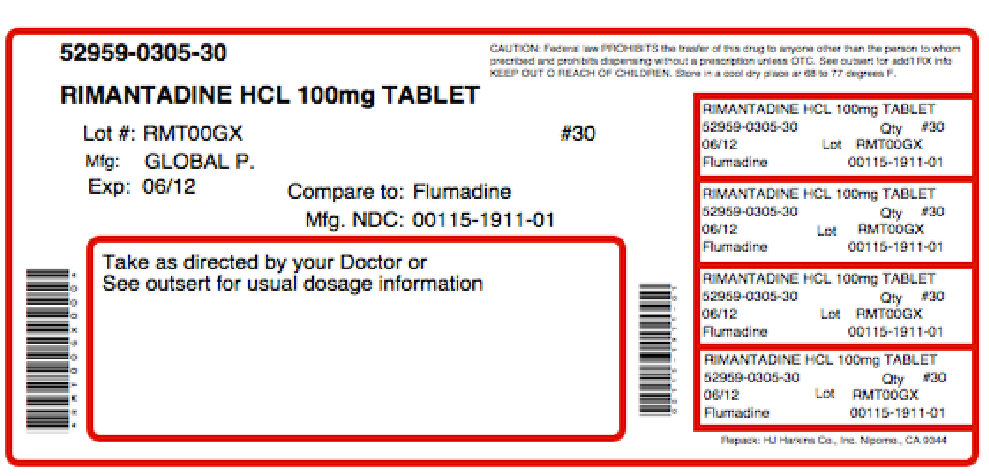
Label: RIMANTALIST- rimantadine hydrochloride, arginine kit
-
Contains inactivated NDC Code(s)
NDC Code(s): 52959-305-30, 68405-110-26 - Packager: Physician Therapeutics LLC
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated August 1, 2011
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
DESCRIPTION
Rimantadine hydrochloride is a synthetic antiviral drug available as a 100 mg film-coated tablet. Each film-coated tablet contains 100 mg of rimantadine hydrochloride. In addition, each tablet contains the following inactive ingredients: hydroxypropyl methylcellulose, magnesium stearate, microcrystalline cellulose, purified water, sodium starch glycolate and FDandC Yellow No. 6 Lake. Film coating material, Opadry (YS-1-19025-A), contains hypromellose and macrogol.
Rimantadine hydrochloride is a white to off-white crystalline powder which is freely soluble in water (50 mg/mL at 20°C). Chemically, rimantadine hydrochloride is alpha-methyltricyclo-[3.3.1.1/3.7]decane-1-methanamine hydrochloride, with an empirical formula of C12H21N•HCl, a molecular weight of 215.77 and the following structural formula: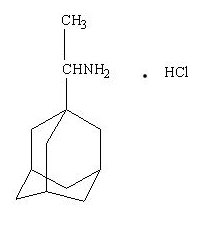
-
CLINICAL PHARMACOLOGY
CLINICAL PHARMACOLOGY
MECHANISM OF ACTION
The mechanism of action of rimantadine is not fully understood. Rimantadine appears to exert its inhibitory effect early in the viral replicative cycle, possibly inhibiting the uncoating of the virus. Genetic studies suggest that a virus protein specified by the virion M2 gene plays an important role in the susceptibility of influenza A virus to inhibition by rimantadine. -
MICROBIOLOGY
MICROBIOLOGY
Rimantadine inhibits the replication in cell culture of influenza A virus isolates from each of the three antigenic subtypes, i.e., H1N1, H2N2 and H3N2, that have been isolated from man. Rimantadine has little or no activity against influenza B virus (Ref. 1, 2). Rimantadine does not appear to interfere with the immunogenicity of inactivated influenza A vaccine.
A quantitative relationship between the susceptibility in cell culture of influenza A virus to rimantadine and clinical response to therapy has not been established.
Susceptibility test results, expressed as the concentration of the drug required to inhibit virus replication by 50% or more in a cell culture system, vary greatly (from 19 nM to 93 µM) depending upon the assay protocol used, size of the virus inoculum, isolates of the influenza A virus strains tested, and the cell types used (Ref. 2).
RESISTANCE
Influenza A virus isolates resistant to rimantadine have been selected in cell culture and in vivo as a result of treatment. Rimantadine-resistant strains of influenza A virus have emerged among freshly isolated strains in closed settings where rimantadine has been used. Resistant viruses have been shown to be transmissible and to cause typical influenza illness. (Ref. 3, 9). Substitutions at any one of five amino acid positions in the transmembrane domain of M2 confer resistance to rimantadine. The most common substitution causing resistance among influenza A (H1N1) and A (H3N2) is S31N. Other less common substitutions that cause resistance include substitutions A30F, V27A, V30A, and L26F.
Rimantadine resistance has been observed in circulating seasonal influenza and pandemic isolates from individuals who have not received rimantadine. Swine-origin influenza A (H1N1) (S-OIV) viruses that were resistant to rimantadine have been shown to contain the S31N substitution. Existing primers used for detection of adamantine resistance in seasonal viruses do not work with all tested S-OIVs (Ref. 11). The CDC should be consulted for questions regarding resistance to rimantadine in circulating influenza strains.
CROSS-RESISTANCE
Cross-resistance among the adamantanes, rimantadine and amantadine, has been observed. Resistance to rimantadine confers cross-resistance to amantadine and vice-versa. Substitutions that confer resistance to rimantadine include (most frequently) M2 S31N, as well as the less common changes V27A, V30A, L26F and A30T (Ref. 10). -
PHARMACOKINETICS
PHARMACOKINETICS
Although the pharmacokinetic profile of rimantadine hydrochloride has been described, no pharmacodynamic data establishing a correlation between plasma concentration and its antiviral effect are available.
Rimantadine hydrochloride is absorbed after oral administration. The mean ± SD peak plasma concentration after a single 100 mg dose of rimantadine hydrochloride was 74 ± 22 ng/mL (range: 45 to 138 ng/mL). The time to peak concentration was 6 ± 1 hours in healthy adults (age 20 to 44 years). The single dose elimination half-life in this population was 25.4 ± 6.3 hours (range: 13 to 65 hours). The single dose elimination half-life in a group of healthy 71 to 79 year-old subjects was 32 ± 16 hours (range: 20 to 65 hours).
After the administration of rimantadine 100 mg twice daily to healthy volunteers (age 18 to 70 years) for 10 days, area under the curve (AUC) values were approximately 30% greater than predicted from a single dose. Plasma trough levels at steady state ranged between 118 and 468 ng/mL. In these patients no age-related differences in pharmacokinetics were detected. However, in a comparison of three groups of healthy older subjects (age 50-60, 61-70 and 71-79 years), the 71 to 79 year-old group had average AUC values, peak concentrations and elimination half-life values at steady state that were 20 to 30% higher than the other two groups. Steady-state concentrations in elderly nursing home patients (age 68 to 102 years) were 2- to 4-fold higher than those seen in healthy young and elderly adults.
The pharmacokinetic profile of rimantadine in children has not been established.
Following oral administration, rimantadine is extensively metabolized in the liver with less than 25% of the dose excreted in the urine as unchanged drug. Three hydroxylated metabolites have been found in plasma. These metabolites, an additional conjugated metabolite and parent drug account for 74 ± 10% (n=4) of a single 200 mg dose of rimantadine excreted in urine over 72 hours.
In a group (n=14) of patients with chronic liver disease, the majority of whom were stabilized cirrhotics, the pharmacokinetics of rimantadine were not appreciably altered following a single 200 mg oral dose compared to six healthy subjects who were sex, age and weight matched to six of the patients with liver disease. After administration of a single 200 mg dose to patients (n=10) with severe hepatic dysfunction, AUC was approximately 3-fold larger, elimination half-life was approximately 2-fold longer and apparent clearance was about 50% lower when compared to historic data from healthy subjects.
Rimantadine pharmacokinetics were evaluated following administration of 100 mg rimantadine hydrochloride twice daily for 14 days to subjects with mild (creatine clearance [CrCl] 50-80 mL/min), moderate ([CrCl] 30-49 mL/min), and severe ([CrCl] 5-29 mL/min) renal impairment and to healthy subjects (CrCl>80 mL/min). There were no clinically relevant differences in rimantadine Cmax, Cmin, and AUC0-τ between subjects with mild or moderate renal impairment compared to healthy subjects. In subjects with severe renal impairment. Rimantadine Cmax, Cmin, and AUC0-τ on Day 14 increased by 75%, 82%, and 81%, respectively, compared to healthy subjects. The rimantadine elimination half-life was slightly prolonged (increased of 18% or less) in subjects with mild and moderate renal impairment but increased by 49% in subjects with severe renal impairment compared to healthy subjects.
After a single 200 mg oral dose of rimantadine was given to eight hemodialysis patients (CrCl 0-10 mL/min), there was a 1.6-fold increase in the elimination half-life and a 40% decrease in apparent clearance compared to age-matched healthy subjects. Hemodialysis did not contribute to the clearance of rimantadine.
The in vitro human plasma protein binding of rimantadine is about 40% over typical plasma concentrations. Albumin is the major binding protein. -
INDICATIONS & USAGE
INDICATIONS AND USAGE
Rimantadine hydrochloride tablet is indicated for the prophylaxis and treatment of illness caused by various strains of influenza A virus in adults (17 years and older).
Rimantadine hydrochloride tablet is indicated for prophylaxis against influenza A virus in children (1 year to 16 years of age).
PROPHYLAXIS
In controlled studies of children (1 year to 16 years of age), healthy adults (17 years and older), and elderly patients (65 years of age and older), rimantadine hydrochloride has been shown to be safe and effective in preventing signs and symptoms of infection caused by various strains of influenza A virus. Since rimantadine hydrochloride does not completely prevent the host immune response to influenza A infection, individuals who take this drug may still develop immune responses to natural disease or vaccination and may be protected when later exposed to antigenically-related viruses. Following vaccination during an influenza outbreak, rimantadine hydrochloride prophylaxis should be considered for the 2 to 4 week time period required to develop an antibody response. However, the safety and effectiveness of rimantadine hydrochloride prophylaxis have not been demonstrated for longer than 6 weeks.
TREATMENT
Rimantadine hydrochloride therapy should be considered for adults (17 years and older) who develop an influenza-like illness during known or suspected influenza A infection in the community. When administered within 48 hours after onset of signs and symptoms of infection caused by influenza A virus strains, rimantadine hydrochloride has been shown to reduce the duration of fever and systemic symptoms.
The following points should be considered before initiating treatment or prophylaxis with rimantadine hydrochloride:
- Rimantadine hydrochloride is not a substitute for early vaccination on an annual basis as recommended by the Centers for Disease Control and Prevention Advisory Committee on Immunization Practices
- Influenza viruses change over time. Emergence of resistance mutations could decrease drug effectiveness. Other factors (for example, changes in viral virulence) might also diminish clinical benefit of antiviral drugs. Prescribers should consider available information on influenza drug susceptibility patterns and treatment effects when deciding whether to use rimantadine hydrochloride. - CONTRAINDICATIONS
-
PRECAUTIONS
PRECAUTIONS
GENERAL
An increased incidence of seizures has been reported in patients with a history of epilepsy who received the related drug amantadine. In clinical trials of rimantadine hydrochloride, the occurrence of seizure-like activity was observed in a small number of patients with a history of seizures who were not receiving anticonvulsant medication while taking rimantadine hydrochloride. If seizures develop, rimantadine hydrochloride should be discontinued.
The safety and pharmacokinetics of rimantadine in hepatic insufficiency have only been evaluated after single dose administration. In a study of 14 subjects with chronic liver disease (mostly stabilized cirrhotics), no alterations in the pharmacokinetics were observed after the administration of a single dose of rimantadine. However, the apparent clearance of rimantadine following a single dose to 10 patients with severe liver dysfunction was 50% lower than reported for healthy subjects. Because of the potential for accumulation of rimantadine and its metabolites in plasma, caution should be exercised when patients with hepatic insufficiency are treated with rimantadine.
Following multiple-dose administration of rimantadine, there were no clinically relevant differences in rimantadine systemic exposure between subjects with mild or moderate renal impairment compared to healthy subjects. In subjects with severe renal impairment, rimantadine systemic exposure increased by 81%, compared with healthy subjects. Because of the potential for increased accumulation of rimantadine metabolites in renally impaired subjects, caution should be exercised when these patients are treated with rimantadine.
Transmission of rimantadine resistant virus should be considered when treating patients whose contacts are at high risk for influenza A illness. Influenza A virus strains resistant to rimantadine can emerge during treatment and such resistant strains have been shown to be transmissible and to cause typical influenza illness (Ref. 3). Although the frequency, rapidity, and clinical significance of the emergence of drug-resistant virus are not yet established, several small studies have demonstrated that 10% to 30% of patients with initially sensitive virus, upon treatment with rimantadine, shed rimantadine resistant virus. (Ref. 3, 4, 5, 6)
Clinical response to rimantadine, although slower in those patients who subsequently shed resistant virus, was not significantly different from those who did not shed resistant virus. (Ref. 3) No data are available in humans that address the activity or effectiveness of rimantadine therapy in subjects infected with resistant virus.
Serious bacterial infections may begin with influenza-like symptoms or may coexist with or occur as complications during the course of influenza. Rimantadine hydrochloride has not been shown to prevent such complications. -
DRUG INTERACTIONS
DRUG INTERACTIONS
Acetaminophen
Rimantadine hydrochloride, 100 mg, was given twice daily for 13 days to 12 healthy volunteers. On day 11, acetaminophen (650 mg four times daily) was started and continued for 8 days. The pharmacokinetics of rimantadine were assessed on days 11 and 13. Coadministration with acetaminophen reduced the peak concentration and AUC values for rimantadine by approximately 11%.
Aspirin
Rimantadine hydrochloride, 100 mg, was given twice daily for 13 days to 12 healthy volunteers. On day 11, aspirin (650 mg, four times daily) was started and continued for 8 days. The pharmacokinetics of rimantadine were assessed on days 11 and 13. Peak plasma concentrations and AUC of rimantadine were reduced approximately 10% in the presence of aspirin.
Cimetidine
When a single 100 mg dose of rimantadine hydrochloride was administered with steady-state cimetidine (300 mg four times a day), there was no statistically significant differences in rimantadine Cmax or AUC between rimantadine hydrochloride alone and rimantadine hydrochloride in the presence of cimetdine.
Live Attenuated Influenza Vaccine (LAIV)
The concurrent use of rimantadine hydrochloride with live attenuated intranasal influenza vaccine has not been evaluated. However, because of potential interference between these products, the live attenuated intranasal influenza vaccine should not be administered until 48 hours after cessation of rimantadine hydrochloride and rimantadine hydrochloride should be not administered until two weeks after the administration of live attenuated intranasal influenza vaccine unless medically indicated. The concern about potential interference arises principally from the potential for antiviral drugs to inhibit replication of live vaccine virus. -
CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY
CARCINOGENESIS, MUTAGENESIS, AND IMPAIRMENT OF FERTILITY
Carcinogenesis
Oral administration of rimantadine to rats for 2 years at doses up to 100 mg/kg/d [approximately 11-14 times the maximum recommended human dose (MRHD) based on AUC] showed no evidence of increased tumor incidence.
Mutagenesis
No mutagenic effects were seen when rimantadine was evaluated in several standard assays for mutagenicity.
Impairment of Fertility
A reproduction study in male and female rats did not show detectable impairment of fertility at dosages up to 60 mg/kg/day (3 times the MRHD based on mg/m2). -
PREGNANCY
PREGNANCY
Teratogenic Effects
Pregnancy Category C
There are no adequate and well-controlled studies in pregnant women. Rimantadine is reported to cross the placenta in mice. Rimantadine has been shown to be embryotoxic in rats when given at a dose of 200 mg/kg/d (11 times the MRHD based mg/m2). At this dose the embryotoxic effect consisted of increased fetal resorption in rats; this dose also produced a variety of maternal effects including ataxia, tremors, convulsions and significantly reduced weight gain. No embryotoxicity was observed when rabbits were given doses up to 50 mg/kg/d (approximately 0.1 times the MRHD based on AUC), but evidence of a developmental abnormality in the form of a chance in the ratio of fetuses with 12 or 13 ribs were noted. This ratio is normally about 50:50 in a litter but was 80:20 after rimantadine treatment. However, in a repeat embryofetal toxicity study in rabbits at doses up to 50 mg/kg/d (approximately 0.1 times the MRHD based on AUC), this abnormality was not observed.
Nonteratogenic Effects
Rimantadine was administered to pregnant rats in a peri- and postnatal reproduction toxicity study at doses of 30, 60 and 120 mg/kg/d (1.7, 3.4 and 6.8 times the MRHD based on mg/m2). Maternal toxicity during gestation was noted at the two higher doses of rimantadine, and at the highest dose, 120 mg/kg/day, there was an increase in pup mortality during the first 2 to 4 days postpartum. Decreased fertility of the F1 generation was also noted for the two higher doses.
For these reasons, rimantadine hydrochloride should be used during pregnancy only if the potential benefit justifies the risk to the fetus. -
NURSING MOTHERS
NURSING MOTHERS
Rimantadine hydrochloride should not be administered to nursing mothers because of the adverse effects noted in offspring of rats treated with rimantadine during the nursing period. Rimantadine is concentrated in rat milk in a dose-related manner: 2 to 3 hours following administration of rimantadine, rat breast milk levels were approximately twice those observed in the serum. -
PEDIATRIC USE
PEDIATRIC USE
In children (1 year to 16 years of age), rimantadine hydrochloride is recommended for the prophylaxis of influenza A. The safety and effectiveness of rimantadine hydrochloride in the treatment of symptomatic influenza infection in children (1 year to 16 years of age) have not been established. Prophylaxis studies with rimantadine hydrochloride have not been performed in children below the age of 1 year. -
ADVERSE REACTIONS
ADVERSE REACTIONS
In 1,027 patients treated with rimantadine hydrochloride in controlled clinical trials at the recommended dose of 200 mg daily, the most frequently reported adverse events involved the gastrointestinal and nervous systems.
Incidence >1%: Adverse events reported most frequently (1-3%) at the recommended dose in controlled clinical trials are shown in the table below.Less frequent adverse events (0.3 to 1%) at the recommended dose in controlled clinical trials were: Gastrointestinal System: diarrhea, dyspepsia; Nervous System: impairment of concentration, ataxia, somnolence, agitation, depression; Skin and Appendages: rash; Hearing and Vestibular: tinnitus; Respiratory: dyspnea.
Rimantadine
(n=1027)
Control
(n=986)
Nervous System
Insomnia
2.1%
0.9%
Dizziness
1.9%
1.1%
Headache
1.4%
1.3%
Nervousness
1.3%
0.6%
Fatigue
1.0%
0.9%
Gastrointestinal System
Nausea
2.8%
1.6%
Vomiting
1.7%
0.6%
Anorexia
1.6%
0.8%
Dry mouth
1.5%
0.6%
Abdominal Pain
1.4%
0.8%
Body as a Whole
Asthenia
1.4%
0.5%
Additional adverse events (less than 0.3%) reported at recommended doses in controlled clinical trials were: Nervous System: gait abnormality, euphoria, hyperkinesia, tremor, hallucination, confusion, convulsions; Respiratory: bronchospasm, cough; Cardiovascular: pallor, palpitation, hypertension, cerebrovascular disorder, cardiac failure, pedal edema, heart block, tachycardia, syncope; Reproduction: non-puerperal lactation; Special Senses: taste loss/change, parosmia.
Rates of adverse events, particularly those involving the gastrointestinal and nervous systems, increased significantly in controlled studies using higher than recommended doses of rimantadine hydrochloride. In most cases, symptoms resolved rapidly with discontinuation of treatment. In addition to the adverse events reported above, the following were also reported at higher than recommended doses: increased lacrimation, increased micturition frequency, fever, rigors, agitation, constipation, diaphoresis, dysphagia, stomatitis, hypesthesia and eye pain.
Adverse Reactions in Trials of Rimantadine and Amantadine: In a six-week prophylaxis study of 436 healthy adults comparing rimantadine with amantadine and placebo, the following adverse reactions were reported with an incidence >1%.
Rimantadine
200 mg/day
(n=145)
Placebo
(n=143)
Amantadine
200 mg/day
(n=148)
Nervous System
Insomia
3.4%
0.7%
7.0%
Nervousness
2.1%
0.7%
2.8%
Impaired
Concentration
2.1%
1.4%
2.1%
Dizziness
0.7%
0.0%
2.1%
Depression
0.7%
0.7%
3.5%
Total % of subjects of adverse reactions
6.9%
4.1%
14.7%
Total % of subjects withdrawn due to adverse
reactions
6.9%
3.4%
14.0%
-
GERIATRIC USE
GERIATRIC USE
Approximately 200 patients over the age of 64 were evaluated for safety in controlled clinical trials with rimantadine hydrochloride. Geriatric subjects who received either 200 mg or 400 mg of rimantadine daily for 1 to 50 days experienced considerably more central nervous system and gastrointestinal adverse events than comparable geriatric subjects receiving placebo. Central nervous system events including dizziness, headache, anxiety, asthenia, and fatigue, occurred up to two times more often in subjects treated with rimantadine than in those treated with placebo. Gastrointestinal symptoms, particularly nausea, vomiting, and abdominal pain occurred at least twice as frequently in subjects receiving rimantadine than in those receiving placebo. The gastrointestinal symptoms appeared to be dose related. In patients over 65, the recommended dose is 100 mg, daily (see CLINCAL PHARMACOLOGY and DOSAGE AND ADMINISTRATION). -
OVERDOSAGE
OVERDOSAGE
As with any overdose, supportive therapy should be administered as indicated. Overdoses of a related drug, amantadine, have been reported with adverse reactions consisting of agitation, hallucinations, cardiac arrhythmia and death. The administration of intravenous physostigmine (a cholinergic agent) at doses of 1 to 2 mg in adults (Ref. 7) and 0.5 mg in children (Ref. 8) repeated as needed as long as the dose did not exceed 2 mg/hour has been reported anecdotally to be beneficial in patients with central nervous system effects from overdoses of amantadine. -
DOSAGE & ADMINISTRATION
DOSAGE AND ADMINISTRATION
FOR PROPHYLAXIS IN ADULTS AND CHILDREN
Adults (17 years and older)
The recommended adult dose of rimantadine hydrochloride is 100 mg twice a day. Study durations ranged from 11 days to 6 weeks in adult and elderly patients. In patients with severe hepatic dysfunction, severe renal impairment (CrCl 5 to 29 mL/min) or renal failure (CrCl ≤ 10 mL/min) and in elderly nursing home patients, a dose reduction to 100 mg daily is recommended. Because of the potential for accumulation of rimantadine metabolites during multiple dosing, patients with hepatic or renal impairment should be monitored for adverse effects.
Children (1 year to 16 years of age)
- Study duration ranged from 5 weeks to 6 weeks in pediatric subjects.
- In children 1 year to 9 years of age, rimantadine hydrochloride should be administered once a day, at a dose of 5 mg/kg but not exceeding 150 mg.
- For children 10 years of age or older, use the adult dose.
(see Directions for Compounding of an Oral Suspension from Rimantadine Hydrochloride Tablets to prepare an oral suspension for administration to children and patients with difficulty swallowing tablets).
Children (Birth to 11 months)
The safety and efficacy of rimantadine hydrochloride for prophylaxis of influenza in pediatric patients younger than 1 year of age have not been established.
FOR TREATMENT IN ADULTS
Adults (17 years and older)
The recommended adult dose of rimantadine hydrochloride is 100 mg twice a day for 7 days. In patients with severe hepatic dysfunction, severe renal impairment (CrCl 5 to 29 mL/min) or renal failure (CrCl ≤ 10 mL/min) and elderly nursing home patients, a dose reduction to 100 mg daily is recommended. Because of the potential for accumulation of rimantadine metabolites during multiple dosing, patients with hepatic or renal impairment should be monitored for adverse effects. Rimantadine hydrochloride therapy should be initiated as soon as possible, preferably within 48 hours after onset of signs and symptoms of influenza A infection. Therapy should be continued for approximately seven days from the initial onset of symptoms.
Children (16 years of age and younger)
Rimantadine hydrochloride is not indicated for treatment of influenza in pediatric patients 16 years or younger.
Directions for the Compounding of an Oral Suspension from Rimantadine Hydrochloride Tablets (Final Concentration = 10 mg/mL)1
These directions are provided for use only during emergency situations, for patients who have difficulty swallowing tablets or where lower doses are needed. The pharmacist may compound a suspension (10 mg/mL) from rimantadine hydrochloride tablets, 100 mg using Ora-Sweet®.1 Other vehicles have not been studied.
To make an oral suspension (10 mg/mL) from 100 mg rimantadine hydrochloride tablets, you will need the following:
- 100 mg tablets of rimantadine hydrochloride
- Ora-Sweet® (a vehicle manufactured by Paddock Laboratories)
- a graduated cylinder
- a mortar and pestle
- an Amber Glass or Polyethylene terephthalate plastic (PET) bottle
- a funnel (optional)
1
Ora-Sweet® is a registered trademark of Paddock Laboratories
Compounding Procedures
A 100 mg tablet of rimantadine hydrochloride is required for each 10 mL of compounded oral suspension to make a concentration of 10 mg/mL.
A compounded oral suspension is stable for 14 days. Therefore, the maximum amount of oral suspension that can be dispensed to a patient should not exceed a 14 day supply.
Step A: Guidance for how to determine the Number of Tablets and Total Volume needed to compound a 10 mg/mL oral suspension for each patient
1. Verify the prescribed dose is correct.
2. Calculate the mg amount of rimantadine hydrochloride needed for the duration of therapy.
(Daily Dose) × (Number of days) = (mg of rimantadine hydrochloride)
For example, 75 mg/day × 10 days = 750 mg
3. Round up the mg of rimantadine hydrochloride amount to the next 100 mg designation.
For example, Round up 750 mg to 800 mg
4. Calculate the Number of 100 mg tablets that are required for the compounded oral suspension.
(Rounded mg of rimantadine hydrochloride) ÷ (100 mg/tablet) = (Number of tablets)
For example, 800 mg ÷100 mg/tablet = 8 tablets
5. Calculate the Total Volume of compounded oral suspension (10 mg/mL) (Rounded mg of rimantadine hydrochloride) ÷ (10 mg/mL) = (Total Volume)
For example, 800 mg ÷ 10 mg/mL = 80 mL
Step B: Once the total Number of Tablets and Volume are determined then follow the procedures below for compounding the oral suspension (10 mg/mL) from rimantadine hydrochloride tablets 100 mg
Verify your calculations before you begin to compound an oral suspension.
A 100 mg tablet of rimantadine hydrochloride is required for each 10 mL's of compounded oral suspension to make a concentration of 10 mg/mL.
1. Place the required number of rimantadine hydrochloride 100 mg tablets into a clean mortar of sufficient size to contain the tablets and volume of vehicle, Ora-Sweet® used in Step 3.
2. Grind the tablets and triturate to a fine powder using a pestle. Powder on the sides of the mortar or pestle should be removed using a spatula and incorporated into the trituration throughout the process.
3. Slowly add approximately one-third (1/3) of the total volume of vehicle to the mortar while triturating until a uniform suspension is achieved.
4. Transfer the suspension to an amber glass or a PET plastic bottle. Other types of bottles, such as non-PET plastic or uncolored bottles, have not been evaluated and should not be used. A funnel may be used to eliminate any spillage.
5. Slowly add the second one-third (1/3) of the total volume of vehicle to the mortar, rinse the pestle and mortar by a triturating motion and transfer the contents into the bottle.
6. Repeat the rinsing (Step 5) with the remaining one-third (1/3) of the vehicle, transferring the remaining contents to the fullest extent possible. Verify that the suspension is at the desired total volume or add additional vehicle if needed.
7. Close the bottle using a child-resistant cap.
8. Shake well to ensure homogeneous suspension. (Note: The active drug, rimantadine HCl readily dissolves in the specified vehicle. The suspension is caused by some of the inert ingredients of rimantadine hydrochloride tablets 100 mg which are insoluble in this vehicle.)
Labeling and Dispensing Information for the Compounded Oral Suspension
1. Include an ancillary label on the bottle indicating "Shake Gently Before Use." This compounded suspension should be gently shaken prior to administration to minimize the tendency for air entrapment with the Ora-Sweet® preparation. The need to shake the compounded oral suspension gently prior to administration should be reviewed with the parent or guardian when the suspension is dispensed.
2. Provide an oral dosing device (a graduated oral syringe or spoon) that will measure the prescribed dose (in mL). If possible, mark or highlight the graduation corresponding to the appropriate dose on the oral syringe or spoon for each patient.
3. Include an Expiration Date label according to storage condition (see below) and a "Discard any Unused Portion" label to the bottle. Instruct the parent or guardian that any remaining material following completion of therapy or after the expiration date on the label must be discarded.
STORAGE OF THE PHARMACY-COMPOUNDED SUSPENSION
Room Temperature: Stable for 14 days when stored in ambient room temperature conditions. Other storage conditions have not been studied.
Note: The storage conditions are based on stability studies of compounded oral suspensions, using the above mentioned vehicle, which was placed in amber glass and PET plastic bottles at 25°C (77°F). Stability studies have not been conducted with other vehicles or bottle types. - HOW SUPPLIED
- STORAGE AND HANDLING
-
REFERENCES
REFERENCES
1. Belshe RB, Burk B, Newman F, et al. J. Infect. Dis. 1989; 159(3):430-435.
2. Sim IS, Cerruti RL, Connell EV. J. Resp. Dis. 1989(Suppl): S46-S51.
3. Hayden FG, Belshe RB, Clover RD, et al. N.Engl. J. Med. 1989; 321(25), 1696-1702.
4. Hall CB, Dolin R, Gala CL, et al. Pediatrics 1987; 80(2): 275-282.
5. Thompson J, Fleet W, Lawrence E et al. J.Med. Virol. 1987; 21(3): 249-255.
6. Belshe RB, Smith MH, Hall CB, et al. J. Virol.1988; 62(5): 1508-1512.
7. Casey DE. N Engl J Med. 1978; 298(9):516.
8. Berkowitz CD. J. Pediatri. 1979; 95(1): 144-145.
9. Hayden FG, Sperber SJ, Belshe RB, et al. Antimicrob Agents Chemother. 1991; 35(9): 1741-1747
10. Deyde VM, Xu X, Bright RA, et al. J Infect Dis. 2007; 196(2): 249-257.
11. CDC MMWR Morb Mortal Wkly Rep. 2009; 58(16): 433-435.
Dist. by:
Global Pharmaceuticals
Division of IMPAX Laboratories, Inc.
Philadelphia, PA 19124 USA
Rev. 04/2010
224-03 -
PRINCIPAL DISPLAY PANEL
GLOBAL NDC 0115-1911-01 Rimantadine Hydrochloride Tablets 100 mg Rx only 100 Tablets USUAL DOSAGE: See accompanying outsert for complete prescribing information.
Each tablet contains 100 mg rimantadine HCI. This is bulk package. Dispense in a tightly-closed, light-resistant container as defined in the USP, with a child-resistant closure, as required.
Store at 20°C to 25°C (68° to 77°F) [see USP Controlled Room Temperature] Dist. by: Global Pharmaceuticals Division of IMPAX Laboratories, Inc. Philadelphia, PA 19124 USA Product of Taiwan Iss. 12/2009 1112-01 N3 0115-1911-01 7 Lot: Exp.:
-
SPL UNCLASSIFIED SECTION
Lister-V™ PRODUCT INFORMATION Lister-V (U.S. patent pending) capsules by oral administration. A specially formulated Medical Food product, consisting of a proprietary blend of amino acids and polyphenol ingredients in specific proportions, for the nutritional management of the metabolic processes associated with viral infection. Must be administered under physician supervision. Medical Foods Medical Food products are often used in hospitals (e.g., for burn victims or kidney dialysis patients) and outside of a hospita setting under a physician’s care for the dietary management of diseases in patients with particular medical or metabolic needs due to their disease or condition. Congress defined "Medical Food" in the Orphan Drug Act and Amendments of 1988 as "a system which is formulated to be consumed or administered enterally [or orally] under the supervision of a physician and which i intended for the specific dietary management of a disease or condition for which distinctive nutritional requirements, based on recognized scientific principles, are established by medical evaluation." Medical Foods are complex formulated products, requiring sophisticated and exacting technology. Lister-V has been developed, manufactured, and labeled in accordance with both the statutory and the FDA regulatory definition of a Medical Food. Lister-V must be used while the patient is under the ongoing care of a physician. DISORDERS OF IMMUNE RESPONSE Disorders of Immune Response as a Metabolic Deficiency Disease A critical component of the definition of a Medical Food is the requirement for a distinctive nutritional deficiency. FDA scientists have proposed a physiologic definition of a distinctive nutritional deficiency as follows: “the dietary management of patients with specific diseases requires, in some instances, the ability to meet nutritional requirements that differ substantially from the needs of healthy persons. For example, in establishing the recommended dietary allowances for general, healthy population, the Food and Nutrition Board of the Institute of Medicine National Academy of Sciences, recognized that different or distinctive physiologic requirements may exist for certain persons with "special nutritional needs arising from metabolic disorders, chronic diseases, injuries, premature birth, other medical conditions and drug therapies. Thus, the distinctive nutritional needs associated with a disease reflect the total amount needed by a healthy person to support life or maintain homeostasis, adjusted for the distinctive changes in the nutritional needs of the patient as a result of the effects of the disease process on absorption, metabolism and excretion.” It was also proposed that in patients with certain disease states who respond to nutritional therapies, a physiologic deficiency of the nutrient is assumed to exist. For example, if a patient with symptoms of influenza responds to arginine as a precursor to nitric oxide, by decreasing the severity and/or duration of symptoms, a deficiency of argine is assumed to exist. Patients with a compromised immune system are known to have nutritional deficiencies of arginine, choline, flavonoids, and certain antioxidants. Lister-V is a specially formulated Medical Food product designed to enhance immune function, activating the white blood cells to seek out and destroy viruses when they invade the body. Arginine serves as the substrate for the nitric oxide synthase enzyme, which catalyzes the oxidation of arginine to produce citrulline and nitric oxide (NO). Nitric oxide is a potent inhibitor of virus growth, particularly viruses that are responsible for the common cold including rhinoviruses, adenoviruses, and corona viruses. Delivery of nitric oxide to viruses in the nasal passages and lungs is likely to inhibit their growth thereby decreasing the duration of a common cold and certain types of virus induced illnesses. Arginine and choline, precursors of nitric oxide and acetylcholine, combined with food-based potentiators, will produce nitric oxide in nasal passages. Echinacea increases white blood cell activity in the nasal passages and the lungs. The combination of nitric oxide production, acetylcholine production, and echinacea induced white blood cell increase, will reduce the duration and symptoms of a common cold. Echinacea extracts have been shown to stimulate the growth and activity of cells of the immue system (macrophages, natural killer cells, T-cells). Such activation of protective mechanisms is thought to increase the body’s defenses to infection by viruses. Flavonoids are thought to be the primary immunostimulatory constituents in the various echinacea species. Grape seeds are a potent source of proanthocyanidins, a powerful antioxidant. The complex metabolic processes that are responsible for optimal functioning of the immune system involve the coordinated efforts of several types of white blood cells.
-
DESCRIPTION
PRODUCT DESCRIPTION Primary Ingredients Lister-V consists of a proprietary blend of amino acids, cocoa, caffeine, cinnamon, and flavonoids in specific proportions. These ingredients fall into the category of “Generally Regarded as Safe” (GRAS) as defined by the Food and Drug Administration (FDA) (Sections 201(s) and 409 of the Federal Food, Drug, and Cosmetic Act). A GRAS substance is distinguished from a food additive on the basis of the common knowledge about the safety of the substance for its intended use. The standard for an ingredient to achieve GRAS status requires not only technical demonstration of non-toxicity and safety, but also general recognition of safety through widespread usage and agreement of that safety by experts in the field. Many ingredients have been determined by the U.S. Food and Drug Administration (FDA) to be GRAS, and are listed as such by regulation, in Volume 21 Code of Federal Regulations (CFR) Sections 182, 184, and 186. Amino Acids Amino Acids are the building blocks of protein. All amino acids are GRAS listed as they have been ingested by humans for thousands of years. The doses of the amino acids in Lister-V are equivalent to those found in the usual human diet; however the formulation uses specific ratios of the key ingredients to elicit a therapeutic response. Patients with a viral disease may require an increased amount of certain amino acids that cannot be obtained from normal diet alone. Choline, for example, is an obligatory amino acid. The body cannot make choline and must obtain choline from the diet. Choline is required to fully potentiate nitric oxide synthesis in the nasal passage and lungs. A deficiency of choline leads to reduced nitric oxide production in the nasal passage and lungs. Flavonoids potentiate the production of nitric oxide in the nasal passage and lungs. Choline deficient diets and diets deficient in flavonoid rich foods can result in inadequate flavonoid concentrations, impeding nitric oxide production. Provision of arginine, choline, and flavonoids with antioxidants, in specific proportions can restore the production of beneficial nitric oxide, thereby inhibiting viral growth. Flavonoids Flavonoids are a group of phytochemical compounds found in all vascular plants including fruits and vegetables. They are a part of a larger class of compounds known as polyphenols. Many of the therapeutic or health benefits of colored fruits and vegetables, cocoa, red wine, and green tea are directly related to their flavonoid content. The specially formulated flavonoids found in Lister-V cannot be obtained from conventional foods in the necessary proportions to elicit a therapeutic response. Physical Description Lister-V is a yellow to light brown powder. Lister-V contains L-Arginine, L-Glutamic Acid, Lecithin, Echinacea Angustifolia, Green Tea Extract, Licorice Extract, Cinnamon, Cocoa, Hydrolyzed Whey Protein, Caffeine, Ginkgo Biloba, and Grape Seed Extract. Other Ingredients Lister-V contains the following inactive or other ingredients, as fillrs, excipients, and colorings: magnesium stearate, microcrystalline cellulose, Maltodextrin NF, gelatin (as the capsule material).
-
CLINICAL PHARMACOLOGY
CLINICAL PHARMACOLOGY Mechanism of Action Lister-V acts by activating white blood cell production of several types of white blood cells to combat and destroy viruses when they invade the body. Lister-V promotes nitric oxide production to inhibit viral replication in the nasal passages and the lungs. Metabolism The amino acids in Lister-V are primarily absorbed by the stomach and small intestines. All cells metabolize the amino acids in Lister-V. Circulating arginine and choline blood levels determine the production of nitric oxide and acetylcholine. Excretion Lister-V is not an inhibitor of cytochrome P450 1A2, 2C9, 2C19, 2D6, or 3A4. These isoenzymes are principally responsible for 95% of all detoxification of drugs, with CYP3A4 being responsible for detoxification of roughly 50% of drugs. Amino acids do not appear to have an effect on drug metabolizing enzymes.
- INDICATIONS & USAGE
-
CLINICAL STUDIES
CLINICAL EXPERIENCE Administration of Lister-V has demonstrated significant functional improvements of influenza symptoms when used for the nutritional management of the metabolic processes associated with certain viral infections. The administration of Lister-V results in increased production of white blood cells and a reduction in the severity and duration of symptoms associated with viral infections.
- CONTRAINDICATIONS
-
ADVERSE REACTIONS
ADVERSE REACTIONS Oral supplementation with L-arginine at high doses up to 15 grams daily is generally well tolerated. The most common adverse reactions of higher doses — from 15 to 30 grams daily — are nausea, abdominal cramps, and diarrhea. Some patients may experience these symptoms at lower doses. The total combined amount of amino acids in each Lister-V capsule does not exceed 400 mg.
- DRUG INTERACTIONS
-
OVERDOSAGE
OVERDOSE There is a negligible risk of overdose with Lister-V as the total dosage of amino acids in a one month supply (60 capsules) is less than 25 grams. Overdose symptoms may include diarrhea, weakness, and nausea. POST-MARKETING SURVEILLANCE Post-marketing surveillance has shown no serious of adverse reactions. Reported cases of mild rash and itching may have been associated with allergies to Lister-V flavonoid ingredients, including cinnamon, cocoa, and chocolate. The reactions were transient in nature and subsided within 24 hours.
-
DOSAGE & ADMINISTRATION
DOSAGE AND ADMINISTRATION Recommended Administration For the nutritional management of the metabolic processes associated with viral infections. Lister-V can be administered to ameliorate the symptoms and shorten the duration of a viral infection. At the first signs of viral infection take (3) capsules four times a day for five days. Lister-V may be used to enhance immune function after exposure to a viral disease. Lister-V may be taken by those susceptible to upper respiratory infections during airline travel. As with most amino acid formulations Lister-V should be taken without food to increase the absorption of key ingredients.
-
HOW SUPPLIED
HOW SUPPLIED Lister-V is supplied in opaque white, size 0 capsules in bottles of 60 capsules. PHYSICIAN SUPERVISION Lister-V is a Medical Food product available by prescription only and must be used while the patient is under ongoing physician supervision. Lister-V is supplied to physicians in a recyclable plastic bottle with a child-resistant cap. U.S. patent pending. Manufactured by Arizona Nutritional Supplements, Inc. Chandler AZ 85225 Distributed by Physician Therapeutics LLC, Los Angeles, CA 90077. www.ptlcentral.com Copyright 2003-2006, Physician Therapeutics LLC, all rights reserved NDC # 68405-1010-02
- STORAGE AND HANDLING
-
PRINCIPAL DISPLAY PANEL
PHYSICIAN THERAPEUTICS LISTER-V Medical Food Rx only 60 Capsules Directions for use: Must be administered under physician supervision. For adults only. As a Medical Food, take two (2) capsules four times daily in between meals or as directed by physician. For the dietary management of viral infection. Contains no added sugar, starch, wheat, yeast, preservatives, or flavor. Storage: Keep tightly closed in a cool dry place 8-320 C (45-900F), relative humidity, below 50%. Warning: Keep this product out of the reach of children. NDC# 68405-1010-02 Ingredients: Each serving (per 2 capsules) contains: Proprietary Amino Acid Blend L-Histadine, L-Arginine, L-Leucine, L-Glutarmine, Hydrolyzed Whey Protein Lecithin Cinnamon Echinacea Grape Seed Extract (80% polyphenols) Green Tea Extract (caffeine) L-Cysteine Cocoa (6% Theobromine) (fruit) Other indgredients: Gelatin,, cellulose, colloidal, silicon dioxide, titanium dioxide. Distributed exclusive by: A Division of Targeted Medical Pharma, Inc Los Angeles, CA 90077 www.ptlcentral.com Patent Pending
-
NONCLINICAL TOXICOLOGY
For the Dietary Management of Viral Infection. Three capsules four times daily or as directed by physician. See product label and insert. Lister-V Medical Food A Convenience Packed Medical Food and Drug Rimantalist PHYSICIAN THERAPEUTICS - Lister-V 60 Capsules - Rimantadine 100 mg 30 Tablets Rx Only No Refills Without Physician Authorization NDC# 68405-8110-26 Of this co-pack. FRONT VIEW As prescribed by physician. See product label and product information insert. Rimantadine 100 mg Rx Drug 68405-8110-26
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
RIMANTALIST
rimantadine hydrochloride, arginine kitProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:68405-110 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68405-110-26 1 in 1 KIT Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 BOTTLE 30 Part 2 1 BOTTLE 60 Part 1 of 2 RIMANTADINE HYDROCHLORIDE
rimantadine hydrochloride tabletProduct Information Item Code (Source) NDC:52959-305(NDC:0115-1911) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength RIMANTADINE HYDROCHLORIDE (UNII: JEI07OOS8Y) (RIMANTADINE - UNII:0T2EF4JQTU) RIMANTADINE HYDROCHLORIDE 100 mg Inactive Ingredients Ingredient Name Strength MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) WATER (UNII: 059QF0KO0R) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) HYPROMELLOSES (UNII: 3NXW29V3WO) POLYETHYLENE GLYCOL (UNII: 3WJQ0SDW1A) Product Characteristics Color orange (ORANGE) Score no score Shape OVAL (convex-faced) Size 11mm Flavor Imprint Code G;1911 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:52959-305-30 30 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA076132 07/07/2011 Part 2 of 2 LISTER-V
arginine capsuleProduct Information Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ARGININE (UNII: 94ZLA3W45F) (ARGININE - UNII:94ZLA3W45F) ARGININE 60 mg Inactive Ingredients Ingredient Name Strength MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) MALTODEXTRIN (UNII: 7CVR7L4A2D) GELATIN (UNII: 2G86QN327L) Product Characteristics Color white (WHITE) Score no score Shape CAPSULE Size 21mm Flavor Imprint Code ; Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 60 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Medical Food 07/07/2011 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 07/07/2011 Labeler - Physician Therapeutics LLC (931940964) Establishment Name Address ID/FEI Business Operations Impax Laboratories Taiwan Inc. 656139511 manufacture Establishment Name Address ID/FEI Business Operations H.J. Harkins Company, Inc. 147681894 repack Establishment Name Address ID/FEI Business Operations Targeted Medical Pharma, Inc. 126962740 manufacture