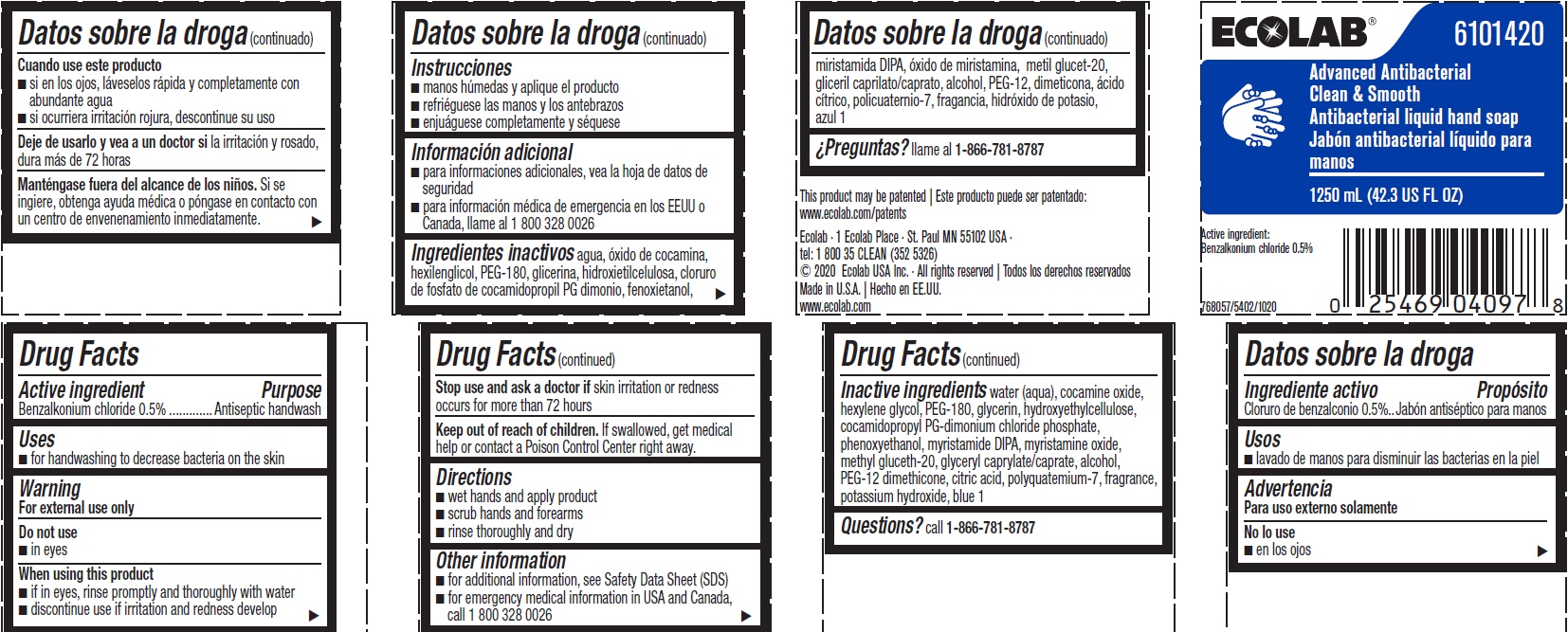
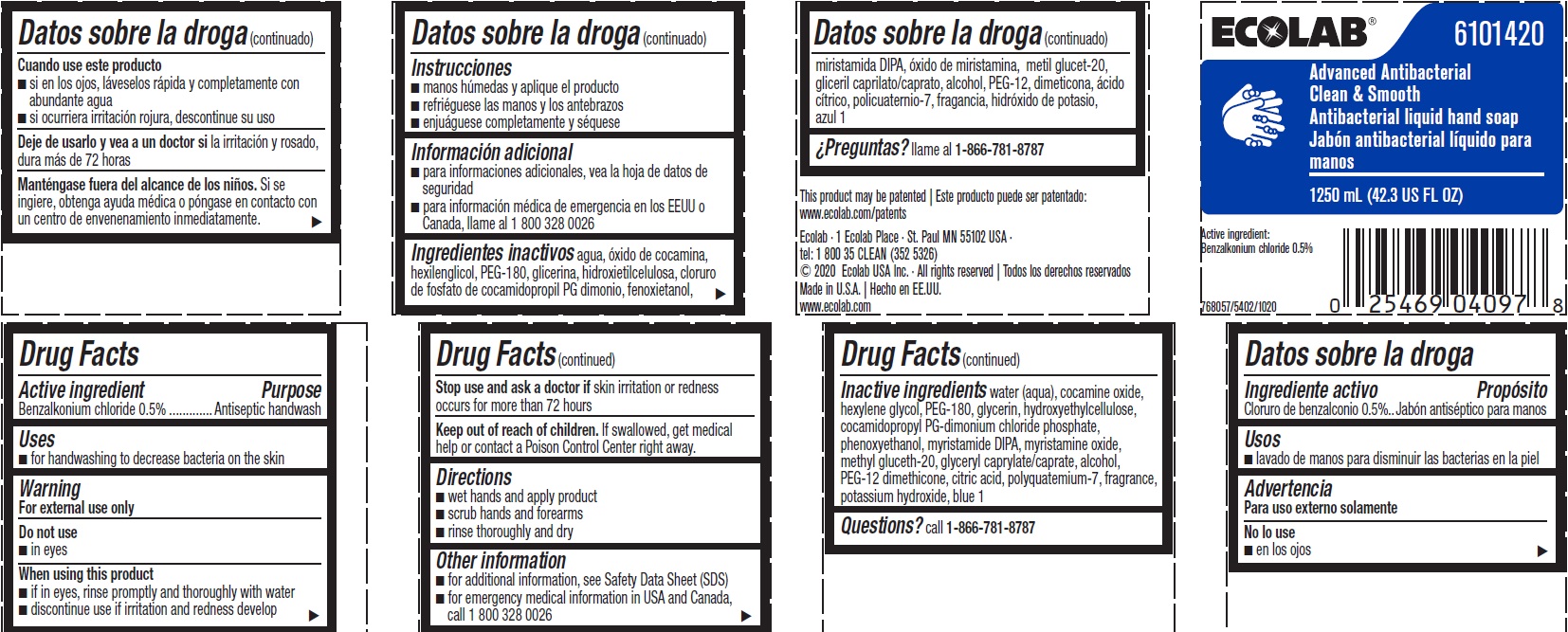
Label: ADVANCED ANTIBACTERIAL CLEAN AND SMOOTH- benzalkonium chloride solution
- NDC Code(s): 47593-575-11, 47593-575-41, 47593-575-59
- Packager: Ecolab Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated November 30, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
- Warnings
- Directions
- Other information
-
INACTIVE INGREDIENT
Inactive ingredients water (aqua), cocamine oxide, hexylene glycol, PEG-180, glycerin, hydroxyethylcellulose, cocamidopropyl PG-dimonium chloride phosphate, phenoxyethanol, myristamide DIPA, myristamine oxide, methyl gluceth-20, glyceryl caprylate/caprate, alcohol, PEG-12 dimethicone, citric acid, polyquatemium-7, fragrance, potassium hydroxide, blue 1
- QUESTIONS
-
Representative label & Primary display panel
ECOLAB 6101420
Advanced Antibacterial
Clean & Smooth
Antibacterial liquid hand soap
1250 mL (42.3 US FL OZ)
Active Ingredient
Benzalkonium chloride 0.5%
This product may be patented | Este producto puede ser patentado: www.ecolab.com/patents
Ecolab · 1 Ecolab Place · St. Paul MN 55102 USA ·
tel: 1 800 35 CLEAN (352 5326)
© 2020 Ecolab USA Inc. · All rights reserved | Todos los derechos reservados
Made in U.S.A. | Hecho en EE.UU.
www.ecolab.com
-
INGREDIENTS AND APPEARANCE
ADVANCED ANTIBACTERIAL CLEAN AND SMOOTH
benzalkonium chloride solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:47593-575 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 5 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) COCAMINE OXIDE (UNII: QWA2IZI6FI) HEXYLENE GLYCOL (UNII: KEH0A3F75J) POLYETHYLENE GLYCOL 8000 (UNII: Q662QK8M3B) GLYCERIN (UNII: PDC6A3C0OX) HYDROXYETHYL CELLULOSE (4000 MPA.S AT 1%) (UNII: ZYD53NBL45) COCAMIDOPROPYL PG-DIMONIUM CHLORIDE PHOSPHATE (UNII: H2KVQ74JM4) PHENOXYETHANOL (UNII: HIE492ZZ3T) MYRISTIC DIISOPROPANOLAMIDE (UNII: 17DN142CTK) MYRISTAMINE OXIDE (UNII: J086PM3RRT) METHYL GLUCETH-20 (UNII: J3QD0LD11P) GLYCERYL CAPRYLATE/CAPRATE (UNII: G7515SW10N) ALCOHOL (UNII: 3K9958V90M) PEG-12 DIMETHICONE (UNII: ZEL54N6W95) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) POLYQUATERNIUM-7 (70/30 ACRYLAMIDE/DADMAC; 1600000 MW) (UNII: 0L414VCS5Y) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:47593-575-11 3780 mL in 1 BOTTLE; Type 0: Not a Combination Product 05/25/2017 2 NDC:47593-575-59 1250 mL in 1 BOTTLE; Type 0: Not a Combination Product 05/25/2017 3 NDC:47593-575-41 750 mL in 1 BOTTLE; Type 0: Not a Combination Product 05/25/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) 05/25/2017 Labeler - Ecolab Inc. (006154611)