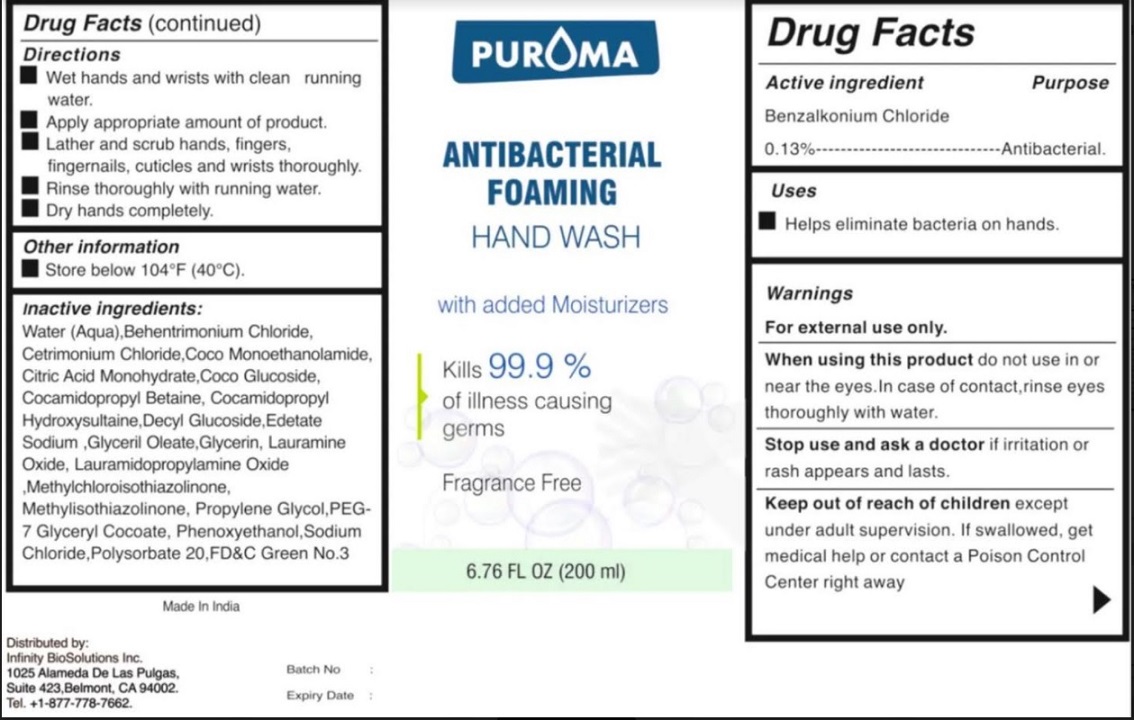
Label: PUROMA ANTIBACTERIAL FOAMING HAND WASH FRAGRANCE FREE- benzalkonium chloride liquid
-
NDC Code(s):
80948-005-01,
80948-005-02,
80948-005-03,
80948-005-04, view more80948-005-05, 80948-005-06, 80948-005-07, 80948-005-08, 80948-005-09, 80948-005-10, 80948-005-11, 80948-005-12, 80948-005-13, 80948-005-14, 80948-005-15, 80948-005-21, 80948-005-22, 80948-005-23, 80948-005-24, 80948-005-25, 80948-005-26, 80948-005-27, 80948-005-28, 80948-005-29, 80948-005-30, 80948-005-31, 80948-005-32, 80948-005-33, 80948-005-34, 80948-005-35, 80948-005-36, 80948-005-37, 80948-005-38, 80948-005-39, 80948-005-40, 80948-005-41, 80948-005-42, 80948-005-43, 80948-005-44, 80948-005-45, 80948-005-46, 80948-005-47, 80948-005-48, 80948-005-49, 80948-005-50, 80948-005-51, 80948-005-52
- Packager: ZENITH MICRO CONTROL
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 13, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredient
- Purpose
- Uses
- Warnings
- Keep out of reach of children
- Directions
-
Inactive ingredients
Water,Glycerin, Methylchloroisothiazolinone, Methylisothiazolinone, Cetrimonium Chloride, Lauramidopropylamine Oxide , Coco Monoethanolamide, Propylene Glycol, Citric Acid Monohydrate, Phenoxyethanol, Edetate Sodium , Decyl Glucoside, Coco Glucoside, Glyceril Oleate, Cocamidopropyl Betaine, Lauramine Oxide, Behentrimonium Chloride, PEG-7 Glyceryl Cocoate, Cocamidopropyl Hydroxysultaine, Polysorbate 20, Sodium Chloride, FD&C Green No.3.
- Other information
- Product label
-
INGREDIENTS AND APPEARANCE
PUROMA ANTIBACTERIAL FOAMING HAND WASH FRAGRANCE FREE
benzalkonium chloride liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:80948-005 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 130 mg in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) GLYCERIN (UNII: PDC6A3C0OX) METHYLCHLOROISOTHIAZOLINONE (UNII: DEL7T5QRPN) METHYLISOTHIAZOLINONE (UNII: 229D0E1QFA) CETRIMONIUM CHLORIDE (UNII: UC9PE95IBP) LAURAMIDOPROPYLAMINE OXIDE (UNII: I6KX160QTV) COCO MONOETHANOLAMIDE (UNII: C80684146D) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) PHENOXYETHANOL (UNII: HIE492ZZ3T) EDETATE SODIUM (UNII: MP1J8420LU) SODIUM CHLORIDE (UNII: 451W47IQ8X) DECYL GLUCOSIDE (UNII: Z17H97EA6Y) COCO GLUCOSIDE (UNII: ICS790225B) GLYCERYL OLEATE (UNII: 4PC054V79P) COCAMIDOPROPYL BETAINE (UNII: 5OCF3O11KX) LAURAMINE OXIDE (UNII: 4F6FC4MI8W) BEHENTRIMONIUM CHLORIDE (UNII: X7GNG3S47T) PEG-7 GLYCERYL COCOATE (UNII: VNX7251543) COCAMIDOPROPYL HYDROXYSULTAINE (UNII: 62V75NI93W) POLYSORBATE 20 (UNII: 7T1F30V5YH) FD&C GREEN NO. 3 (UNII: 3P3ONR6O1S) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:80948-005-01 4 in 1 PACKAGE 03/10/2021 1 70 mL in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:80948-005-02 6 in 1 PACKAGE 03/10/2021 2 70 mL in 1 BOTTLE; Type 0: Not a Combination Product 3 NDC:80948-005-03 8 in 1 PACKAGE 03/10/2021 3 70 mL in 1 BOTTLE; Type 0: Not a Combination Product 4 NDC:80948-005-04 12 in 1 PACKAGE 03/10/2021 4 70 mL in 1 BOTTLE; Type 0: Not a Combination Product 5 NDC:80948-005-05 24 in 1 PACKAGE 03/10/2021 5 70 mL in 1 BOTTLE; Type 0: Not a Combination Product 6 NDC:80948-005-06 4 in 1 PACKAGE 03/10/2021 6 100 mL in 1 BOTTLE; Type 0: Not a Combination Product 7 NDC:80948-005-07 6 in 1 PACKAGE 03/10/2021 7 100 mL in 1 BOTTLE; Type 0: Not a Combination Product 8 NDC:80948-005-08 8 in 1 PACKAGE 03/10/2021 8 100 mL in 1 BOTTLE; Type 0: Not a Combination Product 9 NDC:80948-005-09 12 in 1 PACKAGE 03/10/2021 9 100 mL in 1 BOTTLE; Type 0: Not a Combination Product 10 NDC:80948-005-10 24 in 1 PACKAGE 03/10/2021 10 100 mL in 1 BOTTLE; Type 0: Not a Combination Product 11 NDC:80948-005-11 4 in 1 PACKAGE 03/10/2021 11 150 mL in 1 BOTTLE; Type 0: Not a Combination Product 12 NDC:80948-005-12 6 in 1 PACKAGE 03/10/2021 12 150 mL in 1 BOTTLE; Type 0: Not a Combination Product 13 NDC:80948-005-13 8 in 1 PACKAGE 03/10/2021 13 150 mL in 1 BOTTLE; Type 0: Not a Combination Product 14 NDC:80948-005-14 12 in 1 PACKAGE 03/10/2021 14 150 mL in 1 BOTTLE; Type 0: Not a Combination Product 15 NDC:80948-005-15 24 in 1 PACKAGE 03/10/2021 15 150 mL in 1 BOTTLE; Type 0: Not a Combination Product 16 NDC:80948-005-21 4 in 1 PACKAGE 03/10/2021 16 200 mL in 1 BOTTLE; Type 1: Convenience Kit of Co-Package 17 NDC:80948-005-22 6 in 1 PACKAGE 03/10/2021 17 200 mL in 1 BOTTLE; Type 1: Convenience Kit of Co-Package 18 NDC:80948-005-23 8 in 1 PACKAGE 03/10/2021 18 200 mL in 1 BOTTLE; Type 1: Convenience Kit of Co-Package 19 NDC:80948-005-24 12 in 1 PACKAGE 03/10/2021 19 200 mL in 1 BOTTLE; Type 1: Convenience Kit of Co-Package 20 NDC:80948-005-25 24 in 1 PACKAGE 03/10/2021 20 200 mL in 1 BOTTLE; Type 1: Convenience Kit of Co-Package 21 NDC:80948-005-26 4 in 1 PACKAGE 03/10/2021 21 220 mL in 1 BOTTLE; Type 0: Not a Combination Product 22 NDC:80948-005-27 6 in 1 PACKAGE 03/10/2021 22 220 mL in 1 BOTTLE; Type 0: Not a Combination Product 23 NDC:80948-005-28 8 in 1 PACKAGE 03/10/2021 23 220 mL in 1 BOTTLE; Type 0: Not a Combination Product 24 NDC:80948-005-29 12 in 1 PACKAGE 03/10/2021 24 220 mL in 1 BOTTLE; Type 0: Not a Combination Product 25 NDC:80948-005-30 24 in 1 PACKAGE 03/10/2021 25 220 mL in 1 BOTTLE; Type 0: Not a Combination Product 26 NDC:80948-005-31 2 in 1 PACKAGE 03/10/2021 26 250 mL in 1 BOTTLE; Type 0: Not a Combination Product 27 NDC:80948-005-32 4 in 1 PACKAGE 03/10/2021 27 250 mL in 1 BOTTLE; Type 0: Not a Combination Product 28 NDC:80948-005-33 6 in 1 PACKAGE 03/10/2021 28 250 mL in 1 BOTTLE; Type 0: Not a Combination Product 29 NDC:80948-005-34 8 in 1 PACKAGE 03/10/2021 29 250 mL in 1 BOTTLE; Type 0: Not a Combination Product 30 NDC:80948-005-35 12 in 1 PACKAGE 03/10/2021 30 250 mL in 1 BOTTLE; Type 0: Not a Combination Product 31 NDC:80948-005-36 24 in 1 PACKAGE 03/10/2021 31 250 mL in 1 BOTTLE; Type 0: Not a Combination Product 32 NDC:80948-005-37 2 in 1 PACKAGE 03/10/2021 32 300 mL in 1 BOTTLE; Type 0: Not a Combination Product 33 NDC:80948-005-38 4 in 1 PACKAGE 03/10/2021 33 300 mL in 1 BOTTLE; Type 0: Not a Combination Product 34 NDC:80948-005-39 6 in 1 PACKAGE 03/10/2021 34 300 mL in 1 BOTTLE; Type 0: Not a Combination Product 35 NDC:80948-005-40 8 in 1 PACKAGE 03/10/2021 35 300 mL in 1 BOTTLE; Type 0: Not a Combination Product 36 NDC:80948-005-41 12 in 1 PACKAGE 03/10/2021 36 300 mL in 1 BOTTLE; Type 0: Not a Combination Product 37 NDC:80948-005-42 24 in 1 PACKAGE 03/10/2021 37 300 mL in 1 BOTTLE; Type 0: Not a Combination Product 38 NDC:80948-005-43 2 in 1 PACKAGE 03/10/2021 38 350 mL in 1 BOTTLE; Type 0: Not a Combination Product 39 NDC:80948-005-44 4 in 1 PACKAGE 03/10/2021 39 350 mL in 1 BOTTLE; Type 0: Not a Combination Product 40 NDC:80948-005-45 6 in 1 PACKAGE 03/10/2021 40 350 mL in 1 BOTTLE; Type 0: Not a Combination Product 41 NDC:80948-005-46 8 in 1 PACKAGE 03/10/2021 41 350 mL in 1 BOTTLE; Type 0: Not a Combination Product 42 NDC:80948-005-47 12 in 1 PACKAGE 03/10/2021 42 350 mL in 1 BOTTLE; Type 0: Not a Combination Product 43 NDC:80948-005-48 24 in 1 PACKAGE 03/10/2021 43 350 mL in 1 BOTTLE; Type 0: Not a Combination Product 44 NDC:80948-005-49 5000 mL in 1 CAN; Type 0: Not a Combination Product 03/10/2021 45 NDC:80948-005-50 50000 mL in 1 CAN; Type 0: Not a Combination Product 03/10/2021 46 NDC:80948-005-51 200000 mL in 1 DRUM; Type 0: Not a Combination Product 03/10/2021 47 NDC:80948-005-52 250 mL in 1 BOTTLE; Type 0: Not a Combination Product 10/21/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M003 03/10/2021 Labeler - ZENITH MICRO CONTROL (915625571) Establishment Name Address ID/FEI Business Operations ZENITH MICRO CONTROL 915625571 manufacture(80948-005)