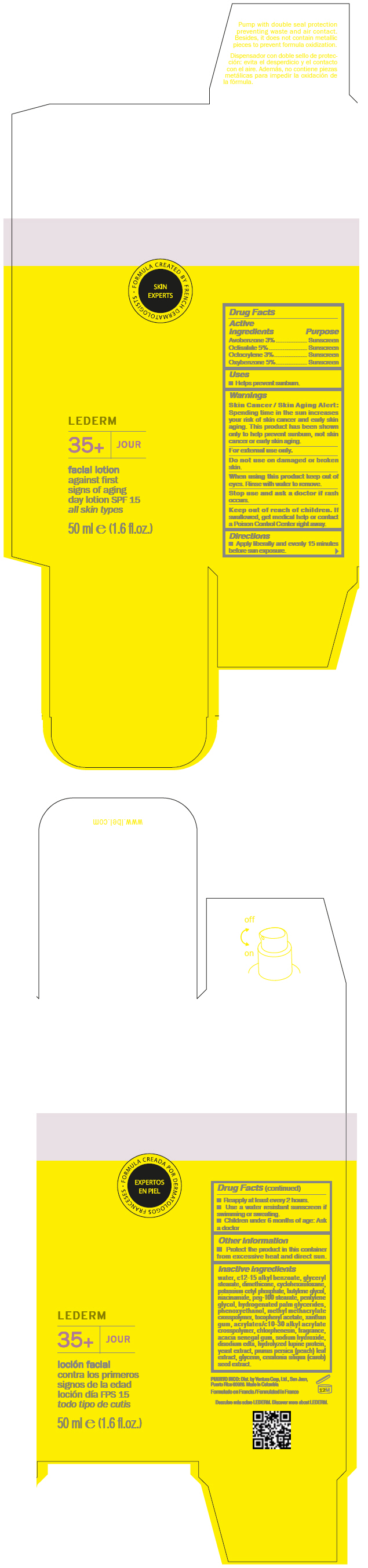
Label: LBEL LEDERM 35 PLUS JOUR FACIAL AGAINST FIRST SIGNS OF AGING- avobenzone, octisalate, octocrylene, and oxybenzone lotion
-
Contains inactivated NDC Code(s)
NDC Code(s): 13537-995-01, 13537-995-02 - Packager: Ventura Corporation LTD
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated November 17, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- Uses
- Warnings
- Directions
- Other information
-
Inactive ingredients
water, c12-15 alkyl benzoate, glyceryl stearate, dimethicone, cyclohexasiloxane, potassium cetyl phosphate, butylene glycol, niacinamide, peg-100 stearate, pentylene glycol, hydrogenated palm glycerides, phenoxyethanol, methyl methacrylate crosspolymer, tocopheryl acetate, xanthan gum, acrylates/c10-30 alkyl acrylate crosspolymer, chlorphenesin, fragrance, acacia senegal gum, sodium hydroxide, disodium edta, hydrolyzed lupine protein, yeast extract, prunus persica (peach) leaf extract, glycerin, ceratonia siliqua (carob) seed extract.
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 50 ml Bottle Box
-
INGREDIENTS AND APPEARANCE
LBEL LEDERM 35 PLUS JOUR FACIAL AGAINST FIRST SIGNS OF AGING
avobenzone, octisalate, octocrylene, and oxybenzone lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:13537-995 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Avobenzone (UNII: G63QQF2NOX) (Avobenzone - UNII:G63QQF2NOX) Avobenzone 0.03 g in 1 mL Octisalate (UNII: 4X49Y0596W) (Octisalate - UNII:4X49Y0596W) Octisalate 0.05 g in 1 mL Octocrylene (UNII: 5A68WGF6WM) (Octocrylene - UNII:5A68WGF6WM) Octocrylene 0.03 g in 1 mL Oxybenzone (UNII: 95OOS7VE0Y) (Oxybenzone - UNII:95OOS7VE0Y) Oxybenzone 0.05 g in 1 mL Inactive Ingredients Ingredient Name Strength water (UNII: 059QF0KO0R) alkyl (c12-15) benzoate (UNII: A9EJ3J61HQ) glyceryl monostearate (UNII: 230OU9XXE4) dimethicone (UNII: 92RU3N3Y1O) cyclomethicone 6 (UNII: XHK3U310BA) potassium cetyl phosphate (UNII: 03KCY6P7UT) butylene glycol (UNII: 3XUS85K0RA) niacinamide (UNII: 25X51I8RD4) peg-100 stearate (UNII: YD01N1999R) pentylene glycol (UNII: 50C1307PZG) hydrogenated palm glycerides (UNII: YCZ8EM144Q) phenoxyethanol (UNII: HIE492ZZ3T) .alpha.-tocopherol acetate (UNII: 9E8X80D2L0) xanthan gum (UNII: TTV12P4NEE) carbomer interpolymer type a (allyl sucrose crosslinked) (UNII: 59TL3WG5CO) chlorphenesin (UNII: I670DAL4SZ) acacia (UNII: 5C5403N26O) sodium hydroxide (UNII: 55X04QC32I) edetate disodium anhydrous (UNII: 8NLQ36F6MM) yeast, unspecified (UNII: 3NY3SM6B8U) prunus persica leaf (UNII: VN3501T41P) glycerin (UNII: PDC6A3C0OX) carob (UNII: 5MG5Z946UO) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13537-995-02 1 in 1 BOX 10/24/2016 1 NDC:13537-995-01 50 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 10/24/2016 Labeler - Ventura Corporation LTD (602751344) Establishment Name Address ID/FEI Business Operations Bel Star S.A. (Colombia) 880160197 MANUFACTURE(13537-995)