Label: LIDOCAINE- lidocaine hydrochloride and epinephrine injection, solution
- NDC Code(s): 0362-0898-05
- Packager: Septodont, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated February 8, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION
Sterile isotonic solutions containing a local anesthetic agent, Lidocaine Hydrochloride, and a vasoconstrictor, epinephrine (as bitartrate) and are administered parenterally by injection. Both solutions are available in single dose cartridges of 1.7 mL (See INDICATIONS AND USAGE for specific uses).
Solutions contain lidocaine hydrochloride which is chemically designated as acetamide,
2-(diethylamino)-N-(2,6-dimethylphenyl)-monohydrochloride, and has the following structural formula: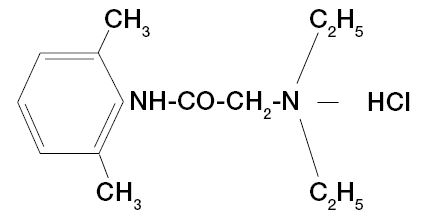 C14H22N20 • HCl • H20 M.W. 288.8
C14H22N20 • HCl • H20 M.W. 288.8Epinephrine is ( - )-3, 4-Dihydroxy-α-[(Methylamino) methyl] benzyl alcohol and has the following structural formula:
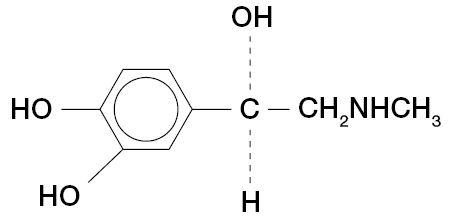 C9H13N03 M.W. 183.21
C9H13N03 M.W. 183.21COMPOSITION OF LIDOCAINE HYDROCHLORIDE AND EPINEPHRINE INJECTION, USP BRAND NAME PRODUCT IDENTIFICATION FORMULA SINGLE DOSE CARTRIDGE Lidocaine
hydrochlorideEpinephrine
(as the bitartrate)Sodium
ChloridePotassium
metabisulfiteEdetate
DisodiumConcentration % Dilution (mg/mL) (mg/mL) (mg/mL) LIDOCAINE HCl 2%
and EPINEPHRINE 1:50,0002 1:50,000 6.5 1.2 0.25 LIDOCAINE HCl 2%
and EPINEPHRINE 1:100,0002 1:100,000 6.5 1.2 0.25 The pH of the lidocaine hydrochloride and epinephrine injection, USP solutions are adjusted to USP limits with sodium hydroxide. -
CLINICAL PHARMACOLOGY
Mechanism of action
Lidocaine stabilizes the neuronal membrane by inhibiting the ionic fluxes required for the initiation and conduction of nerve impulses, thereby effecting local anesthetic action.
Onset and duration of anesthesia
When used for infiltration anesthesia in dental patients, the time of onset averages less than two minutes for each of the two forms of lidocaine hydrochloride and epinephrine injection, USP. Lidocaine HCl 2% and epinephrine 1:50,000 or lidocaine HCl 2% and epinephrine 1:100,000 provide an average pulp anesthesia of at least 60 minutes with an average duration of soft tissue anesthesia of approximately 2 ½ hours.
When used for nerve blocks in dental patients, the time of onset for both forms of lidocaine hydrochloride and epinephrine injection, USP averages 2 - 4 minutes. Lidocaine HCl 2% and epinephrine 1:50,000 or lidocaine HCl 2% and epinephrine 1:100,000 provide pulp anesthesia averaging at least 90 minutes with an average duration of soft tissue anesthesia of 3 to 3 ½ hours.
Hemodynamics
Excessive blood levels may cause changes in cardiac output, total peripheral resistance, and mean arterial pressure. These changes may be attributable to a direct depressant effect of the local anesthetic agent on various components of the cardiovascular system and/or the beta-adrenergic receptor stimulating action of epinephrine when present.
Pharmacokinetics and metabolism
Information derived from diverse formulations, concentrations and usages reveals that lidocaine is completely absorbed following parenteral administration, its rate of absorption depending, for example, upon various factors such as the site of administration and the presence or absence of a vasoconstrictor agent. Except for intravascular administration, the highest blood levels are obtained following intercostal nerve block and the lowest after subcutaneous administration.
The plasma binding of lidocaine is dependent on drug concentration, and the fraction bound decreases with increasing concentration. At concentration of 1 to 4 µg of free base per mL, 60 to 80 percent of lidocaine is protein bound. Binding is also dependent on the plasma concentration of the alpha-l-acid glycoprotein.
Lidocaine crosses the blood-brain and placental barriers, presumably by passive diffusion.
Lidocaine is metabolized rapidly by the liver, and metabolites and unchanged drug are excreted by the kidneys. Biotransformation includes oxidative N-dealkylation, ring hydroxylation, cleavage of the amide linkage, and conjugation. N- dealkylation, a major pathway of biotransformation, yields the metabolites monoethylglycinexylidide and glycinexylidide. The pharmacological/toxicological actions of these metabolites are similar to, but less potent than those of lidocaine. Approximately 90% of lidocaine administered is excreted in the form of various metabolites, and less than 10% is excreted unchanged. The primary metabolite in urine is a conjugate of 4-hydroxy-2, 6-dimethylaniline.
Studies of lidocaine metabolism following intravenous bolus injections have shown that the elimination half-life of this agent is typically 1.5 to 2.0 hours. Because of the rapid rate at which lidocaine is metabolized, any condition that affects liver function may alter lidocaine kinetics. The half-life may be prolonged two-fold or more in patients with liver dysfunction. Renal dysfunction does not affect lidocaine kinetics but may increase the accumulation of metabolites.
Factors such as acidosis and the use of CNS stimulants and depressants affect the CNS levels of lidocaine required to produce overt systemic effects. Objective adverse manifestations become increasingly apparent with increasing venous plasma levels above 6.0 mcg free base per mL. In the rhesus monkey, arterial blood levels of 18 - 21 µg/mL have been shown to be the threshold for convulsive activity.
- INDICATIONS AND USAGE
- CONTRAINDICATIONS
-
WARNINGS
DENTAL PRACTITIONERS WHO EMPLOY LOCAL ANESTHETIC AGENTS SHOULD BE WELL VERSED IN DIAGNOSIS AND MANAGEMENT OF EMERGENCIES WHICH MAY ARISE FROM THEIR USE. RESUSCITATIVE EQUIPMENT, OXYGEN AND OTHER RESUSCITATIVE DRUGS SHOULD BE AVAILABLE FOR IMMEDIATE USE.
To minimize the likelihood of intravascular injection, aspiration should be performed before the local anesthetic solution is injected. If blood is aspirated, the needle must be repositioned until no return of blood can be elicited by aspiration. Note, however, that the absence of blood in the syringe does not assure that intravascular injection will be avoided.
Local anesthetic procedures should be used with caution when there is inflammation and/or sepsis in the region of the proposed injection.
Lidocaine hydrochloride and epinephrine injection, USP solutions contain potassium metabisulfite, a sulfite that may cause allergic-type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown and probably low. Sulfite sensitivity is seen more frequently in asthmatic than in non asthmatic people.
The American Heart Association has made the following recommendation regarding the use of local anesthetics with vasoconstrictors in patients with ischemic heart disease: “Vasoconstrictor agents should be used in local anesthesia solutions during dental practice only when it is clear that the procedure will be shortened or the analgesia rendered more profound. When a vasoconstrictor is indicated, extreme care should be taken to avoid intravascular injection. The minimum possible amount of vasoconstrictor should be used.” (Kaplan, EL. editor: Cardiovascular disease in dental practice, Dallas 1986, American Heart Association).
Methemoglobinemia: Cases of methemoglobinemia have been reported in association with local anesthetic use. Although all patients are at risk for methemoglobinemia, patients with glucose-6-phosphate dehydrogenase deficiency, congenital or idiopathic methemoglobinemia, cardiac or pulmonary compromise, infants under 6 months of age, and concurrent exposure to oxidizing agents or their metabolites are more susceptible to developing clinical manifestations of the condition. If local anesthetics must be used in these patients, close monitoring for symptoms and signs of methemoglobinemia is recommended.
Signs of methemoglobinemia may occur immediately or may be delayed some hours after exposure, and are characterized by a cyanotic skin discoloration and/or abnormal coloration of the blood. Methemoglobin levels may continue to rise; therefore, immediate treatment is required to avert more serious central nervous system and cardiovascular adverse effects, including seizures, coma, arrhythmias, and death. Discontinue LIDOCAINE and any other oxidizing agents. Depending on the severity of the signs and symptoms, patients may respond to supportive care, i.e., oxygen therapy, hydration. A more severe clinical presentation may require treatment with methylene blue, exchange transfusion, or hyperbaric oxygen.
-
PRECAUTIONS
General
The safety and effectiveness of lidocaine depend on proper dosage, correct technique, adequate precautions and readiness for emergencies. Consult standard textbooks for specific techniques and precautions for various regional anesthetic procedures. Resuscitative equipment, oxygen and other resuscitative drugs should be available for immediate use (See WARNINGS AND ADVERSE REACTIONS).
The lowest dosage that results in effective anesthesia should be used to avoid high plasma levels and serious adverse effects. Repeated doses of lidocaine may cause significant increases in blood levels with each repeated dose due to slow accumulation of the drug or its metabolites. Tolerance to elevated blood levels varies with the status of the patient. Debilitated, elderly patients, acutely ill patients, and children should be given reduced doses commensurate with their age and physical condition.
If sedatives are employed to reduce patient apprehension, reduced doses should be used since local anesthetic agents, like sedatives, are central nervous system depressants which in combination may have an additive effect. Young children should be given minimal doses of each agent.
Lidocaine should be used with caution in patients with severe shock or heart block. Lidocaine should also be used with caution in patients with impaired cardiovascular function. Local anesthetic solutions containing a vasoconstrictor should be used with caution in areas of the body supplied by end arteries or having otherwise compromised blood supply. Patients with peripheral vascular disease and those with hypertensive vascular disease may exhibit exaggerated vasoconstrictor response. Ischemic injury (such as exfoliating or ulcerating lesions) or necrosis may result. Preparations containing a vasoconstrictor should be used with caution in patients during or following the administration of potent general anesthetic agents, since cardiac arrhythmias may occur under such conditions.
Cardiovascular and respiratory (adequacy of ventilation) vital signs and the patient’s state of consciousness should be monitored after each local anesthetic injection. Restlessness, anxiety tinnitus, dizziness, blurred vision, tremors, depression or drowsiness should alert the practitioner to the possibility of central nervous system toxicity. Signs and symptoms of depressed cardiovascular function may commonly result from a vasovagal reaction, particularly if the patient is in an upright position: placing the patient in the recumbent position is recommended when an adverse response is noted after injection of a local anesthetic (See ADVERSE REACTIONS - Cardiovascular System). Vasovagal reactions may elicit a range of clinical manifestations, from prodrome signs of pre-syncope (e.g., lightheadedness, pallor, nausea, sweating, visual disturbances, weakness) to brief loss of consciousness (i.e., syncope).
Lidocaine should be used with caution in patients with hepatic disease, since amide-type local anesthetics are metabolized by the liver. Patients with severe hepatic disease, because of their inability to metabolize local anesthetics normally, are at greater risk of developing toxic plasma concentrations.
Many drugs used during the conduct of anesthesia are considered potential triggering agents for familial malignant hyperthermia. Since it is not known whether amide-type local anesthetics may trigger this reaction, and since the need for supplemental general anesthesia cannot be predicted in advance, it is suggested that a standard protocol for management should be available. Early unexplained signs of tachycardia, tachypnea, labile blood pressure and metabolic acidosis may precede temperature elevation. Successful outcome is dependent on early diagnosis, prompt discontinuance of the suspected triggering agent (s) and prompt treatment, including oxygen therapy, dantrolene (consult dantrolene sodium intravenous package insert before using) and other supportive measures.
Lidocaine should be used with caution in persons with known drug sensitivities. Patients allergic to para-aminobenzoic acid derivatives (procaine, tetracaine, benzocaine, etc.) have not shown cross sensitivity to lidocaine.
Use in the Head and Neck Area
Small doses of local anesthetics injected into the head and neck area, including retrobulbar, dental and stellate ganglion blocks, may produce adverse reactions similar to systemic toxicity seen with unintentional intravascular injections of larger doses. Confusion, convulsions, respiratory depression and/or respiratory arrest, and cardiovascular stimulation or depression have been reported. These reactions may be due to intra-arterial injection of the local anesthetic with retrograde flow to the cerebral circulation. Patients receiving these blocks should have their circulation and respiration monitored and be constantly observed. Resuscitative equipment and personnel for treating adverse reactions should be immediately available. Dosage recommendations should not be exceeded (See DOSAGE AND ADMINISTRATION).
Information for Patients/Patient Counseling Information
The patient should be informed of the possibility of temporary loss of sensation and muscle function following infiltration or nerve block injections.
The patient should be advised to exert caution to avoid inadvertent trauma to the lips, tongue, cheek mucosae or soft palate when these structures are anesthetized. The ingestion of food should therefore be postponed until normal function returns. The patient should be advised to consult the dentist if anesthesia persists or if a rash develops. Inform patients that use of local anesthetics may cause methemoglobinemia, a serious condition that must be treated promptly. Advise patients or caregivers to seek immediate medical attention if they or someone in their care experience the following signs or symptoms: pale, gray, or blue colored skin (cyanosis); headache; rapid heart rate; shortness of breath; lightheadedness; or fatigue.Clinically Significant Drug Interactions
he administration of local anesthetic solutions containing epinephrine or norepinephrine to patients receiving monoamine oxidase inhibitors, tricyclic antidepressants or phenothiazines may produce severe prolonged hypotension or hypertension. Concurrent use of these agents should generally be avoided. In situations when concurrent therapy is necessary, careful patient monitoring is essential.
Concurrent administration of vasopressor drugs and ergot-type oxytocic drugs may cause severe, persistent hypertension or cerebrovascular accidents.As the Lidocaine HCl 2% and epinephrine 1:100,000 and the Lidocaine HCl 2% and epinephrine 1:50,000 solutions both contain a vasoconstrictor (epinephrine), concurrent use of either with a Beta-adrenergic blocking agent (propranolol, timolol, etc.) may result in dose-dependent hypertension and bradycardia with possible heart block.
Patients who are administered local anesthetics are at increased risk of developing methemoglobinemia when concurrently exposed to the following drugs, which could include other local anesthetics:
EXAMPLES OF DRUGS ASSOCIATED WITH METHEMOGLOBINEMIA Class Examples Nitrates/Nitrites nitric oxide, nitroglycerin, nitroprusside, nitrous oxide Local anesthetics articaine, benzocaine, bupivacaine, lidocaine, mepivacaine, prilocaine, procaine, ropivacaine, tetracaine Antineoplastic Agents cyclophosphamide, flutamide, hydroxyurea, ifosfamide, rasburicase Antibiotics dapsone, nitrofurantoin, para-aminosalicylic acid, sulfonamides Antimalarials chloroquine, primaquine Anticonvulsants phenobarbital, phenytoin, sodium valproate Other drugs acetaminophen, metoclopramide, quinine, sulfasalazine Drug/Laboratory test interactions
The intramuscular injection of lidocaine may result in an increase in creatine phosphokinase levels. Thus, the use of this enzyme determination, without isoenzyme separation, as a diagnostic test for the presence of acute myocardial infarction may be compromised by the intramuscular injection of lidocaine.
Carcinogenesis, mutagenesis, impairment of fertility
Studies of lidocaine in animals to evaluate the carcinogenic and mutagenic potential or the effect on fertility have not been conducted.
PREGNANCY
Teratogenic Effects
Reproduction studies have been performed in rats at doses up to 6.6 times the human dose and have revealed no evidence of harm to the fetus caused by lidocaine. There are, however, no adequate and well-controlled studies in pregnant women. Animal reproduction studies are not always predictive of human response. General consideration should be given to this fact before administering lidocaine to women of childbearing potential, especially during early pregnancy when maximum organogenesis takes place.
Nursing mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when lidocaine is administered to a nursing woman.
Pediatric use
Dosages in pediatric population should be reduced, commensurate with age, body weight and physical condition (See DOSAGE AND ADMINISTRATION).
-
ADVERSE REACTIONS
Adverse experiences following the administration of lidocaine are similar in nature to those observed with other amide-type local anesthetic agents. These adverse experiences are, in general, dose-related and may result from high plasma levels (which may be caused by excessive dosage, rapid absorption unintended intravascular injection or slow metabolic degradation), injection technique, volume of injection, hypersensitivity, idiosyncrasy or diminished tolerance on the part of the patient. Serious adverse experiences are generally systemic in nature. The following types are those most commonly reported:
Central Nervous System
CNS manifestations are excitatory and/or depressant and may be characterized by lightheadedness, nervousness, apprehension, euphoria, confusion, dizziness, drowsiness, tinnitus, blurred or double vision, vomiting, sensations of heat, cold or numbness, twitching, tremors, convulsions, unconsciousness, respiratory depression and arrest. The excitatory manifestations may be very brief or may not occur at all, in which case the first manifestation of toxicity may be drowsiness merging into unconsciousness and respiratory arrest.
Drowsiness following the administration of lidocaine is usually an early sign of a high blood level of the drug and may occur as a consequence of rapid absorption.
Cardiovascular system
Cardiovascular manifestations in response to lidocaine are usually depressant and are characterized by bradycardia, hypotension, and cardiovascular collapse, which may lead to cardiac arrest. In addition, the beta-adrenergic receptor- stimulating action of epinephrine may lead to excitatory cardiovascular responses, such as tachycardia, palpitations, and hypertension.
Signs and symptoms of depressed cardiovascular function may commonly result from a vasovagal reaction, particularly if the patient is in an upright position. Less commonly, they may result from a direct effect of the drug. Failure to recognize the premonitory signs such as sweating, a feeling of faintness, changes in pulse or sensorium may result in progressive cerebral hypoxia and seizure or serious cardiovascular catastrophe. Management consists of placing the patient in the recumbent position and ventilation with oxygen. Supportive treatment of circulatory depression may require the administration of intravenous fluids and, when appropriate, a vasopressor (e.g, ephedrine) as directed by the clinical situation.
Allergic reactions
Allergic reactions are characterized by cutaneous lesions, urticaria, edema, anaphylactoid reactions, or dyspnea due to bronchoconstriction. Allergic reactions as a result of sensitivity to lidocaine are extremely rare and, if they occur, should be managed by conventional means. The detection of sensitivity by skin testing is of doubtful value.
Neurologic reactions
The incidences of adverse reactions (e.g., persistent neurologic deficit) associated with the use of local anesthetics may be related to the technique employed, the total dose of local anesthetic administered, the particular drug used, the route of administration, and the physical condition of the patient.
Persistent paresthesias of the lips, tongue, and oral tissues have been reported with the use of lidocaine, with slow, incomplete, or no recovery. These post-marketing events have been reported chiefly following nerve blocks in the mandible and have involved the trigeminal nerve and its branches.
-
OVERDOSAGE
Acute emergencies from local anesthetics are generally related to high plasma levels encountered during therapeutic use of local anesthetics or to unintended subarachnoid injection of local anesthetic solution (See ADVERSE REACTIONS, WARNINGS AND PRECAUTIONS).
Management of local anesthetic emergencies
The first consideration is prevention, best accomplished by careful and constant monitoring of cardiovascular and respiratory vital signs and the patient’s state of consciousness after each local anesthetic injection. At the first sign of change, oxygen should be administered.
The first step in the management of convulsions consists of immediate attention to the maintenance of a patent airway and assisted or controlled ventilation with oxygen and a delivery system capable of permitting immediate positive airway pressure by mask.
Immediately after the institution of these ventilatory measures, the adequacy of the circulation should be evaluated, keeping in mind that drugs used to treat convulsions sometimes depress the circulation when administered intravenously. Should convulsions persist despite adequate respiratory support, and if the status of the circulation permits, small increments of an ultra-short acting barbiturate (such as thiopental or thiamylal) or a benzodiazepine (such as diazepam) may be administered intravenously. The clinician should be familiar, prior to use of local anesthetics, with these anticonvulsant drugs. Supportive treatment of circulatory depression may require administration of intravenous fluids and, when appropriate, a vasopressor as directed by the clinical situation (e.g., ephedrine).
If not treated immediately, both convulsions and cardiovascular depression can result in hypoxia, acidosis, bradycardia, arrhythmias and cardiac arrest. If cardiac arrest should occur, standard cardio-pulmonary resuscitative measures should be instituted. Endotracheal intubation, employing drugs and techniques familiar to the clinician, may be indicated, after initial administration of oxygen by mask, if difficulty is encountered in the maintenance of a patent airway or if prolonged ventilatory support (assisted or controlled) is indicated.
Dialysis is of negligible value in the treatment of acute overdosage with lidocaine.
The intravenous LD50 of lidocaine HCl in female mice is 26 (21 - 31) mg/kg and the subcutaneous LD50 is 264 (203 - 304) mg /kg.
-
DOSAGE AND ADMINISTRATION
The dosage of lidocaine HCl 2% and epinephrine depends on the physical status of the patient, the area of the oral cavity to be anesthetized, the vascularity of the oral tissues, and the technique of anesthesia used. The least volume of solution that results in effective local anesthesia should be administered; time should be allowed between injections to observe the patient for manifestations of an adverse reaction. For specific techniques and procedures of a local anesthesia in the oral cavity, refer to standard textbooks.
For most routine dental procedures, lidocaine HCl 2% and epinephrine 1:100,000 is preferred. However, when greater depth and a more pronounced hemostasis are required, lidocaine HCl 2% and epinephrine 1:50,000 should be used.
Dosage requirements should be determined on an individual basis. In oral infiltration and / or mandibular block, initial dosages of 1.0 - 5.0 mL (½ to 2 ½ cartridges) of lidocaine HCl 2% and epinephrine 1:50,000 or lidocaine HCl 2% and epinephrine 1:100,000 are usually effective.
In children under 10 years of age, it is rarely necessary to administer more than one-half cartridge (0.9 - 1.0 mL or 18 - 20 mg of lidocaine) per procedure to achieve local anesthesia for a procedure involving a single tooth. In maxillary infiltration, this amount will often suffice to the treatment of two or even three teeth. In the mandibular block, however, satisfactory anesthesia achieved with this amount of drug, will allow treatment of the teeth of an entire quadrant. Aspiration is recommended since it reduces the possibility of intravascular injection, thereby keeping the incidence of side effects and anesthetic failures to a minimum. Moreover, injection should always be made slowly.
Maximum recommended dosages for lidocaine HCl 2% and epinephrine 1:50,000 or lidocaine HCl 2% and epinephrine 1:100,000.
Adult
For normal healthy adults, the amount of lidocaine HCl administered should be kept below 500 mg, and in any case, should not exceed 7 mg/kg (3.2 mg/lb) of body weight.
Pediatric
Pediatric patients: It is difficult to recommend a maximum dose of any drug for pediatric patients since this varies as a function of age and weight. For pediatric patients of less than ten years who have a normal lean body mass and normal body development, the maximum dose may be determined by the application of one of the standard pediatric drug formulas (e.g., Clark’s rule). For example, in pediatric patients of five years weighing 50 Ibs, the dose of lidocaine hydrochloride should not exceed 75 - 100 mg when calculated according to Clark’s rule. In any case, the maximum dose of lidocaine hydrochloride should not exceed 7 mg/kg (3.2 mg/lb) of body weight.
NOTE: Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever the solution and container permit. Solutions that are discolored and / or contain particulate matter should not be used; and any unused portion of a cartridge of lidocaine hydrochloride and epinephrine injection, USP should be discarded.
-
HOW SUPPLIED
- Lidocaine hydrochloride 2% (34 mg/1.7 mL) (20 mg/mL) and Epinephrine 1:50,000 injection is available in cartons containing 5 blisters of 10 x 1.7 mL single-dose cartridges (NDC 0362-0262-05).
- Lidocaine hydrochloride 2% (34 mg/1.7 mL) (20 mg/mL) and Epinephrine 1:100,000 injection is available in cartons containing 5 blisters of 10 x 1.7 mL single-dose cartridges (NDC 0362-0898-05).
Store at controlled room temperature, below 25°C (77°F). Protect from light. Do not permit to freeze.
BOXES: For protection from light, retain in box until time of use. Once opened, the box should be reclosed by closing the end flap. Do not use if color is pinkish or darker than slightly yellow or if it contains a precipitate.
STERILIZATION : STORAGE AND TECHNICAL PROCEDURES
- Cartridges should not be autoclaved, because the closures employed cannot withstand autoclaving temperatures and pressures.
- If chemical disinfection of anesthetic cartridges is desired, either isopropyl alcohol (91%) or 70% ethyl alcohol is recommended. Many commercially available brands of rubbing alcohol, as well as solutions of ethyl alcohol not of U.S.P grade, contain denaturants that are injurious to rubber and, therefore, are not to be used. It is recommended that chemical disinfection be accomplished just prior to use by wiping the cartridge cap thoroughly with a pledge of cotton that has been moistened with recommended alcohol.
- Certain metallic ions (mercury, zinc, copper, etc.) have been related to swelling and edema after local anesthesia in dentistry. Therefore, chemical disinfectants containing or releasing these ions are not recommended. Antirust tablets usually contain sodium nitrite or some similar agents that may be capable of releasing metal ions. Because of this, aluminium sealed cartridges should not be kept in such solutions.
- Quaternary ammonium salts, such as benzalkonium chloride, are electrolytically incompatible with aluminium. Cartridges of lidocaine hydrochloride and epinephrine injection, USP are sealed with aluminium caps and therefore should not be immersed in any solution containing these salts.
- To avoid leakage of solutions during injection, be sure to penetrate the center of the rubber diaphragm when loading the syringe. An off-center penetration produces an oval shaped puncture that allows leakage around the needle. Other causes of leakage and breakage include badly worn syringes, aspirating syringes with bent harpoons, the use of syringes not designed to take 1.7 mL cartridges, and inadvertent freezing.
- Cracking of glass cartridges is most often the result of an attempt to use a cartridge with an extruded plunger. An extruded plunger loses its lubrication and can be forced back into the cartridge only with difficulty. Cartridges with extruded plungers should be discarded.
- Store at controlled room temperature, below 25°C (77°F).
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 1.7 mL Cartridge Carton
-
INGREDIENTS AND APPEARANCE
LIDOCAINE
lidocaine hydrochloride and epinephrine injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0362-0898 Route of Administration SUBMUCOSAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Lidocaine Hydrochloride (UNII: V13007Z41A) (Lidocaine - UNII:98PI200987) LIDOCAINE HYDROCHLORIDE ANHYDROUS 20 mg in 1 mL Epinephrine Bitartrate (UNII: 30Q7KI53AK) (Epinephrine - UNII:YKH834O4BH) Epinephrine 0.01 mg in 1 mL Inactive Ingredients Ingredient Name Strength Potassium Metabisulfite (UNII: 65OE787Q7W) 1.2 mg in 1 mL Sodium Chloride (UNII: 451W47IQ8X) 6.5 mg in 1 mL Edetate Disodium (UNII: 7FLD91C86K) 0.25 mg in 1 mL Sodium Hydroxide (UNII: 55X04QC32I) HYDROCHLORIC ACID (UNII: QTT17582CB) Water (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0362-0898-05 50 in 1 CARTON 07/18/2018 1 1.7 mL in 1 CARTRIDGE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA088390 07/18/2018 Labeler - Septodont, Inc. (627058738) Registrant - Novocol Pharmaceutical of Canada, Inc. (201719960) Establishment Name Address ID/FEI Business Operations Novocol Pharmaceutical of Canada, Inc. 201719960 MANUFACTURE(0362-0898)


