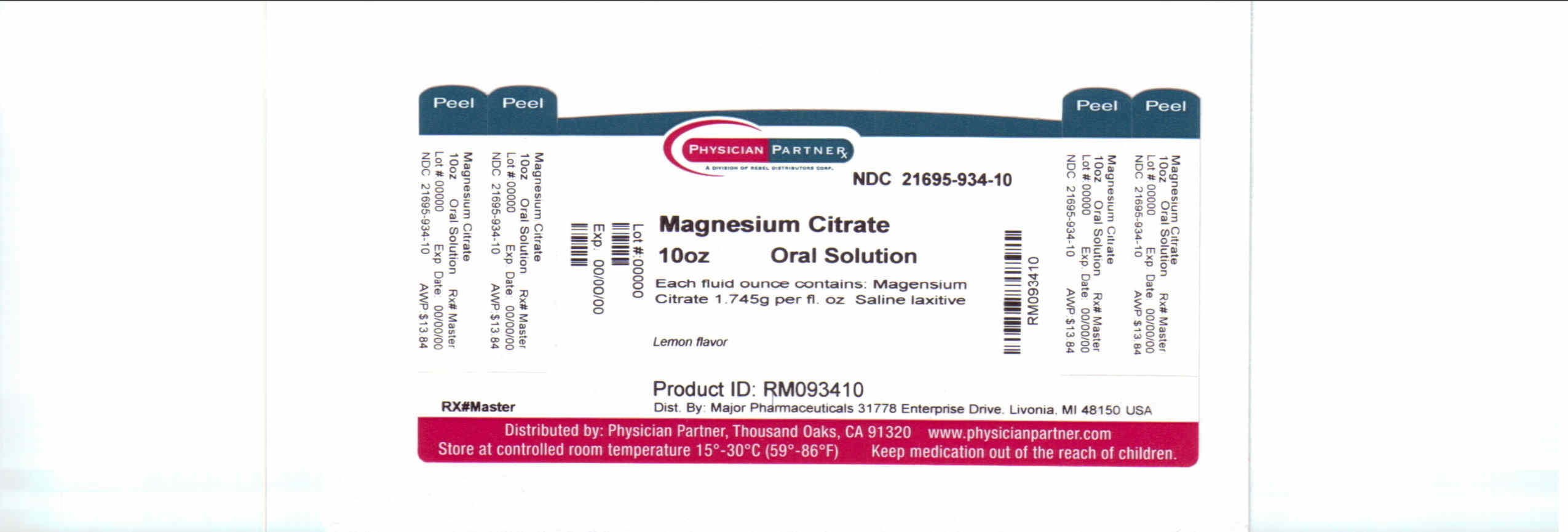
Label: MAGNESIUM CITRATE liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 21695-934-10 - Packager: Rebel Distributors Corp
- This is a repackaged label.
- Source NDC Code(s): 0904-0304
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated April 28, 2011
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
- Warnings
- Ask a doctor or pharmacist before use if you are
- when using this product
- Stop use and ask a doctor if
- If pregnant or breast feeding
- keep this and all drugs out of the reach of children.
-
Directions
Adults and Children 12 years of age and older 1/2 to 1 bottle (10 fl oz). Drink a full 8 oz glass of liquid with each dose. The dose may be taken as a single daily dose or in divided doses.
children 6 to under 12 years of age 1/3 to 1/2 bottle with a full 8 oz glass of liquid.
children under 6 years of age consult a doctor
- Other Information
- Inactive Ingredients
- Questions or Comments?
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
MAGNESIUM CITRATE
magnesium citrate liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:21695-934(NDC:0904-0304) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MAGNESIUM CITRATE (UNII: RHO26O1T9V) (Magnesium Cation - UNII:T6V3LHY838) MAGNESIUM CITRATE 2.62 g in 100 mL Inactive Ingredients Ingredient Name Strength water (UNII: 059QF0KO0R) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) SACCHARIN SODIUM ANHYDROUS (UNII: I4807BK602) POTASSIUM BICARBONATE (UNII: HM5Z15LEBN) Product Characteristics Color Score Shape Size Flavor LEMON (Lemon) Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:21695-934-10 296 mL in 1 BOTTLE, GLASS Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part334 07/31/2009 Labeler - Rebel Distributors Corp (118802834) Establishment Name Address ID/FEI Business Operations Rebel Distributors Corp 118802834 RELABEL, REPACK