner of the registered trademark Chloraseptic ®
Tamper-Evident: Do not use if imprinted shrink band is broken or missing.
Distributed by:
-
INGREDIENTS AND APPEARANCE
CVS SORE THROAT CHERRY
phenol sprayProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:51316-306 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PHENOL (UNII: 339NCG44TV) (PHENOL - UNII:339NCG44TV) PHENOL 1.4 g in 100 mL Inactive Ingredients Ingredient Name Strength FD&C RED NO. 40 (UNII: WZB9127XOA) GLYCERIN (UNII: PDC6A3C0OX) WATER (UNII: 059QF0KO0R) SUCRALOSE (UNII: 96K6UQ3ZD4) Product Characteristics Color red Score Shape Size Flavor CHERRY (NATURAL) Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:51316-306-06 177 mL in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product 03/11/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M022 08/18/2015 Labeler - CVS Pharmacy,Inc. (062312574)