Label: KINRAY VITAMIN A D- petrolatum ointment
-
Contains inactivated NDC Code(s)
NDC Code(s): 63777-214-02, 63777-214-07 - Packager: Kinray Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated February 11, 2014
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
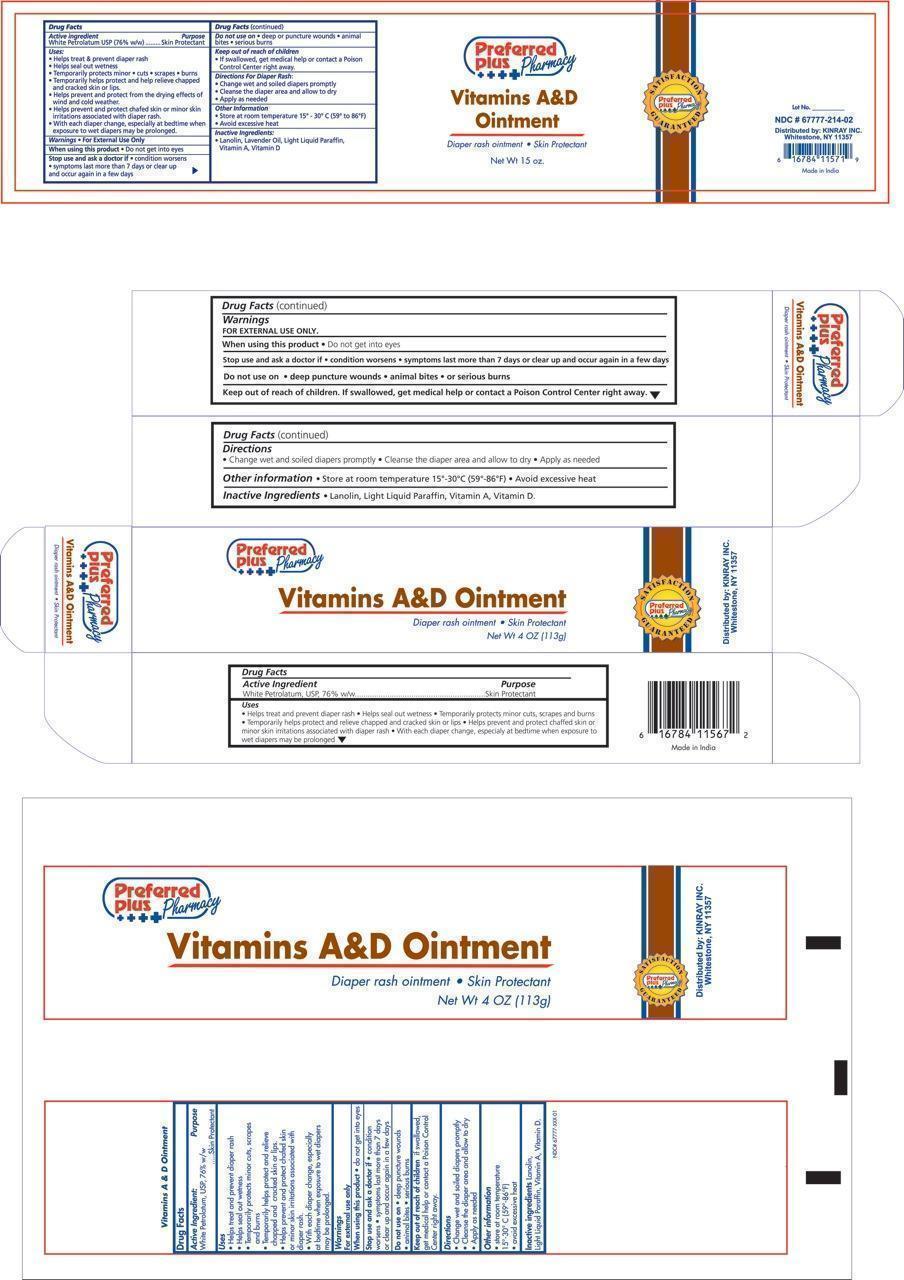
- ACTIVE INGREDIENT
-
Kinray Uses:
- Helps treat and prevent diaper rash
- Helps seal out wetness
- Temporarily protects minor * cuts * scrapes * burns
- Temporarily helps protect and help relieve chapped and cracked skin and lips
- Helps prevent and protect from the drying effects of wind and cold weather
- Helps prevent and protect chafed skin or minor skin irritations associated with diaper rash
- With each diaper change, especially at bedtime when exposure to wet diapers may be prolonged
- Warnings:
- When using this product:
- Stop use and ask a doctor if:
- Keep out of reach of children
- Do Not Use
- Directions for diaper rash:
- Other information:
- Inactive Ingredients:
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
KINRAY VITAMIN A D
petrolatum ointmentProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:63777-214 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PETROLATUM (UNII: 4T6H12BN9U) (PETROLATUM - UNII:4T6H12BN9U) PETROLATUM .76 g in 1 g Inactive Ingredients Ingredient Name Strength VITAMIN A PALMITATE (UNII: 1D1K0N0VVC) LANOLIN (UNII: 7EV65EAW6H) LIGHT MINERAL OIL (UNII: N6K5787QVP) CHOLECALCIFEROL (UNII: 1C6V77QF41) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63777-214-02 425.25 g in 1 JAR 2 NDC:63777-214-07 1 in 1 BOX 2 113.4 g in 1 TUBE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part347 04/19/2010 Labeler - Kinray Inc. (012574513) Registrant - Dynarex Corporation (008124539) Establishment Name Address ID/FEI Business Operations Galentic Pharma India Private Limited 918531450 manufacture(63777-214) Establishment Name Address ID/FEI Business Operations Jell Pharmaceuticals Pvt. Ltd. 726025211 manufacture(63777-214)