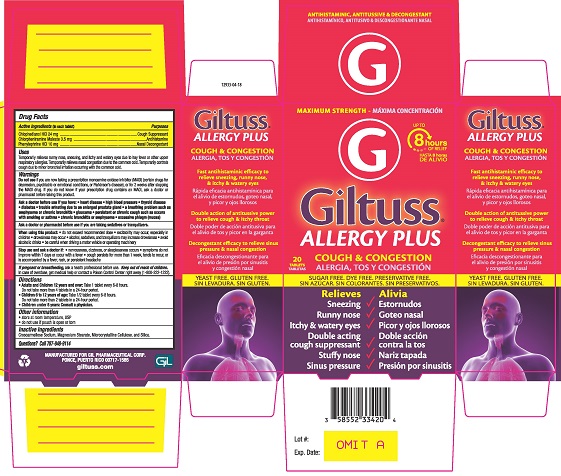
Label: GILTUSS ALLERGY PLUS- chlophedianol hcl, chlorpheniramine maleate, phenylephrine hcl tablet
- NDC Code(s): 58552-334-20
- Packager: Gil Pharmaceutical Corp
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated October 24, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- PURPOSE
- INDICATIONS & USAGE
-
WARNINGS
Warnings
Do not use if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have:
- heart disease
- high blood pressure
- thyroid disease
- diabetes
- trouble urinating due to an enlarged prostate gland
- a breathing problem such as emphysema or chronic bronchitis
- glaucoma
- persistent or chronic cough such as occurs with smoking, asthma
- chronic bronchitis, or emphysema
- excessive phlegm (mucus)
When using this product:
- do not exceed recommended dose
- excitability may occur, especially in children
- drowsiness may occur
- alcohol, sedatives, and tranquilizers may increase drowsiness
- avoid alcoholic drinks
- be careful when driving a motor vehicle or operating machinery
- DOSAGE & ADMINISTRATION
- STORAGE AND HANDLING
- INACTIVE INGREDIENT
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
GILTUSS ALLERGY PLUS
chlophedianol hcl, chlorpheniramine maleate, phenylephrine hcl tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:58552-334 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE HYDROCHLORIDE 10 mg CHLORPHENIRAMINE MALEATE (UNII: V1Q0O9OJ9Z) (CHLORPHENIRAMINE - UNII:3U6IO1965U) CHLORPHENIRAMINE MALEATE 3.5 mg CHLOPHEDIANOL HYDROCHLORIDE (UNII: 69QQ58998Y) (CHLOPHEDIANOL - UNII:42C50P12AP) CHLOPHEDIANOL HYDROCHLORIDE 24 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) MAGNESIUM STEARATE (UNII: 70097M6I30) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) Product Characteristics Color white Score no score Shape ROUND Size 8mm Flavor Imprint Code GIL;323 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58552-334-20 10 in 1 BOX 04/01/2021 1 2 in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M012 04/01/2021 Labeler - Gil Pharmaceutical Corp (176826592)