Label: BUSCOPAN- n-butylscopolammonium bromide injection
- NDC Code(s): 0010-3701-01
- Packager: Boehringer Ingelheim Animal Health USA Inc.
- Category: PRESCRIPTION ANIMAL DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Animal Drug Application
Drug Label Information
Updated March 27, 2025
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Caution:
-
Description:
BUSCOPAN Injectable Solution is an antispasmodic (spasmolytic) and anticholinergic drug which suppresses spasms of the digestive system.
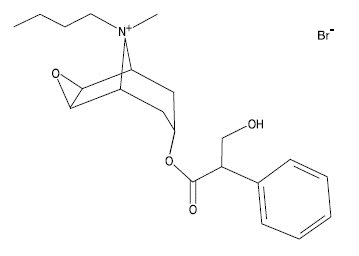
The chemical name for the active constituent of BUSCOPAN is N-butylscopolammonium bromide. It is a water soluble, white crystalline substance with a molecular weight of 440.40. Each mL of BUSCOPAN contains 20 mg N-butylscopolammonium bromide, 1.8 mg methylparaben, 0.2 mg propylparaben, 6.0 mg sodium chloride, and water for injection. The chemical structure is:
- Indications:
-
Dosage and Administration:
Administer a single injection of 0.3 mg/kg body weight (0.14 mg/lb), slowly IV. This is equivalent to 30 mg N-butylscopolammonium bromide per 100 kg (220 pounds) bodyweight or 1.5 mL of BUSCOPAN per 100 kg (220 pounds) bodyweight.
Dosing Table (Dosage 0.68 mL/100 lbs.) Body Weight (nearest 100 pounds)
Dose (mL)
Body Weight (nearest 100 pounds)
Dose (mL)
Body Weight (nearest 100 pounds)
Dose (mL)
200
1.4
1000
6.8
1800
12.2
300
2.0
1100
7.5
1900
12.9
400
2.7
1200
8.2
2000
13.6
500
3.4
1300
8.8
2100
14.3
600
4.1
1400
9.5
2200
15.0
700
4.8
1500
10.2
2300
15.6
800
5.4
1600
10.9
2400
16.3
900
6.1
1700
11.6
2500
17.0
- Contraindications:
- Warnings:
-
Precautions:
BUSCOPAN is not recommended for use in nursing foals or in pregnant or lactating mares, as safety has not been established. The effects of BUSCOPAN may be potentiated by the concomitant use of other anticholinergic drugs. Studies of concomitant administration of BUSCOPAN with other drugs have not been conducted. Drug compatibility should be monitored closely in patients requiring adjunctive therapy. The safety of BUSCOPAN has not been established for intramuscular (IM) administration. Administration of BUSCOPAN results in heart rate elevation. Heart rate can not be used as a valid indicator of severity of pain for 30 minutes following IV injection.
-
Adverse Reactions:
Transient tachycardia and decreased borborygmal sounds lasted approximately 30 minutes following administration. Transient pupillary dilation may also be observed.
To report suspected adverse reactions, to obtain a Safety Data Sheet (SDS), or for technical assistance, call 1-888-637-4251.
For additional information about adverse drug experience reporting for animal drugs, contact FDA by telephone 1-888-FDA-VETS or online at www.fda.gov/reportanimalae.
- Clinical Pharmacology:
-
Pharmacokinetics:
Following single IV administration of 14C-Buscopan (0.4 mg/kg, side chain labeled) in 3 horses, the major route of elimination of total radioactivity was via urine and feces almost equally. The bulk of the radioactivity (>96%) that represents all 14C-labeled moieties derived from the 14C-Buscopan dose was eliminated (urine and feces) within the first 48 hours post-dosing. The elimination half-life of total plasma 14C was estimated to be approximately 6 hours. Therefore, the elimination half-life of the parent drug in plasma is equal to or shorter than 6 hours. The main radioactive component in the urine, which co-chromatographed with BUSCOPAN using thin layer chromatography (TLC), accounted for approximately 85% of the detected radioactivity.
-
Effectiveness:
A multi-centered, field study was conducted to establish the clinical effectiveness of BUSCOPAN (0.3 mg/kg body weight) for the control of abdominal pain (colic) associated with spasmodic, flatulent and simple impaction colics in horses. A total of 217 cases were randomly assigned to the BUSCOPAN or placebo group; investigators were masked with respect to treatment.
Horses underwent a pretreatment colic examination, with repeated exams at 5, 15, and 30 minutes following test article administration. A total colic score was assigned to each case based on 5 individual criteria (sweating, pawing, head and body movement, kicking, and desire to lie down). A general clinical impression was made at the 30-minute post-treatment evaluation. No further evaluations were performed after 30 minutes.
Quarter Horses, Arabians, and Thoroughbreds accounted for the majority of the cases. Fifty-three percent of the horses were geldings, 39% were mares and 8% were stallions. Body weights ranged from 300-1700 lb (136-772 kg), with a mean of 968 lb (440 kg). Ages ranged between 4 months and 35 years, with an average of 10.6 years.
Total colic scores decreased throughout the 30-minute post-treatment observation period for both BUSCOPAN (N-butylscopolammonium bromide, 20 mg/mL) and placebo. Scores were significantly lower (p≤0.001) for the BUSCOPAN treated horses. Also, 88% of the BUSCOPAN cases were rated as a "success" (excellent, good, or moderate). This was significantly greater (p≤0.0001) than the 42% success rate for the placebo group.
The effectiveness of BUSCOPAN was also supported by the overall improvement in the behavioral attitudes of the horses. A significantly higher percentage of BUSCOPAN treated horses were rated as "alert/calm" at 15 and 30 minutes post-treatment (p≤0.005 and 0.0058, respectively), and a significantly (p≤0.006) greater proportion of placebo treated horses were "nervous/restless." A comparable number of cases for both the BUSCOPAN and placebo groups were classified as "violent" or "drowsy/depressed" at all time points.
Post-treatment heart rates were significantly elevated (p≤0.0001) for BUSCOPAN treated horses at 5 and 15 minutes, compared to the placebo treated horses, as expected due to BUSCOPAN's parasympatholytic effects. By 30 minutes post-treatment, the heart rates for the BUSCOPAN group did not differ significantly from their pre-treatment heart rates. The intensity and frequency of borborygmi in all four abdominal quadrants was decreased for the BUSCOPAN group at most post-treatment evaluation points (p≤0.02).
-
Animal Safety:
Target animal safety was evaluated in several studies, including dose tolerance, target animal safety, hemodynamics, and field safety. There were no signs of toxicity or adverse reactions. The pharmacological effects seen in these studies were consistent with those of anticholinergic drugs (see Clinical Pharmacology section).
In a target animal safety study, BUSCOPAN was evaluated in two phases. In the first phase, the drug was administered intravenously at dosages of 1, 3, and 5 times the recommended level (30, 90 and 150 mg per 100 kg body weight) once daily for three consecutive days. In the second phase it was given at the recommended therapeutic dosage (30 mg per kg body weight) at hourly intervals for three consecutive hours. Horses receiving BUSCOPAN tended to have transient, reduced intensity of auscultated borborygmi, which can be attributed to the intended therapeutic effect of the drug. There was no evidence of gut stasis or colic. No other clinically significant effects were found. Though a transient effect on heart rate was noted in other studies, observations in this study (1-4 hours after drug administration) were made after the primary pharmacological activity for this drug had passed. Neither was there a significant association of dosage with pupil/pupillary light response as has been noted in other studies. Pupillary dilation is an expected pharmacological response to this class of drug (anticholinergic). The lack of a clinical response in this study is also likely related to the longer time after treatment at which the initial observations were made. In any event, this study shows that any effect on pupillary light response is indeed transient. There were no drug associated findings with respect to blood analyses, nor were there any effects evident at necropsy and histopathology.
In a tolerance study, four horses were administered 10 times the recommended BUSCOPAN dosage (300 mg/100 kg) and were examined at 10 minutes and then at 1, 2, 3, 4, 24, and 72 hours post injection. All horses temporarily experienced dilated and fixed pupils at 10 minutes post injection. Normal pupillary light reflex returned in two horses at 4 hours and in the other two horses at 24 hours. Heart rates were increased at 10 and 60 minutes, but had returned to normal at 2 hours. Oral mucous membranes were dry at 10 minutes, but were normal at 2 hours. Gut motility, as judged by auscultated borborygmi, was absent at 10 minutes post injection, but had returned to baseline frequency and intensity by 3 hours in 2 horses and by 4 hours in the remaining horses. There were two incidences of colic, with one horse showing mild colic at 1 hour, and another with mild colic at 11 hours. Both colic episodes were transient and required no medication. There were no drug associated changes in CBC, blood coagulation, or serum chemistry. Neither were there any drug-related gross or histopathological changes.
In a study of hemodynamic parameters, BUSCOPAN decreased right atrial pressure, while cardiac output was maintained. The hemodynamic changes which occurred following BUSCOPAN administration were of little clinical significance, being qualitatively similar to those reported for low doses of other anticholinergics such as atropine.
- Storage:
- How Supplied:
- References:
- SPL UNCLASSIFIED SECTION
- 50 mL Container Label
-
50 mL Display Carton
NDC 0010-3701-01
Busopan® Injectable Solution
(N-butylscopolammonium bromide, 20 mg/mL)
Caution: Federal law restricts this drug to use by or on the order of a licensed veterinarian.
Antispasmodic (spasmolytic) and anticholinergic drug for intravenous use in horses only
Net Contents: 50 mL
Approved by FDA under NADA # 141-228
-
INGREDIENTS AND APPEARANCE
BUSCOPAN
n-butylscopolammonium bromide injectionProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC:0010-3701 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BUTYLSCOPOLAMINE BROMIDE (UNII: 0GH9JX37C8) (BUTYLSCOPOLAMINE - UNII:2Z3E1OF81V) BUTYLSCOPOLAMINE BROMIDE 20 mg in 1 mL Inactive Ingredients Ingredient Name Strength METHYLPARABEN (UNII: A2I8C7HI9T) 1.8 mg in 1 mL PROPYLPARABEN (UNII: Z8IX2SC1OH) 0.2 mg in 1 mL SODIUM CHLORIDE (UNII: 451W47IQ8X) 6.0 mg in 1 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0010-3701-01 1 in 1 CARTON 1 50 mL in 1 VIAL Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NADA NADA141228 06/04/2004 Labeler - Boehringer Ingelheim Animal Health USA Inc. (007134091)