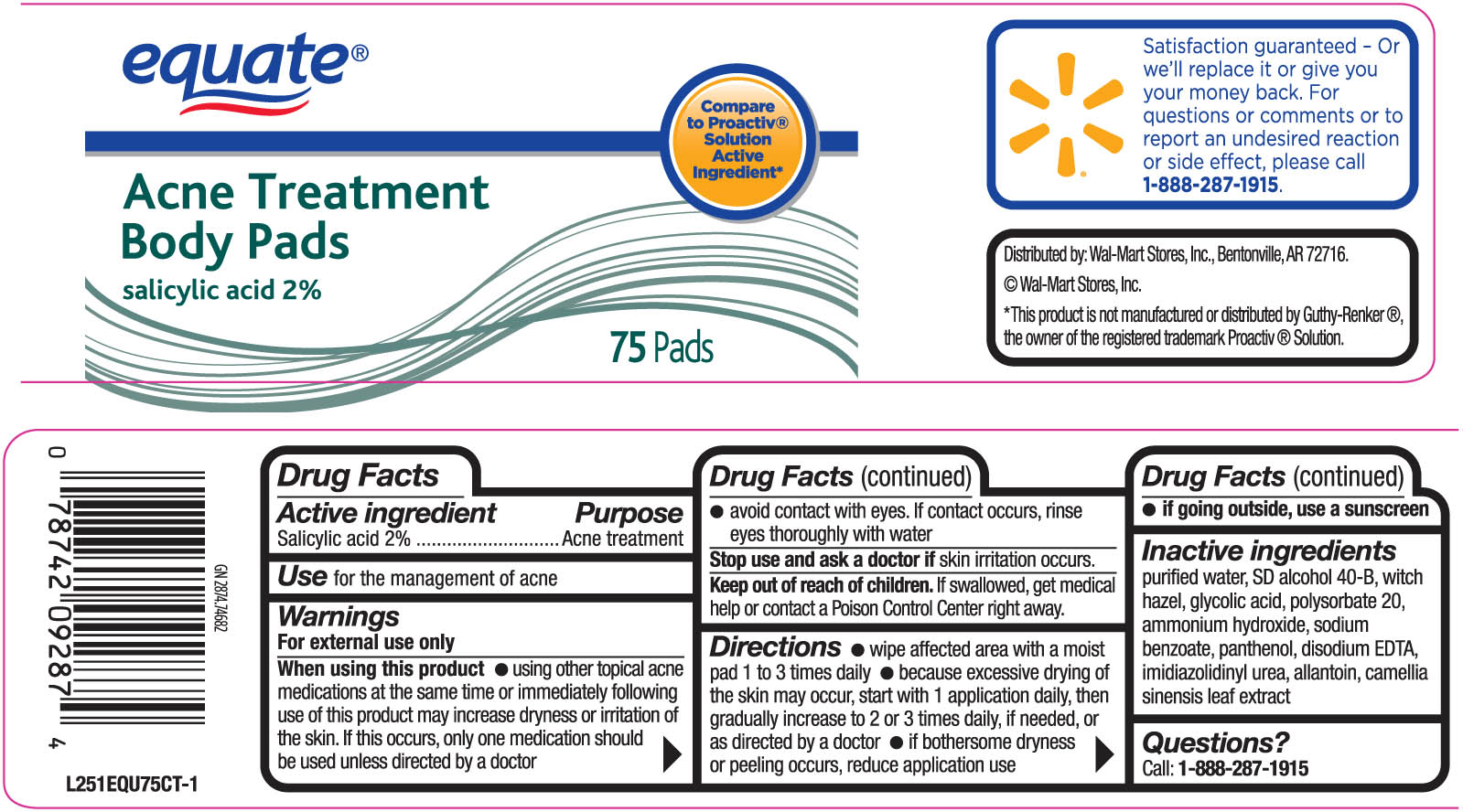
Label: EQUATE ACNE TREATMENT BODY PADS- salicylic acid swab
-
Contains inactivated NDC Code(s)
NDC Code(s): 49035-024-33 - Packager: Wal-Mart Stores Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated July 13, 2010
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
-
INDICATIONS & USAGE
Directions
- wipe affected area with a moist pad 1 to 3 times daily
- because excessive drying of the skin may occur, start with one application daily, then gradually increase to 2 or 3 times daily, if needed or as directed by a doctor.
- if bothersome drying or peeling occurs, reduce application use.
If going outside, use a sunscreen
Questions?
Call 1-888-287-1915
-
WARNINGS
Warnings
For external use only.
When using this product
- using other topical acne medications at the same time or immediately following use of this product may
increase dryness or irritation of the skin. If this occurs, only one medication should be used unless directed by a doctor.
- avoid contact with the eyes. If contact occurs, rinse eyes thoroughly with water.
Stop use and ask a doctor if skin irritation occurs.
Keep out of reach of children. If swallowed, get medical help or contact a Poison
Control Center right away.
-
DOSAGE & ADMINISTRATION
Directions
- wipe affected area with a moist pad 1 to 3 times daily
- because excessive drying of the skin may occur, start with one application daily, then gradually increase to 2 or 3 times daily, if needed or as directed by a doctor.
- if bothersome drying or peeling occurs, reduce application use.
If going outside, use a sunscreen
Questions?
Call 1-888-287-1915
- INACTIVE INGREDIENT
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
EQUATE ACNE TREATMENT BODY PADS
salicylic acid swabProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49035-024 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 20 mg Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) ALCOHOL (UNII: 3K9958V90M) ALLANTOIN (UNII: 344S277G0Z) GREEN TEA LEAF (UNII: W2ZU1RY8B0) EDETATE DISODIUM (UNII: 7FLD91C86K) GLYCOLIC ACID (UNII: 0WT12SX38S) WITCH HAZEL (UNII: 101I4J0U34) PANTHENOL (UNII: WV9CM0O67Z) POLYSORBATE 20 (UNII: 7T1F30V5YH) SODIUM BENZOATE (UNII: OJ245FE5EU) DIAZOLIDINYL UREA (UNII: H5RIZ3MPW4) AMMONIA (UNII: 5138Q19F1X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49035-024-33 75 in 1 JAR Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333D 07/13/2011 Labeler - Wal-Mart Stores Inc (051957769) Registrant - Pharma Pac, LLC (140807475) Establishment Name Address ID/FEI Business Operations Pharma Pac, LLC 140807475 manufacture