Label: ZOMACTON- somatropin kit
- NDC Code(s): 55566-1801-1, 55566-1901-1, 55566-1902-1
- Packager: Ferring Pharmaceuticals Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Biologic Licensing Application
Drug Label Information
Updated May 2, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use ZOMACTON® safely and effectively. See full prescribing information for ZOMACTON.
ZOMACTON® (somatropin) for injection, for subcutaneous use
Initial U.S. Approval: 1995INDICATIONS AND USAGE
ZOMACTON is a recombinant human growth hormone indicated for:
- Pediatric: Treatment of pediatric patients with growth failure due to inadequate secretion of endogenous growth hormone (GH), short stature associated with Turner syndrome, idiopathic short stature (ISS), short stature or growth failure in short stature homeobox-containing gene (SHOX) deficiency, and short stature born small for gestational age (SGA) with no catch-up growth by 2 years to 4 years. (1.1)
- Adult: Replacement of endogenous GH in adults with GH deficiency (1.2)
DOSAGE AND ADMINISTRATION
- Administer by subcutaneous injection to the back of upper arm, abdomen, buttock, or thigh with regular rotation of injection sites (2.1)
- Pediatric dosage: Divide the calculated weekly dosage into equal doses given either 3, 6, or 7 days per week (2.2)
-
Adult dosage: Either of the following two dosing regimens may be used:
- -
- Non-weight based dosing: Initiate with a dose of approximately 0.2 mg/day (range, 0.15 mg/day to 0.3 mg/day) and increase the dose every 1 to 2 months by increments of approximately 0.1 mg/day to 0.2 mg/day, according to individual patient requirements (2.3)
- -
- Weight-based dosing (Not recommended for obese patients): Initiate at 0.006 mg/kg daily and increase the dose according to individual patient requirements to a maximum of 0.0125 mg/kg daily (2.3)
- See Full Prescribing Information for reconstitution instructions (2.4)
DOSAGE FORMS AND STRENGTHS
CONTRAINDICATIONS
- Acute critical illness (4,5.1)
- Pediatric patients with Prader-Willi syndrome who are severely obese, have a history of upper airway obstruction or sleep apnea, or have severe respiratory impairment due to risk of sudden death (4, 5.2)
- Active malignancy (4)
- Hypersensitivity to ZOMACTON, its excipients, or diluents (4)
- Active proliferative or severe non-proliferative diabetic retinopathy (4)
- Pediatric patients with closed epiphyses (4)
WARNINGS AND PRECAUTIONS
- Increased Risk of Neoplasm: Second neoplasms have occurred in childhood cancer survivors. Monitor patients with preexisting tumors for progression or recurrence. (5.3)
- Glucose Intolerance and Diabetes Mellitus: ZOMACTON may decrease insulin sensitivity, particularly at higher doses. Monitor glucose levels periodically in all patients receiving ZOMACTON, especially in patients with existing diabetes mellitus or at risk for development. (5.4)
- Intracranial Hypertension (IH): Has been reported usually within 8 weeks of initiation. Perform fundoscopic examinations prior to initiation and periodically thereafter. If papilledema occurs, stop treatment. (5.5)
- Hypersensitivity: Serious hypersensitivity reactions may occur. In the event of an allergic reaction, seek prompt medical attention. (5.6)
- Fluid Retention: May occur in adults and may be dose dependent. (5.7)
- Hypoadrenalism: Monitor patients for reduced serum cortisol levels and/or need for glucocorticoid dose increases in those with known hypoadrenalism. (5.8)
- Hypothyroidism: Monitor thyroid function periodically as hypothyroidism may occur or worsen after initiation of somatropin. (5.9)
- Slipped Capital Femoral Epiphysis in Pediatric Patients: May occur; evaluate patients with onset of a limp or hip/knee pain. (5.10)
- Progression of Preexisting Scoliosis in Pediatric Patients: Monitor patients with scoliosis for progression. (5.11)
- Pancreatitis: Has been reported; consider pancreatitis in patients with abdominal pain, especially pediatric patients. (5.12)
- Risk of Serious Adverse Reactions in Infants due to Benzyl Alcohol Preservative: Serious and fatal adverse reactions can occur in neonates and infants treated with benzyl alcohol-preserved drugs, including the diluent for ZOMACTON 5 mg. If administering ZOMACTON 5 mg to infants, reconstitute with 0.9% sodium chloride injection. (5.13)
ADVERSE REACTIONS
Common adverse reactions reported in adult and pediatric patients include: upper respiratory infection, fever, pharyngitis, headache, otitis media, edema, arthralgia, paresthesia, myalgia, carpal tunnel syndrome, peripheral edema, flu syndrome, hypothyroidism, hyperglycemia, and impaired glucose tolerance. (6)
To report SUSPECTED ADVERSE REACTIONS, contact Ferring Pharmaceuticals Inc. at 1-888-337-7464 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- Glucocorticoids: Patients treated with glucocorticoid for hypoadrenalism may require an increase in their maintenance or stress doses following initiation of ZOMACTON (7)
- Pharmacologic Glucocorticoid Therapy and Supraphysiologic Glucocorticoid Treatment: Adjust glucocorticoid replacement dosing in pediatric patients receiving glucocorticoid treatment to avoid both hypoadrenalism and an inhibitory effect on growth. (7)
- Cytochrome P450-Metabolized Drugs: ZOMACTON may alter the clearance. Monitor carefully if used with ZOMACTON (7)
- Oral Estrogen: Larger doses of ZOMACTON may be required (7)
- Insulin and/or Other Hypoglycemic Agents: Dose adjustment of insulin or hypoglycemic agent may be required (5.4, 7)
USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 4/2024
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
1.1 Pediatric Patients
1.2 Adult Patients
2 DOSAGE AND ADMINISTRATION
2.1 Administration and Use Instructions
2.2 Pediatric Dosage
2.3 Adult Dosage
2.4 Reconstitution
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Increased Mortality in Patients with Acute Critical Illness
5.2 Sudden Death in Pediatric Patients with Prader-Willi Syndrome
5.3 Increased Risk of Neoplasms
5.4 Glucose Intolerance and Diabetes Mellitus
5.5 Intracranial Hypertension
5.6 Severe Hypersensitivity
5.7 Fluid Retention
5.8 Hypoadrenalism
5.9 Hypothyroidism
5.10 Slipped Capital Femoral Epiphysis in Pediatric Patients
5.11 Progression of Preexisting Scoliosis in Pediatric Patients
5.12 Pancreatitis
5.13 Risk of Serious Adverse Reactions in Infants due to Benzyl Alcohol Preserved Solution
5.14 Lipoatrophy
5.15 Laboratory Tests
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
9 DRUG ABUSE AND DEPENDENCE
9.1 Controlled Substance
9.2 Abuse
9.3 Dependence
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
12.6 Immunogenicity
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Pediatric Patients with Growth Failure Due to Inadequate Secretion of Endogenous Growth Hormone
14.2 Pediatric Patients with Short Stature Associated with Turner Syndrome
14.3 Pediatric Patients with Idiopathic Short Stature
14.4 Pediatric Patients with Short Stature or Growth Failure with SHOX Deficiency
14.5 Pediatric Patients Born Small for Gestational Age (SGA) Who Fail to Demonstrate Catch-up Growth by 2 Years to 4 Years of Age
14.6 Adult Patients with Growth Hormone Deficiency
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
16.2 Storage and Handling
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
1.1 Pediatric Patients
ZOMACTON is indicated for the treatment of pediatric patients with:
- growth failure due to inadequate secretion of endogenous growth hormone (GH),
- short stature associated with Turner syndrome,
- idiopathic short stature (ISS), height standard deviation score (HSDS) ≤-2.25 and associated with growth rates unlikely to permit attainment of adult height in the normal range,
- short stature or growth failure in short stature homeobox-containing gene (SHOX) deficiency,
- short stature born small for gestational age (SGA) with no catch-up growth by 2 years to 4 years of age.
-
2 DOSAGE AND ADMINISTRATION
2.1 Administration and Use Instructions
- Therapy with ZOMACTON should be supervised by a physician who is experienced in the diagnosis and management of patients with the conditions for which ZOMACTON is indicated [see Indications and Usage (1)].
- Fundoscopic examination should be performed routinely before initiating treatment with ZOMACTON to exclude preexisting papilledema, and periodically thereafter [see Warnings and Precautions (5.5)].
- Administer ZOMACTON by subcutaneous injection to the back of the upper arm, abdomen, buttock, or thigh with regular rotation of injection sites to avoid lipoatrophy.
- ZOMACTON 5 mg and 10 mg can be administered using a standard sterile disposable syringe. For proper use, please refer to the Instructions for Use provided with the administration device. The volume of the syringe should be small enough so that the prescribed dose can be withdrawn from the vial with reasonable accuracy.
2.2 Pediatric Dosage
- Individualize dosage for each patient based on the growth response.
- Divide the calculated weekly ZOMACTON dosage into equal doses given either 3, 6, or 7 days per week.
- The recommended weekly dose in milligrams (mg) per kilogram (kg) of body weight for pediatric patients is:
- Pediatric GH Deficiency: 0.18 mg/kg/week to 0.3 mg/kg/week (0.026 mg/kg/day to 0.043 mg/kg/day)
- Turner syndrome: Up to 0.375 mg/kg/week (up to 0.054 mg/kg/day)
- Idiopathic short stature: Up to 0.37 mg/kg/week (up to 0.053 mg/kg/day)
- SHOX Deficiency: 0.35 mg/kg/week (0.05 mg/kg/day)
-
Small for Gestational Age (SGA): Up to 0.47 mg/kg/week (up to 0.067 mg/kg/day)
- In very short pediatric patients, HSDS less than -3, and older pubertal pediatric patients consider initiating treatment with a larger dose of ZOMACTON (up to 0.067 mg/kg/day). Consider a gradual reduction in dosage if substantial catch-up growth is observed during the first few years of therapy. In pediatric patients less than 4 years of age with less severe short stature, baseline HSDS values between -2 and -3, consider initiating treatment at 0.033 mg/kg/day and titrate the dose as needed.
- Assess compliance and evaluate other causes of poor growth such as hypothyroidism, under-nutrition, advanced bone age and antibodies to recombinant human GH if patients experience failure to increase height velocity, particularly during the first year of treatment.
- Discontinue ZOMACTON for stimulation of linear growth once epiphyseal fusion has occurred [see Contraindications (4)].
2.3 Adult Dosage
- Patients who were treated with somatropin for GH deficiency in childhood and whose epiphyses are closed should be reevaluated before continuation of somatropin for GH deficient adults.
- Consider using a lower starting dose and smaller dose increment increases for geriatric patients as they may be at increased risk for adverse reactions with ZOMACTON than younger individuals [see Use in Specific Populations (8.5)].
- Estrogen-replete women and patients receiving oral estrogen may require higher doses [see Drug Interactions (7)].
- Administer the prescribed dose daily
- Either of two ZOMACTON dosing regimens may be used:
-
Non-weight based
- Initiate ZOMACTON with a dose of approximately 0.2 mg/day (range, 0.15 mg/day to 0.3 mg/day) and increase the dose every 1 to 2 months by increments of approximately 0.1 mg/day to 0.2 mg/day, according to individual patient requirements based on the clinical response and serum insulin-like growth factor 1 (IGF-1) concentrations.
- Decrease the dose as necessary on the basis of adverse reactions and/or serum IGF-1 concentrations above the age- and gender-specific normal range.
- Maintenance dosages will vary considerably from person to person, and between male and female patients.
-
Weight-based
- Initiate ZOMACTON at 0.006 mg/kg daily and increase the dose according to individual patient requirements to a maximum of 0.0125 mg/kg daily.
- Use the patient's clinical response, adverse reactions, and determination of age- and gender-adjusted serum IGF-1 concentrations as guidance in dose titration.
- Not recommended for obese patients as they are more likely to experience adverse reactions with this regimen.
-
Non-weight based
2.4 Reconstitution
- Reconstitute ZOMACTON 5 mg with 1 mL to 5 mL of diluent, Bacteriostatic Sodium Chloride Injection, USP with 0.9% benzyl alcohol as a preservative. Do not use diluent if the patient has a known hypersensitivity to benzyl alcohol [see Contraindications (4)] or in neonates [see Warnings and Precautions (5.13)], or pregnant or lactating women [see Use in Specific Populations (8.1,8.2)] instead reconstitute with 0.9% Sodium Chloride Injection, USP, use only one dose per vial, and discard the unused portion.
- Reconstitute ZOMACTON 10 mg with 1 mL syringe of diluent, Bacteriostatic Water for Injection, USP with 0.33% metacresol as a preservative. Do not use diluent if the patient has a known hypersensitivity to metacresol [see Contraindications (4)].
- Aim the stream of diluent against the side of the vial to prevent foaming and gently swirl the vial with a rotary motion until the contents are completely dissolved and the solution is clear. Do not shake the vial since shaking or vigorous mixing will cause the solution to be cloudy.
- Inspect visually for particulate matter and discoloration. If the resulting solution is cloudy or contains particulate matter do not use.
- Occasionally, after refrigeration, some cloudiness may occur. Allow the product to warm to room temperature. If cloudiness persists or particulate matter is noted do not use.
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
ZOMACTON is contraindicated in patients with:
- Acute critical illness after open heart surgery, abdominal surgery or multiple accidental trauma, or those with acute respiratory failure due to the risk of increased mortality with use of pharmacologic doses of somatropin [see Warnings and Precautions (5.1)].
- Pediatric patients with Prader-Willi syndrome who are severely obese, have a history of upper airway obstruction or sleep apnea, or have severe respiratory impairment due to the risk of sudden death [see Warnings and Precautions (5.2)].
- Active malignancy [see Warnings and Precautions (5.3)].
- Known hypersensitivity to somatropin or to any excipients of ZOMACTON. Systemic hypersensitivity reactions, including anaphylaxis and angioedema, have been reported with postmarketing use of somatropins [see Dosage and Administrations (2.4), Warnings and Precautions (5.6)].
- Active proliferative or severe non-proliferative diabetic retinopathy.
- Pediatric patients with closed epiphyses.
-
5 WARNINGS AND PRECAUTIONS
5.1 Increased Mortality in Patients with Acute Critical Illness
Increased mortality in patients with acute critical illness due to complications following open heart surgery, abdominal surgery or multiple accidental trauma, or those with acute respiratory failure has been reported after treatment with pharmacologic doses of somatropin [see Contraindications (4)]. Two placebo-controlled clinical trials in non-GH deficient adult patients (n=522) with these conditions in intensive care units revealed a significant increase in mortality (42% vs. 19%) among somatropin-treated patients (doses 5.3 mg/day-8 mg/day) compared to those receiving placebo. The safety of continuing ZOMACTON treatment in patients receiving replacement doses for approved indications who concurrently develop these illnesses has not been established. ZOMACTON is not indicated for the treatment of non-GH deficient adults.
5.2 Sudden Death in Pediatric Patients with Prader-Willi Syndrome
There have been reports of sudden death after initiating therapy with somatropin in pediatric patients with Prader-Willi syndrome who had one or more of the following risk factors: severe obesity, history of upper airway obstruction or sleep apnea, or unidentified respiratory infection. Male patients with one or more of these factors may be at greater risk than females. Patients with Prader-Willi syndrome should be evaluated for signs of upper airway obstruction and sleep apnea before initiation of treatment with somatropin. If, during treatment with somatropin, patients show signs of upper airway obstruction (including onset of, or increased, snoring) and/or new onset sleep apnea, treatment should be interrupted. All patients with Prader-Willi syndrome treated with somatropin should also have effective weight control and be monitored for signs of respiratory infection, which should be diagnosed as early as possible and treated aggressively [see Contraindications (4)]. ZOMACTON is not indicated for the treatment of pediatric patients who have growth failure due to Prader-Willi syndrome.
5.3 Increased Risk of Neoplasms
Active Malignancy
There is an increased risk of malignancy progression with somatropin treatment in patients with active malignancy [see Contraindications (4)]. Any preexisting malignancy should be inactive and its treatment complete prior to instituting therapy with ZOMACTON. Discontinue ZOMACTON if there is evidence of recurrent activity.
Risk of Second Neoplasm in Pediatric Patients
There is an increased risk of a second neoplasm in pediatric cancer survivors who were treated with radiation to the brain/head and who developed subsequent GH deficiency and were treated with somatropin. Intracranial tumors, in particular meningiomas, were the most common of these second neoplasms. In adults, it is unknown whether there is any relationship between somatropin replacement therapy and CNS tumor recurrence. Monitor all patients receiving ZOMACTON who have a history of GH deficiency secondary to an intracranial neoplasm for progression or recurrence of the tumor.
New Malignancy During Treatment
Because pediatric patients with certain rare genetic causes of short stature have an increased risk of developing malignancies, thoroughly consider the risks and benefits of starting ZOMACTON in these patients. If ZOMACTON is initiated, these patients should be carefully monitored for development of neoplasms.
Monitor all patients receiving ZOMACTON carefully for increased growth, or potential malignant changes, of preexisting nevi. Advise patients/caregivers to report marked changes in behavior, onset of headaches, vision disturbances and/or changes in skin pigmentation or changes in the appearance of pre-existing nevi.
5.4 Glucose Intolerance and Diabetes Mellitus
Treatment with somatropin may decrease insulin sensitivity, particularly at higher doses. New onset type 2 diabetes mellitus has been reported in patients taking somatropin. Previously undiagnosed impaired glucose tolerance and overt diabetes mellitus may be unmasked. Monitor glucose levels periodically in all patients receiving ZOMACTON, especially in those with risk factors for diabetes mellitus, such as obesity, Turner syndrome, or a family history of diabetes mellitus. Patients with preexisting type 1 or type 2 diabetes mellitus or impaired glucose tolerance should be monitored closely. The doses of antidiabetic agents may require adjustment when ZOMACTON is initiated.
5.5 Intracranial Hypertension
Intracranial hypertension (IH) with papilledema, visual changes, headache, nausea, and/or vomiting has been reported in a small number of patients treated with somatropin. Symptoms usually occurred within the first eight (8) weeks after the initiation of somatropin. In all reported cases, IH-associated signs and symptoms resolved rapidly after cessation of therapy or a reduction of the somatropin dose. Fundoscopic examination should be performed routinely before initiating treatment with ZOMACTON to exclude preexisting papilledema, and periodically thereafter. If papilledema is observed by fundoscopy, treatment should be stopped. If somatropin-induced IH is diagnosed, treatment with ZOMACTON can be restarted at a lower dose after IH-associated signs and symptoms have resolved. Patients with Turner syndrome may be at increased risk for the development of IH.
5.6 Severe Hypersensitivity
Serious systemic hypersensitivity reactions including anaphylactic reactions and angioedema have been reported with postmarketing use of somatropins. Patients and caregivers should be informed that such reactions are possible and that prompt medical attention should be sought if an allergic reaction occurs [see Contraindications (4)].
5.7 Fluid Retention
Fluid retention during somatropin replacement therapy in adults may frequently occur. Clinical manifestations of fluid retention (e.g. edema, arthralgia, myalgia, nerve compression syndromes including carpal tunnel syndrome/paresthesias) are usually transient and dose dependent.
5.8 Hypoadrenalism
Patients receiving somatropin therapy who have or are at risk for pituitary hormone deficiency(s) may be at risk for reduced serum cortisol levels and/or unmasking of central (secondary) hypoadrenalism. In addition, patients treated with glucocorticoid replacement for previously diagnosed hypoadrenalism may require an increase in their maintenance or stress doses following initiation of ZOMACTON. Monitor patients for reduced serum cortisol levels and/or need for glucocorticoid dose increases in those with known hypoadrenalism [see Drug Interactions (7)].
5.9 Hypothyroidism
Undiagnosed or untreated hypothyroidism may prevent response to ZOMACTON, in particular, the growth response in pediatric patients. Patients with Turner syndrome have an increased risk of developing autoimmune thyroid disease and primary hypothyroidism. In patients with GH deficiency, central (secondary) hypothyroidism may first become evident or worsen during somatropin treatment. Therefore, patients should have periodic thyroid function tests performed, and thyroid hormone replacement therapy should be initiated or appropriately adjusted when indicated.
5.10 Slipped Capital Femoral Epiphysis in Pediatric Patients
Slipped capital femoral epiphysis may occur more frequently in patients undergoing rapid growth. Evaluate pediatric patients with the onset of a limp or complaints of hip or knee pain.
5.11 Progression of Preexisting Scoliosis in Pediatric Patients
Somatropin increases the growth rate and progression of existing scoliosis can occur in patients who experience rapid growth. Somatropin has not been shown to increase the occurrence of scoliosis. Monitor patients with a history of scoliosis for progression of scoliosis.
5.12 Pancreatitis
Cases of pancreatitis have been reported in pediatric patients and adults receiving somatropin. The risk may be greater in pediatric patients compared with adults. Published literature indicates that girls who have Turner syndrome may be at greater risk than other pediatric patients receiving somatropin. Pancreatitis should be considered in patients who develop abdominal pain.
5.13 Risk of Serious Adverse Reactions in Infants due to Benzyl Alcohol Preserved Solution
Serious and fatal adverse reactions including "gasping syndrome" can occur in neonates and infants treated with benzyl alcohol-preserved drugs, including the bacteriostatic 0.9% sodium chloride diluent provided with ZOMACTON 5 mg. The "gasping syndrome" is characterized by central nervous system depression, metabolic acidosis, and gasping respirations.
When administering ZOMACTON 5 mg to infants, reconstitute with 0.9% sodium chloride injection, not with the diluent provided. Use only one dose per vial and discard the unused portion [see Use in Specific Populations (8.4)].
5.14 Lipoatrophy
When somatropin is administered subcutaneously at the same site over a long period of time, tissue atrophy may result. Rotate injection sites when administering ZOMACTON to reduce this risk [see Dosage and Administration (2.2)].
-
6 ADVERSE REACTIONS
The following important adverse reactions are also described elsewhere in the labeling:
- Increased mortality in patients with acute critical illness [see Warnings and Precautions (5.1)]
- Fatalities in pediatric patients with Prader-Willi syndrome [see Warnings and Precautions (5.2)]
- Neoplasms [see Warnings and Precautions (5.3)]
- Glucose intolerance and diabetes mellitus [see Warnings and Precautions (5.4)]
- Intracranial hypertension [see Warnings and Precautions (5.5)]
- Severe hypersensitivity [see Warnings and Precautions (5.6)]
- Fluid retention [see Warnings and Precautions (5.7)]
- Hypoadrenalism [see Warnings and Precautions (5.8)]
- Hypothyroidism [see Warnings and Precautions (5.9)]
- Slipped capital femoral epiphysis in pediatric patients [see Warnings and Precautions (5.10)]
- Progression of preexisting scoliosis in pediatric patients [see Warnings and Precautions (5.11)]
- Pancreatitis [see Warnings and Precautions (5.12)]
- Risk of Serious Adverse Reactions in Infants due to Benzyl Alcohol Preservative [see Warnings and Precautions (5.13)]
- Lipoatrophy [see Warnings and Precautions (5.14)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under varying conditions, adverse reaction rates observed during the clinical trials performed with one somatropin formulation cannot always be directly compared to the rates observed during the clinical trials performed with a different approved somatropin formulation and may not reflect the adverse reaction rates observed in practice.
Pediatric Patients
Growth Failure due to Inadequate Secretion of Endogenous Growth Hormone
ZOMACTON was evaluated in 164 pediatric patients with short stature due to GHD in an open-label study for 24 weeks. The subjects ranged in age from 2.1 to 17.7 years with a mean of 10.8 years. One hundred twenty (73%) of the subjects were male and 44 (27%) were female. Two subjects were Asian, 12 were Black, 130 were Caucasian, and 20 were categorized as 'other'.
Table 1: Adverse Reactions ≥ 5% in Pediatric Patients with Growth Failure Due to GHD Treated with ZOMACTON through 24 Weeks Adverse Reaction 24 Week Exposure to ZOMACTON
(n=164)Upper respiratory infection 32% Fever 16% Pharyngitis 12% Headache 11% Otitis Media 10% Increased cough 9% Abdominal pain 7% Anemia 6% Maculopapular rash 6% Diarrhea 5% Pain 5% Rhinitis 5% All pediatric patients were carefully observed for signs or laboratory abnormalities of hypothyroidism. Fifteen patients had T4 values which occasionally fell below the central laboratory's lower limit of normal; T4 levels rose to normal when tested during the next visit for all patients except one who continued to be monitored. Six of the 15 patients received thyroxine therapy before and throughout the study period, and thyroxine dose adjustments were made during the study in 3/6 subjects.
In studies with GH deficient pediatric patients, injection site pain was reported in 1.6% of patients. A mild and transient edema, which appeared in 1.6% of patients, was observed early during the course of treatment.
Short Stature Associated with Turner Syndrome
In a randomized, concurrent-controlled, open-label study, there was an increase in the occurrence of otitis media, ear disorders and surgical procedures in patients receiving another somatropin product at a dose of 0.3 mg/kg/week, compared with untreated control patients (Table 2). A similar increase in otitis media was observed in an 18-month placebo-controlled study.
Table 2: Adverse Reactions Occurring in Patients with Turner Syndrome Treated with 0.3 mg/kg/week of Another Somatropin product During a 4.7 Year Randomized Open-Label Study Untreated
(n=62)Somatropin
(n=74)Surgical procedure 27% 45% Otitis media 26% 43% Ear disorders 5% 18% Idiopathic Short Stature
Adverse reactions from a randomized, placebo-controlled study of another somatropin product dosed at 0.22 mg/kg/week are presented in Table 3. Mean fasting serum insulin concentration increased 10% in the group treated with another somatropin product at the end of treatment relative to baseline, but remained within the normal reference range.
Table 3: Adverse Reactions Occurring in Patients with Idiopathic Short Stature Treated with Another Somatropin product during a 4.4 Year Randomized Placebo-Controlled Study Adverse Reactions Placebo
(n=31)Another Somatropin product
(n=37)Scoliosis 13% 19% Otitis media 7% 16% Hyperlipidemia 3% 8% Gynecomastia 3% 5% Hip pain 0% 3% Arthralgia 3% 11% Arthrosis 7% 11% Myalgia 13% 24% Hypertension 0% 3% In a dose-response study with 239 patients treated for 2 years, mean fasting blood glucose, mean glycosylated hemoglobin, and the incidence of elevated fasting blood glucose concentrations were similar among dose groups. One patient developed glucose intolerance and high serum HbA1c.
Short Stature or Growth Failure in SHOX Deficiency
Adverse reactions from a 2-year open-label study with another somatropin product compared to no treatment are presented in Table 4. During the study, the proportion of patients who had at least one IGF-1 concentration greater than 2.0 SD above the age- and gender-appropriate mean was 37.0% for the somatropin-treated group vs. 0% patients for the untreated group. The proportion of patients who had at least one IGFBP-3 concentration greater than 2.0 SD above the age and gender appropriate mean was 59% for the somatropin treated group vs. 29% for the untreated group.
Table 4: Adverse Reactions Occurring in Patients with SHOX Deficiency Treated with Another Somatropin product for 2 years Untreated
(n=25)Another Somatropin product
(n=27)Arthralgia 8% 11% Gynecomastia 0% 8% Excessive number of cutaneous nevi 0% 7% Scoliosis 0% 4% Small for Gestational Age (SGA) with No Catch-up Growth by 2 Years to 4 Years of Age
In a 2-year study, 193 pediatric patients were treated with another somatropin product using 2 different treatment regimens: a fixed dose of 0.067 mg/kg/day (FHD group) or an individually adjusted dose regimen (IAD group; starting dose 0.035 mg/kg/day which could be increased as early as Month 3 to 0.067 mg/kg/day based on a validated growth prediction model). Adverse reactions included common childhood infectious diseases, otitis media, headaches, and slipped capital femoral epiphysis (n=1). Six patients (4 in the FHD group and 2 in the IAD group whose dose was increased from 0.035 mg/kg/day to 0.067 mg/kg/day [one at Month 3 and one at Year 1])) had impaired fasting glucose at Year 2. Two of 6 had impaired fasting glucose during the study, and 1 discontinued treatment at month 15 as a consequence. At study completion, 20-25% of patients had serum IGF-1 SDS values > +2.
The following adverse reactions were reported from an observational study of 340 pediatric patients who received another somatropin product with an average dosage of 0.041 mg/kg/day for an average of 3 years: type 2 diabetes mellitus (n=1), carpal tunnel syndrome (n=1) and exacerbation of preexisting scoliosis (n=1).
Adult Patients
Adult-Onset GH Deficiency
In the first 6 months of controlled blinded trials during which patients received either another somatropin product or placebo, patients who received this other somatropin product experienced a statistically significant increase in edema (another somatropin product 17% vs. placebo 4%, p=0.043) and peripheral edema (12% vs. 0%, respectively, p=0.017). Edema, muscle pain, joint pain, and joint disorder were reported early in therapy and tended to be transient or responsive to dosage titration.
Two of 113 patients developed carpal tunnel syndrome after beginning maintenance therapy without a low dose (0.00625 mg/kg/day) lead-in phase. Symptoms abated in these patients after dosage reduction.
All adverse reactions with ≥5% overall occurrence rate during 12 or 18 months of replacement therapy with another somatropin product are shown in Table 5 (adult-onset patients) and in Table 6 (childhood-onset patients).
Adult patients treated with another somatropin product who had been diagnosed with GH deficiency in childhood reported adverse reactions less frequently than those with adult-onset GH deficiency.
Table 5: Adverse Reactions Occurring ≥5% in Adult-Onset Growth Hormone-Deficient Patients Treated with Another Somatropin product for 18 Months as Compared with 6-Month Placebo and 12-Month Exposure to Another Somatropin product* 18 Months Exposure Adverse Reaction [Placebo (6 Months)/GH (12 Months)] 18 Months GH Exposure (n=46) (n=52) Edema† 15% 21% Arthralgia 15% 17% Paresthesia 13% 17% Myalgia 13% 14% Pain 13% 14% Rhinitis 11% 14% Peripheral edema‡ 17% 12% Back pain 11% 10% Headache 11% 8% Hypertension 4% 8% Acne 0% 6% Joint disorder 2% 6% Surgical procedure 2% 6% Flu syndrome 7% 4% Childhood-Onset GH Deficiency
Two double-blind, placebo-controlled trials were conducted in 67 adult patients who had received previous somatropin treatment during childhood. Patients were randomized to receive either placebo injections or another somatropin product (0.00625 mg/kg/day for the first 4 weeks, then 0.0125 mg/kg/day thereafter) for the first 6 months, followed by open-label use of another somatropin product for the next 12 months for all patients. The patients in these studies reported side effects less frequently than those with adult-onset GH deficiency. During the placebo-controlled phase (first 6 months) of the study, elevations of serum glutamic oxaloacetic transferase were reported significantly more often for somatropin-treated (12.5%) than placebo-treated patients (0.0%, p=0.031). No other events were reported significantly more often for somatropin-treated patients during the placebo-controlled phase.
Table 6: Adverse Reactions Occurring ≥5% in Childhood-Onset Growth Hormone-Deficient Patients Treated with Another Somatropin product for 18 Months as Compared with 6-Month Placebo and 12-Month Exposure to Another Somatropin product* 18 Months Exposure 18 Months GH Exposure Adverse Reaction [Placebo (6 Months)/GH (12 Months)] (n=35) (n=32) Flu syndrome 23% 16% AST increased† 6% 13% Headache 11% 9% Asthenia 3% 6% Cough increased 0% 6% Edema 9% 6% Hypesthesia 0% 6% Myalgia 6% 6% Pain 9% 6% Rhinitis 6% 6% ALT increased 6% 6% Respiratory disorder 6% 3% Gastritis 6% 0% Pharyngitis 14% 3% In an ongoing post-marketing observational study of treatment with another somatropin product in 3,102 GH-deficient adults, hypertension, dyspnea, and sleep apnea were reported by 1% to less than 10% of patients after various durations of treatment.
6.2 Postmarketing Experience
Because the following adverse events are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Severe Hypersensitivity Reactions — Serious systemic hypersensitivity reactions including anaphylactic reactions and angioedema
Neurologic — Headaches (common in pediatric patients and occasional in adults)
Skin — Increase in size or number of cutaneous nevi
Endocrine — Gynecomastia.
Gastrointestinal — Pancreatitis
Metabolic — New-onset type 2 diabetes mellitus
Neoplasia — Leukemia has been reported in a small number of GH deficient pediatric patients treated with somatropin, somatrem (methionylated rhGH), and GH of pituitary origin.
-
7 DRUG INTERACTIONS
Table 7 includes a list of drugs with clinically important drug interactions when administered concomitantly with ZOMACTON and instructions for preventing or managing them.
Table 7: Clinically Important Drug Interactions with ZOMACTON Glucocorticoids Clinical Impact: Microsomal enzyme 11β-hydroxysteroid dehydrogenase type 1 (11βHSD-1) is required for conversion of cortisone to its active metabolite, cortisol, in hepatic and adipose tissue. ZOMACTON inhibits 11βHSD-1. Consequently, individuals with untreated GH deficiency have relative increases in 11βHSD-1 and serum cortisol. Initiation of ZOMACTON may result in inhibition of 11βHSD-1 and reduced serum cortisol concentrations. Intervention: Patients treated with glucocorticoid replacement for hypoadrenalism may require an increase in their maintenance or stress doses following initiation of ZOMACTON [see Warnings and Precautions (5.8)]. Examples: Cortisone acetate and prednisone may be affected more than others since conversion of these drugs to their biologically active metabolites is dependent on the activity of 11βHSD-1. Pharmacologic Glucocorticoid Therapy and Supraphysiologic Glucocorticoid Treatment Clinical Impact: Pharmacologic glucocorticoid therapy and supraphysiologic glucocorticoid treatment may attenuate the growth promoting effects of ZOMACTON in pediatric patients. Intervention: Carefully adjust glucocorticoid replacement dosing in pediatric patients receiving glucocorticoid treatments to avoid both hypoadrenalism and an inhibitory effect on growth. Cytochrome P450-Metabolized Drugs Clinical Impact: Limited published data indicate that somatropin treatment increases cytochrome P450 (CP450)-mediated antipyrine clearance. ZOMACTON may alter the clearance of compounds known to be metabolized by CP450 liver enzymes. Intervention: Careful monitoring is advisable when ZOMACTON is administered in combination with drugs metabolized by CP450 liver enzymes. Oral Estrogen Clinical Impact: Oral estrogens may reduce the serum IGF-1 response to ZOMACTON. Intervention: Patients receiving oral estrogen replacement may require greater ZOMACTON dosages [see Dosage and Administration (2.2)]. Insulin and/or Other Hypoglycemic Agents Clinical Impact: Treatment with ZOMACTON may decrease insulin sensitivity, particularly at higher doses. Intervention: Patients with diabetes mellitus may require adjustment of their doses of insulin and/or other hypoglycemic agents [see Warnings and Precautions (5.4)]. -
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
The ZOMACTON 5 mg diluent contains benzyl alcohol. Because benzyl alcohol is rapidly metabolized by a pregnant woman, benzyl alcohol exposure in the fetus is unlikely. However, adverse reactions have occurred in premature neonates and low birth weight infants who received intravenously administered benzyl alcohol-containing drugs [see Warnings and Precautions (5.13) and Use in Specific Populations (8.4)]. Therefore, if ZOMACTON 5mg is needed during pregnancy, reconstitute with 0.9% sodium chloride injection, use only one dose per vial, and discard the unused portion, or use a ZOMACTON 10 mg benzyl alcohol-free formulation.
Limited published data do not report an association with somatropin and major birth defects, miscarriage, or adverse maternal or fetal outcomes when somatropin is used in pregnancy. Published reports indicate that somatropin does not cross the placenta. Animal reproduction studies have not been conducted with ZOMACTON.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
8.2 Lactation
Risk Summary
The ZOMACTON 5mg diluent contains benzyl alcohol. Because benzyl alcohol is rapidly metabolized by a lactating woman, benzyl alcohol exposure in the breastfed infant is unlikely. However, adverse reactions have occurred in premature neonates and low birth weight infants who received intravenously administered benzyl alcohol-containing drugs [see Warnings and Precautions (5.13) and Use in Specific Populations (8.4)]. If ZOMACTON 5mg is needed during lactation, reconstitute with 0.9% sodium chloride injection, use only one dose per vial, and discard after use or use a ZOMACTON 10 mg benzyl alcohol-free formulation.
There is no information regarding the presence of somatropin in human milk. Limited published data indicate that exogenous somatropin does not increase normal breastmilk concentrations of growth hormone. No adverse effects on the breastfed infant have been reported with somatropin. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for ZOMACTON and any potential adverse effects on the breastfed child from ZOMACTON or from the underlying maternal condition.
8.4 Pediatric Use
Safety and effectiveness of ZOMACTON in pediatric patients have been established in growth failure due to inadequate secretion of endogenous growth hormone, short stature associated with Turner syndrome, idiopathic short stature (ISS), short stature or growth failure in SHOX deficiency, and short stature in children born small for gestational age (SGA) with no catch-up growth by 2 years to 4 years of age.
Growth Failure due to Inadequate Secretion of Endogenous Growth Hormone
Safety and effectiveness of ZOMACTON have been established in pediatric patients with growth failure due to growth hormone deficiency based on data from a multi-center, open-label study in 164 pediatric patients conducted for a two-year period [see Clinical Studies (14.1)].
Short Stature associated with Turner Syndrome
Safety and effectiveness of ZOMACTON have been established in pediatric patients with short stature associated with Turner syndrome based on data from one long-term, randomized, open-label, Canadian multicenter, concurrently controlled study; two long-term, open-label multicenter, historically controlled US studies; and one long-term, randomized, US dose-response study with another somatropin product in 181 pediatric patients [see Clinical Studies (14.2)].
Idiopathic Short Stature (ISS)
Safety and effectiveness of ZOMACTON have been established in pediatric patients with ISS based on data from two randomized, multicenter studies, one placebo-controlled study and one dose-response study with another somatropin product in 310 pediatric patients [see Clinical Studies (14.3)].
Short Stature or Growth Failure in SHOX Deficiency
Safety and effectiveness of ZOMACTON have been established in pediatric patients with short stature or growth failure in SHOX deficiency based on data from a randomized, controlled, two-year, three-arm, open-label study with another somatropin product in 52 pediatric patients [see Clinical Studies (14.4)].
Short Stature in Children Born Small for Gestational Age (SGA) with No Catch-up Growth by Age 2 Years to 4 Years of Age
Safety and effectiveness of ZOMACTON have been established in pediatric patients with short stature born SGA with no catch-up growth based on data from two clinical studies with another somatropin product in 228 pediatric patients [see Clinical Studies (14.5)].
Toxicity (Gasping Syndrome) with Benzyl Alcohol-Preserved Solution
Serious adverse reactions including fatal reactions and the "gasping syndrome" occurred in premature neonates and infants in the intensive care unit who received drugs containing benzyl alcohol as a preservative. In these cases, benzyl alcohol dosages of 99 mg/kg/day to 234 mg/kg/day produced high levels of benzyl alcohol and its metabolites in the blood and urine (blood levels of benzyl alcohol were 0.61 mmol/L to 1.378 mmol/L). The maximal daily uptake of benzyl alcohol is 39 mg at ZOMACTON doses of 0.067 mg/kg/day.
Additional adverse reactions included gradual neurological deterioration, seizures, intracranial hemorrhage, hematologic abnormalities, skin breakdown, hepatic and renal failure, hypotension, bradycardia, and cardiovascular collapse. Preterm, low-birth weight infants may be more likely to develop these reactions because they may be less able to metabolize benzyl alcohol. When administering ZOMACTON 5 mg to infants, reconstitute with 0.9% sodium chloride injection, not with the diluent provided. Use only one dose per vial and discard the unused portion [see Warnings and Precautions (5.13)].
8.5 Geriatric Use
The safety and effectiveness of somatropin in patients aged 65 years and over has not been evaluated in clinical studies. Elderly patients may be more sensitive to the action of somatropin, and therefore may be more prone to development of adverse reactions. A lower starting dose and smaller dose increments should be considered for geriatric patients [see Dosage and Administration (2.3)].
- 9 DRUG ABUSE AND DEPENDENCE
- 10 OVERDOSAGE
-
11 DESCRIPTION
Somatropin is a human growth hormone (GH) produced by recombinant DNA technology using Escherichia coli. The protein is comprised of 191 amino acid residues and has a molecular weight of about 22,124 daltons. The amino acid sequence is identical to that of human growth hormone of pituitary origin.
ZOMACTON (somatropin) for injection is a sterile, white lyophilized powder, intended for subcutaneous injection after reconstitution with the accompanying diluent.
ZOMACTON 5 mg single-patient-use vial contains 5 mg of somatropin and mannitol (30 mg). Each 5 mg vial is co-packaged with a 5 mL vial of diluent containing Bacteriostatic Sodium Chloride Injection, USP, with 0.9% benzyl alcohol as a preservative, and Water for Injection, USP.
ZOMACTON 10 mg single-patient-use vial contains 10 mg of somatropin, mannitol (10 mg), dibasic sodium phosphate (1.415 mg), and monobasic sodium phosphate (0.608 mg). Each 10 mg vial is co-packaged with a 1 mL syringe of diluent containing Bacteriostatic Water for Injection, USP with 0.33% metacresol as a preservative.
Reconstituted solutions have a pH in the range of 7 to 9.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Somatropin binds to dimeric GH receptors located within the cell membranes of target tissue cells. This interaction results in intracellular signal transduction and subsequent induction of transcription and translation of GH-dependent proteins including IGF-1, IGF BP-3 and acid-labile subunit. Somatropin has direct tissue and metabolic effects or effects mediated indirectly by IGF-1, including stimulation of chondrocyte differentiation, and proliferation, stimulation of hepatic glucose output, protein synthesis, and lipolysis.
Somatropin stimulates skeletal growth in pediatric patients with GHD as a result of effects on the growth plates (epiphyses) of long bones. The stimulation of skeletal growth increases linear growth rate (height velocity) in most somatropin-treated pediatric patients. Linear growth is facilitated in part by increased cellular protein synthesis.
12.2 Pharmacodynamics
Subcutaneous administration of a single dose of 4 mg ZOMACTON in healthy subjects (n=54) with suppressed endogenous growth hormone results in an increased mean (SD) IGF-1 level from 233 (95) ng/mL predose to maximal level of 414 (120) ng/mL after approx. 24 hours. After 96 hours, the subjects displayed a mean (SD) IGF-1 concentration of 228 (74) ng/mL, comparable to the predose value.
12.3 Pharmacokinetics
Absorption — Somatropin has been studied following subcutaneous, and intravenous administration in adult healthy subjects. A single subcutaneous dose of 4 mg ZOMACTON in healthy subjects (n=54) with suppressed endogenous growth hormone resulted in a mean (SD) Cmax of 38.1 (19.3) ng/mL after approximately 4.5 hours. The absolute bioavailability of somatropin is approximately 70% after subcutaneous administration.
Distribution — The mean (SD) apparent volume of distribution of somatropin after single dose subcutaneous administration of 4 mg ZOMACTON in healthy subjects is 53.3 (24.6) L.
Elimination
Metabolism — Extensive metabolism studies have not been conducted. The metabolic fate of somatropin involves classical protein catabolism in both the liver and kidneys.
Excretion - In healthy subjects, mean somatropin clearance is 0.133 L/min following intravenous administration. The mean elimination half-life of intravenous somatropin is 0.42 hours, whereas subcutaneously administered somatropin has a mean half-life of 2.3 hours. The longer half-life observed after subcutaneous administration is due to slow absorption from the injection site. Urinary excretion of intact somatropin has not been measured.
Specific Populations
Geriatric patients — The pharmacokinetics of somatropin have not been studied in patients greater than 65 years of age.
Pediatric patients — The pharmacokinetics of somatropin in pediatric patients are similar to those of adults.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of ZOMACTON or other somatropins.
In a clinical trial with another recombinant growth hormone during the first 6 months of somatropin therapy in 314 naive patients, 1.6% developed specific antibodies to somatropin (binding capacity ≥0.02 mg/L). None had antibody concentrations which exceeded 2 mg/L. Throughout 8 years of this same study, two patients (0.6%) had binding capacity >2 mg/L. Neither patient demonstrated a decrease in growth velocity at or near the time of increased antibody production. It has been reported that growth attenuation from pituitary-derived GH may occur when antibody concentrations are >1.5 mg/L.
- 13 NONCLINICAL TOXICOLOGY
-
14 CLINICAL STUDIES
14.1 Pediatric Patients with Growth Failure Due to Inadequate Secretion of Endogenous Growth Hormone
A 2-year open-label, multi-center study with ZOMACTON was conducted in the United States and in Israel in 164 pediatric patients with short stature due to GHD. The subjects ranged in age from 2.1 to 17.7 years with a mean of 10.8 years. One hundred twenty (73%) of the subjects were male and 44 (27%) were female. Two subjects were Asian, 12 were Black, 130 were Caucasian, and 20 were categorized as 'other'.
The primary efficacy of the product was assessed by calculating height velocity. Mean cumulative increases in height velocity from baseline of 6.6, 4.6, and 6.3 cm/year were attained by 24 weeks of treatment (p < 0.01) in Naïve Type I (serum GH < 10 ng/mL in response to at least two provocative pharmacological tests), Naïve Type II (integrated GH level < 3.5 ng/mL with or without at least one serum GH ≥ 10 ng/mL), and Non-Naïve (treated with GH up to study Day 1, or previously treated and discontinued GH treatment at least 6 months prior to study Day 1) subjects, respectively. After 12 months of treatment, the mean cumulative increases in height velocity from baseline were 5.7, 4.4 and 5.3 cm/year (p= 0.01) in Naïve Type I, Naïve Type II, and Non-Naïve subjects, respectively.
14.2 Pediatric Patients with Short Stature Associated with Turner Syndrome
One long-term, randomized, open-label, Canadian multicenter, concurrently controlled study; two long-term, open-label multicenter, historically controlled US studies; and one long-term, randomized, US dose-response study were conducted to evaluate the efficacy of another somatropin product for treatment of short stature due to Turner syndrome.
The Canadian randomized study compared near-adult height outcomes for patients treated with another somatropin product to those of a concurrent control group who received no injections. Patients received a dosage of 0.3 mg/kg/week given in divided doses 6 times per week from a mean age of 12 years for a mean duration of 5 years. Puberty was induced with a standardized estrogen regimen initiated at 13 years of age for both treatment groups.
In two of the US studies, the effect of long-term treatment with another somatropin product (0.375 mg/kg/week given in divided doses either 3 times per week or daily) on adult height was determined by comparing adult heights in the treated patients with those of age-matched historical controls with Turner syndrome who received no growth-promoting therapy. Puberty was induced with a standardized estrogen regimen initiated after 14 years of age in one study; in the second study patients treated early (before 11 years of age) were randomized to begin pubertal induction at either age 12 (n=26) or 15 (n=29) years (conjugated estrogens, 0.3 mg escalating to 0.625 mg daily); those whose treatment was initiated after 11 years of age began estrogen replacement after 1 year of treatment with another somatropin product.
In a dose-response study in the US, patients were treated from a mean age of 11 years for a mean duration of 5 years with a weekly dosage of either 0.27 mg/kg or 0.36 mg/kg of another somatropin product administered in divided doses 3 or 6 times weekly.
In summary, patients with Turner syndrome (total n=181 from the 4 studies above) treated to adult height achieved statistically significant average height gains ranging from 5.0 to 8.3 cm (see Table 8).
Table 8: Height Gain Results* of Studies with Another Somatropin product in Patients with Turner Syndrome Study Group Study Design† Another Somatropin product Treated Number at Adult Height GH Age (yr) Estrogen Age (yr) GH Duration (yr) Adult Height Gain (cm)‡ - *
- Data shown are mean values.
- †
- RCT: randomized controlled trial; MHT: matched historical controlled trial; RDT: randomized dose-response trial.
- ‡
- Analysis of covariance vs. controls.
- §
- GH age <11 yr, estrogen age 15 yr.
- ¶
- GH age <11 yr, estrogen age 12 yr.
- #
- GH age >11 yr, estrogen at month 12.
- Þ
- Compared with historical data.
Canadian RCT 27 11.7 13 4.7 5.4 US 1 MHT 17 9.1 15.2 7.6 7.4 US 2 A§ MHT 29 9.4 15 6.1 8.3 B¶ 26 9.6 12.3 5.6 5.9 C# 51 12.7 13.7 3.8 5 US 3 RDT 31 11.1 8-13.5 5.3 ~5Þ 14.3 Pediatric Patients with Idiopathic Short Stature
Two randomized, multicenter studies, 1 placebo-controlled and 1 dose-response, were conducted with another somatropin product in pediatric patients with idiopathic short stature. The diagnosis of idiopathic short stature was made after excluding other known causes of short stature, as well as GH deficiency. The placebo-controlled study enrolled 71 pediatric patients (55 males, 16 females) 9 to 15 years old (mean age 12 ± 1.5 years), with short stature, 68 of whom received another somatropin product. Patients were predominately prepubertal (Tanner I, 45%) or in early puberty (Tanner II, 47%) at baseline. In this double-blind study, patients received subcutaneous injections of either another somatropin product 0.222 mg/kg/week, or placebo given in divided doses 3 times per week until height velocity decreased to ≤1.5 cm/year ("final height"). Final height measurements were available for 33 subjects (22 treated, 11 placebo) after a mean treatment duration of 4.4 years (range 0.1-9.1 years).
Results are presented in Table 9. The number of patients whose final height was above the 5th percentile of the general population height standard for age and sex was significantly greater in the treated group than the placebo group (41% vs. 0%), as was the number of patients who gained at least 1 SDS unit in height across the duration of the study (50% vs. 0%).
Table 9: Baseline Height Characteristics and Effect of Another Somatropin product on Final Height in Placebo-Controlled Study*,† Placebo
(n=11)
Mean (SD)Another Somatropin product
(n=22)
Mean (SD)Treatment Effect
Mean
(95% CI)p-value - *
- Abbreviations: BPH=baseline predicted height; CI=confidence interval; FH=final height; NA=not applicable; SDS=standard deviation score.
- †
- For final height population.
- ‡
- Between-group comparison was performed using analysis of covariance with baseline predicted height SDS as the covariate. Treatment effect is expressed as least squares mean (95% CI).
Baseline height SDS -2.75 (0.6) -2.7 (0.6) NA 0.77 BPH SDS -2.3 (0.8) -2.1 (0.7) NA 0.53 Final height SDS‡ -2.3 (0.6) -1.8 (0.8) 0.51 (0.10, 0.92) 0.017 FH SDS – baseline height SDS 0.4 (0.2) 0.9 (0.7) 0.51 (0.04, 0.97) 0.034 FH SDS – BPH SDS -0.1 (0.6) 0.3 (0.6) 0.46 (0.02, 0.89) 0.043 The dose-response study included 239 pediatric patients (158 males, 81 females), 5 to 15 years old, (mean age 10 ± 2 years). Mean ± SD baseline characteristics included: height SDS -3.21 ± 0.70, predicted adult height SDS -2.63 ± 1.08, and height velocity
SDS -1.09 ± 1.15. All but 3 patients were prepubertal. Patients were randomized to one of three treatment groups:
0.24 mg/kg/week; 0.24 mg/kg/week for 1 year, followed by 0.37 mg/kg/week; and 0.37 mg/kg/week. Height velocity was assessed by dose during the first 2 years of therapy. After completing the initial 2-year dose-response phase of the study, 50 patients were followed to final height.
Patients who received the dosage of 0.37 mg/kg/week had a significantly greater increase in mean height velocity after 2 years of treatment than patients who received 0.24 mg/kg/week (4.04 vs. 3.27 cm/year, p=0.003). The results are presented in Table 10. While no patient had height above the 5th percentile in any dosage group at baseline, 82% of the patients who received 0.37 mg/kg/week and 47% of the patients who received 0.24 mg/kg/week achieved final heights above the 5th percentile of the general population height standards (p=NS).
Table 10: Idiopathic Short Stature Study Results: Final Height Minus Baseline Predicted Height* Placebo-controlled Trial
3× per week dosingDose Response Trial
6× per week dosingPlacebo Another Somatropin product Another Somatropin product Another Somatropin product Another Somatropin product (n=10) 0.22 mg/kg
(n=22)0.24 mg/kg
(n=13)0.24/0.37 mg/kg
(n=13)0.37 mg/kg
(n=13)- *
- Abbreviations: FH=final height; PH=predicted height; CI=confidence interval; cm=centimeters.
FH – Baseline PH
Mean (95% CI), cm-0.7 (-3.6, 2.3) +2.2 (0.4, 3.9) +5.4 (2.8, 7.9) +6.7 (4.1, 9.2) +7.2 (4.6, 9.8) 14.4 Pediatric Patients with Short Stature or Growth Failure with SHOX Deficiency
A randomized, controlled, two-year, three-arm, open-label study was conducted to evaluate the efficacy of another somatropin product for treatment of short stature in pediatric patients with SHOX deficiency who were not GH–deficient. A total of 52 patients (24 male, 28 female) with SHOX deficiency, 3 to 12 years of age, were randomized to either a treated arm (27 patients; mean age 7 ± 2 years) or an untreated control arm (25 patients; mean age 8 ± 3 years). To determine the comparability of treatment effect between patients with SHOX deficiency and patients with Turner syndrome, the third study arm enrolled 26 patients with Turner syndrome, 5 to 12 years of age (mean age 8 ± 2 years), to treatment. All patients were prepubertal at study entry. Patients in the treated group(s) received daily subcutaneous injections of 0.05 mg/kg of another somatropin product, equivalent to 0.35 mg/kg/week. Patients in the untreated group received no injections. The results are presented in Table 11.
Table 11: Summary of Results in Patients with SHOX deficiency and Turner Syndrome SHOX Deficiency Turner Syndrome Untreated
(n=24)
Mean (SD)Another Somatropin product
(n=27)
Mean (SD)Treatment Difference*
Mean (95% CI)Another Somatropin product
(n=26)
Mean (SD)Height Velocity (cm/yr) 1st Year 5.2 (1.1) 8.7 (1.6)† +3.5 (2.8, 4.2) 8.9 (2.0) 2nd Year 5.4 (1.2) 7.3 (1.1)† +2.0 (1.3, 2.6) 7.0 (1.1) Height Gain (cm) Baseline to 1st Year +5.4 (1.2) +9.1 (1.5)† +3.7 (2.9, 4.5) +8.9 (1.9) Baseline to 2nd Year +10.5 (1.9) +16.4 (2.0)† +5.8 (4.6, 7.1) +15.7 (2.7) Height SDS Gain Baseline to 1st Year +0.1 (0.5) +0.7 (0.5)† +0.5 (0.3, 0.8) +0.8 (0.5) Baseline to 2nd Year +0.2 (0.5) +1.2 (0.7)† +1.0 (0.7, 1.3) +1.2 (0.2) Patients with height SDS > -2.0 at 2 years 1 (4%) 11 (41%)‡ -- 8 (31%) 14.5 Pediatric Patients Born Small for Gestational Age (SGA) Who Fail to Demonstrate Catch-up Growth by 2 Years to 4 Years of Age
A 2-year, open-label, multicenter, European study enrolled 193 prepubertal, non-GH deficient children with mean chronological age 7 ± 2 years (range: 3 to 12). Study entry criteria included birth weight <10th percentile and/or birth length SDS <-2 for gestational age, and height SDS for chronological age ≤-3. Exclusion criteria included syndromal conditions (e.g., Turner syndrome), chronic disease (e.g., diabetes mellitus), and tumor activity. The primary objective was to demonstrate that the increase from baseline in height SDS after 1 year of treatment with another somatropin product would be similar when treatment is administered according to an individually adjusted dose (IAD) regimen or a fixed high dose (FHD) regimen. The height increases would be considered similar if the lower bound of the 95% confidence interval (CI) for the mean difference between the groups (IAD – FHD) was greater than -0.5 height SDS. Pediatric patients were randomized to either a FHD (0.067 mg/kg/day; n=99) or an IAD treatment group (n=94). The initial dosage in the IAD treatment group was 0.035 mg/kg/day. The dosage was increased to 0.067 mg/kg/day in those patients in the IAD group whose 1-year height gain predicted at Month 3 was <0.75 height SDS (n=40) or whose actual height gain measured at Year 1 was <0.75 height SDS (n=11). Approximately 85% of the randomized patients completed 2 years of therapy.
The results are presented in Table 12. The results were similar when children who entered puberty during the study were removed from the analysis.
Table 12: Results for Height SDS and Change from Baseline in Height SDS at Year 1 and Year 2 After Treatment with Another Somatropin product of Short Children Born SGA Who Fail to Demonstrate Catch-up Growth* IAD Group
0.035 to 0.067 mg/kg/day
Mean (SD)FHD Group
0.067 mg/kg/day
Mean (SD)Between-Group
Difference
IAD - FHD†- *
- Abbreviations: IAD=individually adjusted dose; FHD=fixed high dose; SD=standard deviation; SDS=standard deviation score
- †
- Least squares mean difference ± standard error and 95% confidence interval based on ANCOVA model with treatment and gender as fixed effects, and baseline height SDS, baseline chronological age, baseline bone age, and mid-parental target height SDS as covariates.
- ‡
- Only children with actual height measurements were included in the Year 1 and Year 2 analyses.
Baseline (n=86) (n=93) -0.0 ± 0.1 -3.9 (0.6) -3.9 (0.7) (-0.2, 0.2) p-value = 0.95 Year 1 (n=86)‡ (n=93)‡ -0.3 ± 0.1 Height SDS -3.0 (0.7) -2.7 (0.7) (-0.4, -0.2) Change from baseline 0.9 (0.4) 1.1 (0.4) p-value <0.001 Year 2 (n=82)‡ (n=88)‡ -0.3 ± 0.1 Height SDS -2.5 (0.8) -2.2 (0.7) (-0.4, -0.1) Change from baseline 1.4 (0.5) 1.6 (0.5) p-value = 0.003 An open-label, multicenter, single arm study conducted in France, during which 35 prepubertal, non-GH deficient children were treated for 2 years with another somatropin product 0.067 mg/kg/day. Mean chronological age at baseline was 9 ± 0.9 years (range: 7 to 11). Additional study entry criteria included birth length SDS <-2 or <3rd percentile for gestational age, and height SDS for chronological age <-2. Exclusion criteria included syndromal conditions (e.g., Turner syndrome), chronic disease (e.g., diabetes mellitus), and any active disease. All 35 patients completed the study. Mean height SDS increased from a baseline value of -2.7 (SD 0.5) to -1.5 (SD 0.6) after 2 years of treatment.
14.6 Adult Patients with Growth Hormone Deficiency
Two studies in patients with adult-onset GH deficiency (total n=98) and two studies in adult patients with childhood-onset GH deficiency (total n=67) were designed to assess the effects of replacement therapy with another somatropin product. Adult-onset patients and childhood-onset patients differed by diagnosis (organic vs. idiopathic pituitary disease), body size (average vs. small [mean height and weight]), and age (mean 44 vs. 29 years). These four studies each included a 6-month randomized, blinded, placebo-controlled phase, during which approximately half of the patients received placebo injections, while the other half received injections with another somatropin product. The 6-month, double-blind phase was followed by 12 months of open-label somatropin treatment for all patients. The dosages of this other somatropin product for all studies were identical: 1 month of treatment at 0.00625 mg/kg/day followed by 0.0125 mg/kg/day for the next 5 months. The primary efficacy measures were body composition (lean body mass and fat mass) and lipid parameters. Lean body mass was determined by bioelectrical impedance analysis (BIA), validated with potassium 40. Body fat was assessed by BIA and sum of skinfold thickness. Lipid subfractions were analyzed by standard assay methods in a central laboratory.
In patients with adult-onset GH deficiency, treatment with another somatropin product (vs. placebo) resulted in an increase in mean lean body mass (2.59 vs. -0.22 kg, p<0.001) and a decrease in body fat (-3.27 vs. 0.56 kg, p<0.001). Similar changes were seen in childhood-onset GH deficient patients. Changes in lean body mass persisted throughout the 18-month period for both the adult-onset and childhood-onset groups; the changes in fat mass persisted in the childhood-onset group. Serum concentrations of high-density lipoprotein (HDL) cholesterol which were low at baseline (mean, 30.1 mg/mL and 33.9 mg/mL in adult-onset and childhood-onset patients, respectively) had normalized by the end of 18 months of treatment with this other somatropin product (mean change of 13.7 mg/dL and 11.1 mg/dL for the adult-onset and childhood-onset groups, respectively p<0.001).
-
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
ZOMACTON (somatropin) for injection is a white, lyophilized powder available as:
NDC ZOMACTON Diluent Additional Items NDC 55566-1801-1 5 mg single-patient-use vial 5 mL vial of diluent, Bacteriostatic Sodium Chloride Injection, USP with 0.9% benzyl alcohol NDC 55566-1901-1 10 mg single-patient-use vial 1 mL syringe of diluent, Bacteriostatic Water for Injection, USP with 0.33% metacresol 25G reconstitution needle 16.2 Storage and Handling
Before Reconstitution
Store ZOMACTON vials refrigerated at 36° to 46°F (2° to 8°C) in the original carton to protect from light. Avoid freezing the accompanying diluent.
After Reconstitution
If needed, store ZOMACTON 5 mg vials that have been reconstituted with co-packaged Bacteriostatic Sodium Chloride Injection, USP (with 0.9% benzyl alcohol) refrigerated at 36° to 46°F (2° to 8°C) for up to 14 days. Do not freeze.
If needed, store ZOMACTON 10 mg vials that have been reconstituted with co-packaged Bacteriostatic Water for Injection, USP (with 0.33% metacresol) refrigerated at 36° to 46°F (2° to 8°C) for up to 28 days. Do not freeze.
-
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Instructions for Use).
- Neoplasms – Advise childhood cancer survivors/caregivers that individuals treated with brain/head radiation are at increased risk of secondary neoplasms and as a precaution need to be monitored for recurrence. Advise patients/caregivers to report marked changes in behavior, onset of headaches, vision disturbances and/or changes in skin pigmentation or changes in the appearance of pre-existing nevi.
- Fluid Retention - Advise patients that fluid retention during ZOMACTON replacement therapy in adults may frequently occur. Inform patients of the clinical manifestations of fluid retention (e.g. edema, arthralgia, myalgia, nerve compression syndromes including carpal tunnel syndrome/paresthesias) and to report to their healthcare provider any of these signs or symptoms occur during treatment with ZOMACTON.
- Pancreatitis - Advise patients/caregivers that pancreatitis may develop and to report to their healthcare provider any new onset abdominal pain.
- Hypoadrenalism - Advise patients/caregivers who have or who are at risk for pituitary hormone deficiency(s) that hypoadrenalism may develop and to report to their healthcare provider if they experience hyperpigmentation, extreme fatigue, dizziness, weakness, or weight loss.
- Hypothyroidism - Advise patients/caregivers that undiagnosed/untreated hypothyroidism may prevent an optimal response to ZOMACTON. Advise patients/caregivers they may require periodic thyroid function tests.
- Intracranial Hypertension - Advise patients/caregivers to report to their healthcare provider any visual changes, headache, and nausea and/or vomiting.
- Hypersensitivity Reactions – Advise patients/caregivers that serious systemic hypersensitivity reactions (anaphylaxis and angioedema) are possible and that prompt medical attention should be sought if an allergic reaction occurs.
- Glucose Intolerance/ Diabetes Mellitus – Advise patients/caregivers that new onset impaired glucose intolerance/diabetes mellitus or exacerbation of preexisting diabetes mellitus can occur and monitoring of blood glucose during treatment with ZOMACTON may be needed.
- Females of Reproductive Potential – Instruct patients to inform their healthcare provider if they are pregnant or planning to become pregnant as they may potentially require the use of a different formulation of ZOMACTON.
- SPL UNCLASSIFIED SECTION
-
Instructions for Use
ZOMACTON®
(zoh-MACK-ton)
[somatropin]
for InjectionRead the Instructions for Use that come with your ZOMACTON® before you start using it and each time you get a refill. There may be new information. This leaflet does not take the place of talking to your healthcare provider about your medical condition or treatment. Before you use ZOMACTON for the first time, make sure your healthcare provider shows you the right way to use it.
Supplies needed for your ZOMACTON Injection
-
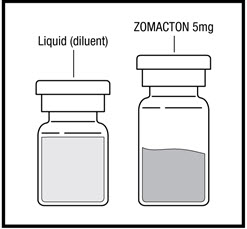
ZOMACTON 5mg (See Figure A) containing:
- 1 vial of ZOMACTON 5mg growth hormone in a powder
- 1 vial of liquid (diluent) containing Bacteriostatic Sodium Chloride Injection, USP (with 0.9% benzyl alcohol as a preservative) (5mL). This is used to mix your ZOMACTON 5mg.
or
-
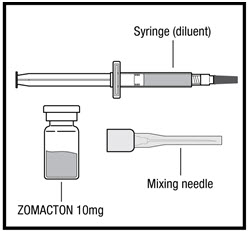
ZOMACTON 10mg (See Figure B) containing:
- 1 vial of ZOMACTON 10mg growth hormone in a powder
- 1 syringe of liquid (diluent) containing Bacteriostatic Water for Injection, USP (with 0.33% Metacresol as a preservative) (1 mL). This is used to mix your ZOMACTON 10mg.
- 25 gauge mixing needle
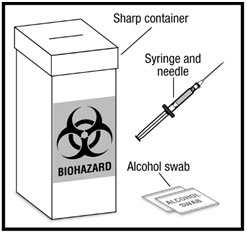
The following additional supplies (See Figure C) will be needed:
- Syringe and needle for injection. Your healthcare provider will tell you the size of the syringe and needle to use.
- Alcohol swab
- Puncture-resistant container (See Step 4: Disposing of used syringes, needles, and vials)
Preparing for Your ZOMACTON Injection
- Place the supplies you will need on a clean, flat surface in a well-lit area.
- Wash your hands thoroughly with soap and water.
Important: The liquids are different for the 5 mg and 10 mg vials.
- Do not use the 5 mg liquid with the 10 mg ZOMACTON.
- Do not use the 10 mg liquid with the 5 mg ZOMACTON.
Preparing ZOMACTON 5mg Liquid for Injection:
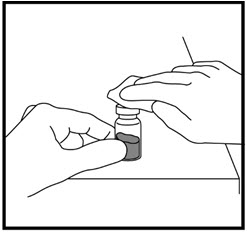
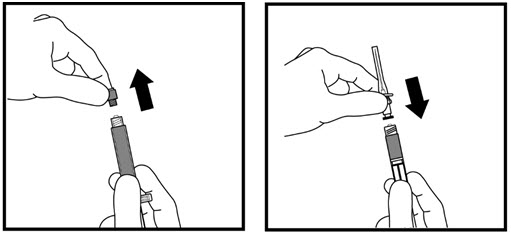
- Remove the hard plastic cap from the top of the liquid vial by gently pushing up on the edge of the cap (See Figure D). Do not remove the rubber stopper.
- Use an alcohol swab to wipe off the top of the liquid vial (See Figure E). After cleaning, do not touch the rubber stopper.
- Remove the needle cap from the syringe while making sure you do not touch the needle (See Figure F). Do not throw away the needle cap.
- Hold the barrel of the syringe with 1 hand and pull back on the plunger with the other hand until you have drawn up the amount of air that is the same as the amount of liquid your healthcare provider has prescribed (See Figure G).
- Insert the needle into the liquid vial through the center of the clean rubber stopper. Push down on the plunger until all the air is released into the vial (See Figure H).
- Hold the vial with 1 hand and carefully turn the vial upside down, making sure the syringe needle stays in the vial. The tip of the needle should be below the surface of the liquid.
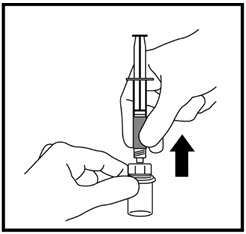
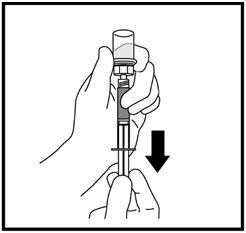
- With your other hand, gently pull back the plunger until the amount of liquid your healthcare provider prescribed is in the syringe (See Figure I).
When the syringe is correctly filled with the liquid, remove the syringe and needle from the vial and recap the needle.
Preparing ZOMACTON 10mg Liquid for Injection:
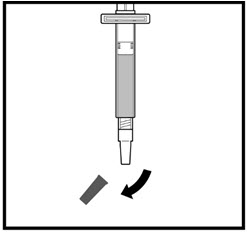
- Remove the syringe tip cap from the top of the pre-filled liquid syringe and attach the 25G mixing needle that comes with your ZOMACTON (See Figure J).
Diluting Your ZOMACTON
- Only use the liquid that comes with the 5 mg ZOMACTON to mix the 5 mg growth hormone. Only use the liquid that comes with the 10 mg ZOMACTON to mix the 10 mg growth hormone.
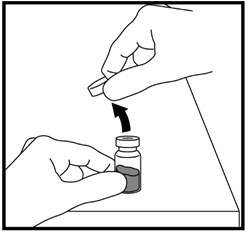
- Remove the hard plastic cap of the growth hormone vial (See Figure K).
- Clean the top of the growth hormone vial with an alcohol swab (See Figure L).
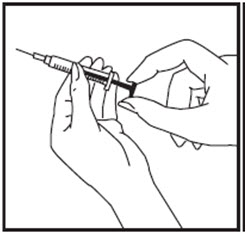
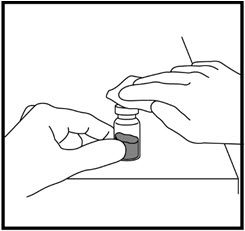
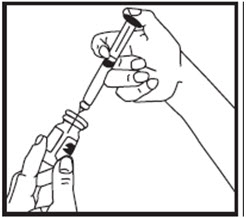
- Remove the needle cap from the syringe filled with liquid and insert the needle into the center of the rubber stopper on the growth hormone vial (See Figure M).
- Point the needle toward the side of the vial and slowly push the plunger so that the liquid squirts onto the side of the vial and not directly onto the powder.
- When all the liquid is in the growth hormone vial, remove the needle from the vial (See Figure N).
- Recap the needle and throw away the syringe.
Mixing ZOMACTON
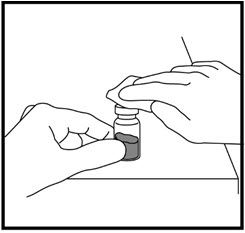
- Hold the vial between your hands and gently roll it until the mixture is clear. Do not shake the vial. Your ZOMACTON is ready for injection.
- Sometimes the vial may need to sit a few seconds before the mixture becomes clear. Do not use the mixture in the vial if it remains cloudy or you see particles floating in the mixture. If air bubbles appear, let the growth hormone sit for a while until they disappear.
- Write the date you mixed the growth hormone on the vial label. The 5 mg vial must be used within 14 days. The 10 mg vial must be used within 28 days.
- Store your mixed growth hormone and all unopened vials of growth hormone in the refrigerator at 36°F to 46°F (2°C to 8°C). Do not freeze.
Step 1: Preparing the Injection
You are now ready for your ZOMACTON injection.
- Wash your hands thoroughly with soap and water.
- Check that the vial of growth hormone you are using is clear and that the date of mixing is within 14 days if you are using ZOMACTON 5 mg or 28 days if you are using ZOMACTON 10 mg.
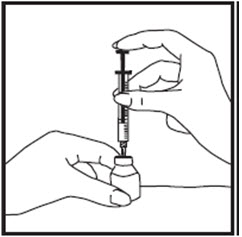
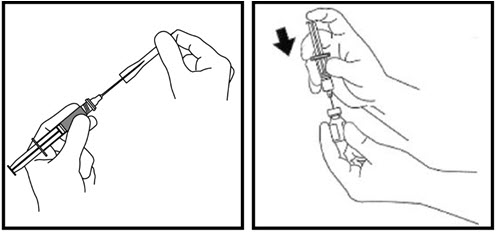
- Clean the top of the growth hormone vial with an alcohol swab. Do not touch the rubber stopper after cleaning (See Figure O).
- Remove the needle cap from the syringe and insert the needle into the center of the rubber stopper on the growth hormone vial (See Figure P).
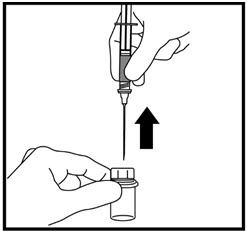
- Gently pull back the plunger until the amount of growth hormone solution your healthcare provider has prescribed is in the syringe (See Figure Q).
- Remove the needle from the vial when the syringe is correctly filled with the solution (See Figure R). Recap the needle.
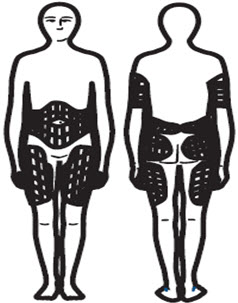
Step 2: Choosing an Injection Site
- There are different sites you can use for your injections. These sites should be rotated (See Figure S).
If you notice any of the following signs, contact your healthcare provider:
- A lump, bruise or redness at the injection site that does not go away.
- Any sign of infection at the injection site (pus, redness, heat or persistent pain).
- Severe, sharp pain or ache at injection site that does not go away.
- Rash at the injection site.
Step 3: Injecting ZOMACTON
Using a circular motion, clean the injection site with an alcohol swab, starting at the injection site and moving outward about 2 inches. Let the skin air dry.
- Check that the correct dose is in the syringe.
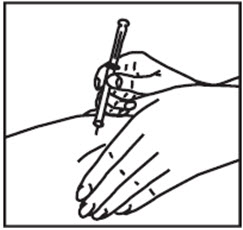
- Remove the needle cap. Hold the syringe like a pencil in 1 hand.
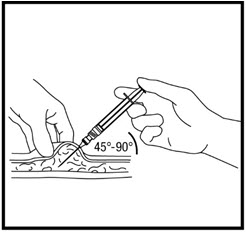
- With your free hand, pinch the skin around the site with the thumb and forefinger of the other hand (See Figure T). Quickly insert the needle into the skin at a 45° - 90° angle with a quick, dart-like motion.
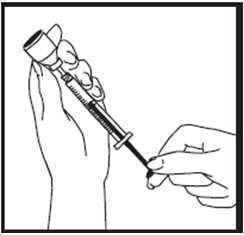
- Holding the syringe in place, pull back a little on the plunger and check to see if any blood flows into the syringe (See Figure U). If you see blood in the syringe, it means that you have entered a blood vessel. Do not inject ZOMACTON. Withdraw the needle. Throw away the syringe and needle in a puncture-resistant container. Do not use the same syringe or any of the other supplies that you used for this injection. Repeat the steps to prepare a new syringe for injection. Choose and clean a new injection site.
- If no blood appears in the syringe, slowly push down plunger all the way until the syringe is completely empty (See Figure V).
- Quickly remove the needle from the skin and apply pressure to the injection site with a dry sterile gauze pad or cotton ball. A drop of blood may appear. Apply a small bandage if needed. Throw away the needle and syringe in your puncture-resistant disposal container.
- Do not share your syringes, needles, or vials with anyone else. You may give them or get an infection from them.
Step 4: Disposing of used syringes, needles, and vials
- To prevent needle-stick injury and spread of infection, do not try to re-cap the needle.
- Place used needles, syringes, and vials in a closeable, puncture-resistant container. You may use a sharps container (such as a red biohazard container), hard plastic container (such as a detergent bottle), or metal container (such as an empty coffee can). Do not use glass or clear plastic containers. Ask your healthcare provider for instructions on the right way to throw away (dispose of) the container. There may be state and local laws about how you should throw away used needles and syringes.
- If you do not have a FDA-cleared sharps disposal container, you may use a household container that is:
- made of a heavy-duty plastic,
- can be closed with a tight-fitting, puncture-resistant lid, without sharps being able to come out,
- upright and stable during use,
- leak-resistant, and
- properly labeled to warn of hazardous waste inside the container.
- When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and syringes. For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal
- Do not throw used needles, syringes, or vials in your household trash or recycle.
- Keep the disposal container, needles, syringes, and vials of ZOMACTON out of reach of children.
This Instructions for Use has been approved by the Food and Drug Administration.
MANUFACTURED BY:
FERRING PHARMACEUTICALS INC.
PARSIPPANY, NJ 07054U.S. License No. 2112
XXXXXXXXXX
Rev. 4/2024 -
ZOMACTON 5mg (See Figure A) containing:
-
PRINCIPAL DISPLAY PANEL - 5 mg Kit Carton
NDC 55566-1801-1
FERRING
PHARMACEUTICALSZOMACTON®
(somatropin) for Injection5 mg per vial
For subcutaneous use only
1 single-patient-use vial
Reconstitute with Bacteriostatic Sodium Chloride Injection,
USP containing 0.9% benzyl alcohol. Gently swirl. Do not shake.Storage before and after reconstitution: Refrigerate at 2°C
to 8°C (36°C to 46°F). Do not freeze. Protect from light.
After reconstitution: Use within 14 days.Rx only

-
PRINCIPAL DISPLAY PANEL - 10 mg Kit Carton - 1901
NDC 55566-1901-1
FERRING
PHARMACEUTICALSZOMACTON®
(somatropin) for Injection10 mg per vial
For subcutaneous use only
1 single-patient-use vial
Reconstitute with Bacteriostatic Water for injection, USP containing 0.33% metacresol.
Gently swirl. Do not shake.Storage before and after reconstitution: Refrigerate at 2°C to 8°C (36°F to 46°F).
Do not freeze. Protect from light. After reconstitution: Use within 28 days.Rx only

-
PRINCIPAL DISPLAY PANEL - 10 mg Kit Carton - 1902
NDC 55566-1902-1
FERRING
PHARMACEUTICALSZOMACTON®10mg
(somatropin) for InjectionFor subcutaneous use only
Reconstitute with the prefilled diluent syringe (Bacteriostatic water for
injection, containing 0.33% metacresol as an antimicrobial preservative).Refrigerate at 2° to 8°C (36° to 46°F).
Reconstituted vials should be refrigerated and used within 28 days.
Do not freeze. Protect from light.
For use with needle-free ZOMA-Jet® 10 only
Rx only

-
INGREDIENTS AND APPEARANCE
ZOMACTON
somatropin kitProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:55566-1801 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:55566-1801-1 1 in 1 CARTON 05/07/2015 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 VIAL 1 Part 2 1 VIAL 5 mL Part 1 of 2 ZOMACTON
somatropin injection, powder, lyophilized, for solutionProduct Information Route of Administration SUBCUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SOMATROPIN (UNII: NQX9KB6PCL) (SOMATROPIN - UNII:NQX9KB6PCL) SOMATROPIN 5 mg Inactive Ingredients Ingredient Name Strength MANNITOL (UNII: 3OWL53L36A) 30 mg Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 1 in 1 VIAL; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA019774 05/07/2015 Part 2 of 2 STERILE DILUENT
diluent solutionProduct Information Route of Administration SUBCUTANEOUS Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) BENZYL ALCOHOL (UNII: LKG8494WBH) SODIUM CHLORIDE (UNII: 451W47IQ8X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 5 mL in 1 VIAL; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA019774 05/07/2015 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA019774 05/07/2015 ZOMACTON
somatropin kitProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:55566-1901 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:55566-1901-1 1 in 1 CARTON 05/07/2015 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 VIAL 1 Part 2 1 SYRINGE, PLASTIC 1 mL Part 1 of 2 ZOMACTON
somatropin injection, powder, lyophilized, for solutionProduct Information Route of Administration SUBCUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SOMATROPIN (UNII: NQX9KB6PCL) (SOMATROPIN - UNII:NQX9KB6PCL) SOMATROPIN 10 mg Inactive Ingredients Ingredient Name Strength MANNITOL (UNII: 3OWL53L36A) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 1 in 1 VIAL; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA019774 05/07/2015 Part 2 of 2 STERILE DILUENT
diluent injection, solutionProduct Information Route of Administration SUBCUTANEOUS Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) METACRESOL (UNII: GGO4Y809LO) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 1 mL in 1 SYRINGE, PLASTIC; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA019774 05/07/2015 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA019774 05/07/2015 ZOMACTON
somatropin kitProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:55566-1902 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:55566-1902-1 1 in 1 CARTON 01/30/2018 05/31/2022 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 VIAL 1 Part 2 1 SYRINGE, PLASTIC 1 mL Part 1 of 2 ZOMACTON
somatropin injection, powder, lyophilized, for solutionProduct Information Route of Administration SUBCUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SOMATROPIN (UNII: NQX9KB6PCL) (SOMATROPIN - UNII:NQX9KB6PCL) SOMATROPIN 10 mg Inactive Ingredients Ingredient Name Strength MANNITOL (UNII: 3OWL53L36A) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 1 in 1 VIAL; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA019774 01/30/2018 Part 2 of 2 STERILE DILUENT
diluent solutionProduct Information Route of Administration SUBCUTANEOUS Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) METACRESOL (UNII: GGO4Y809LO) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 1 mL in 1 SYRINGE, PLASTIC; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA019774 01/30/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA019774 01/30/2018 05/31/2022 Labeler - Ferring Pharmaceuticals Inc. (103722955) Establishment Name Address ID/FEI Business Operations Wasserburger Arzneimittelwerk GmbH 326482247 manufacture(55566-1801, 55566-1901, 55566-1902) , pack(55566-1901) Establishment Name Address ID/FEI Business Operations Ferring GmbH 328609615 manufacture(55566-1801) , pack(55566-1801) , ANALYSIS(55566-1801, 55566-1901) Establishment Name Address ID/FEI Business Operations Ferring International Center SA 481210362 pack(55566-1901, 55566-1801) Establishment Name Address ID/FEI Business Operations Bio-Technology General (Israel) Ltd 600004527 api manufacture(55566-1801, 55566-1901, 55566-1902) , pack(55566-1801) , ANALYSIS(55566-1801, 55566-1901)