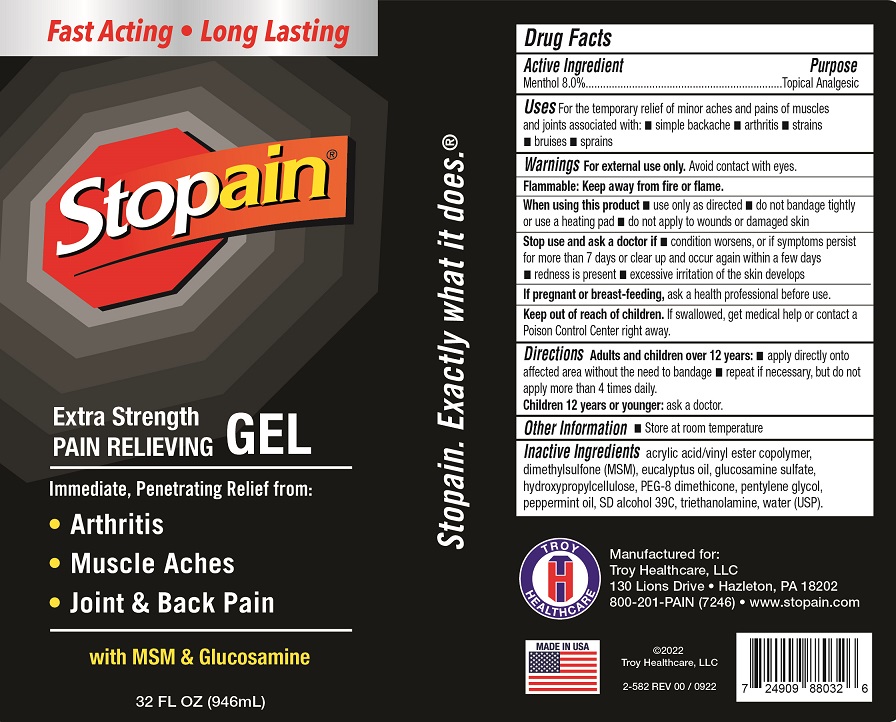
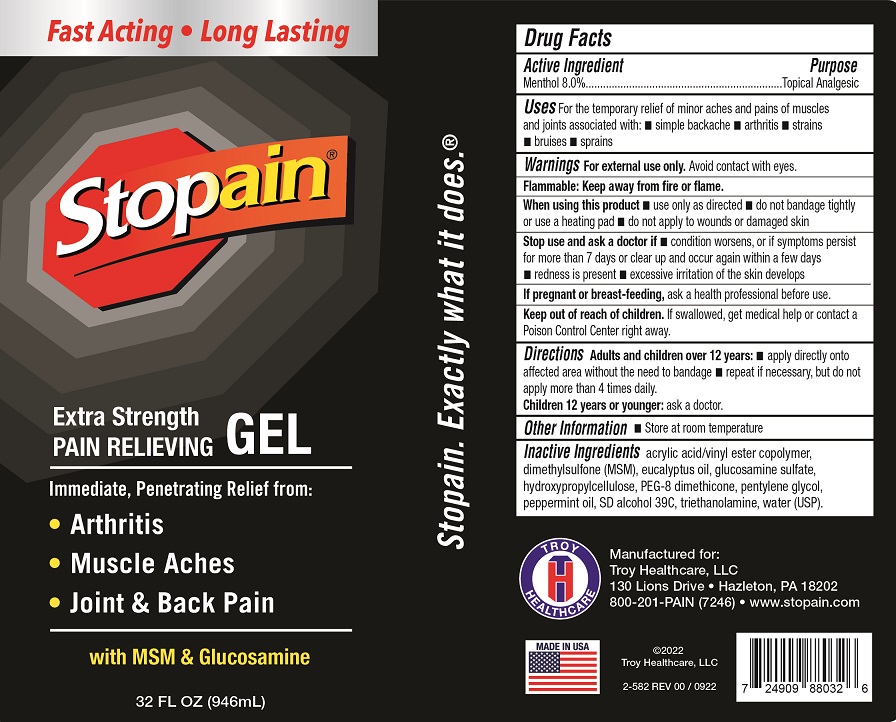
Label: STOPAIN EXTRA STRENGTH GEL- menthol gel
- NDC Code(s): 63936-8800-1, 63936-8800-2, 63936-8800-3, 63936-8800-4
- Packager: Troy Manufacturing, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated November 3, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active Ingredient
- Uses
-
Warnings
For external use only.Avoid contact with eyes
Flammable: Keep away from fire or flame.
When using this product
• use only as directed • do not bandage tightly or use a heating pad • do not apply to wounds or damaged skin
- Directions
- Other Information
- Inactive Ingredients
- Package Labeling:63936-8800-3
- Package Labeling:63936-8800-2
- Package Labeling:63936-8800-1
- Package Labeling:63936-8800-4
-
INGREDIENTS AND APPEARANCE
STOPAIN EXTRA STRENGTH GEL
menthol gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:63936-8800 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 80 mg in 1 mL Inactive Ingredients Ingredient Name Strength DIMETHYL SULFONE (UNII: 9H4PO4Z4FT) EUCALYPTUS OIL (UNII: 2R04ONI662) GLUCOSAMINE SULFATE (UNII: 1FW7WLR731) HYDROXYPROPYL CELLULOSE, UNSPECIFIED (UNII: 9XZ8H6N6OH) PEG-8 DIMETHICONE (UNII: GIA7T764OD) PENTYLENE GLYCOL (UNII: 50C1307PZG) PEPPERMINT OIL (UNII: AV092KU4JH) TROLAMINE (UNII: 9O3K93S3TK) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63936-8800-3 473 mL in 1 BOTTLE; Type 0: Not a Combination Product 10/10/2022 2 NDC:63936-8800-2 946 mL in 1 BOTTLE; Type 0: Not a Combination Product 10/10/2022 3 NDC:63936-8800-1 3785 mL in 1 BOTTLE; Type 0: Not a Combination Product 10/10/2022 4 NDC:63936-8800-4 88 mL in 1 TUBE; Type 0: Not a Combination Product 10/01/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 10/10/2022 Labeler - Troy Manufacturing, Inc. (160075248)