Label: THERASPEC ACNE CLEANSER- benzoyl peroxide soap
-
Contains inactivated NDC Code(s)
NDC Code(s): 52205-003-00 - Packager: Salient Labs, LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated November 16, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
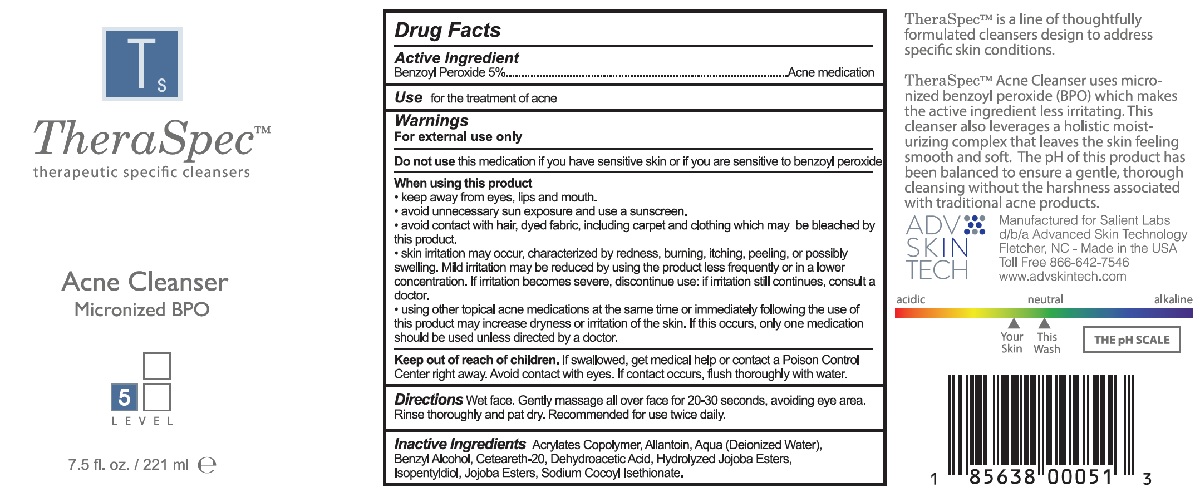
- TheraSpec therapeutic specific cleansers Acne Cleanser Micronized BPO Drug Facts Panel
- Active Ingredient
- PURPOSE
- Use
- Warnings
- DO NOT USE
-
WHEN USING
When using this product
- keep away from eyes, lips and mouth
- avoid unnecessary sun exposure and use a sunscreen
- avoid contact with hair, dyed fabric, including carpet and clothing which may be bleached by this product
- skin irritation may occur, characterized by redness, burning, itching, peeling, or possibly swelling. Mild irritation may be reduced by using the product less frequently or in a lower concentration. lf irritation becomes severe, discontinue use: if irritation still continues, consult a doctor.
- using other topical acne medications at the same time or immediately following the use of this product may increase dryness or irritation of the skin. If this occurs, only one medication should be used unless directed by a doctor.
- KEEP OUT OF REACH OF CHILDREN
- DOSAGE & ADMINISTRATION
- INACTIVE INGREDIENT
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
THERASPEC ACNE CLEANSER
benzoyl peroxide soapProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:52205-003 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZOYL PEROXIDE (UNII: W9WZN9A0GM) (BENZOYL PEROXIDE - UNII:W9WZN9A0GM) BENZOYL PEROXIDE 5 g in 100 mL Inactive Ingredients Ingredient Name Strength BUTYL ACRYLATE/METHYL METHACRYLATE/METHACRYLIC ACID COPOLYMER (18000 MW) (UNII: JZ1374NL9E) HYDROLYZED JOJOBA ESTERS (ACID FORM) (UNII: UDR641JW8W) ALLANTOIN (UNII: 344S277G0Z) WATER (UNII: 059QF0KO0R) SODIUM COCOYL ISETHIONATE (UNII: 518XTE8493) DEHYDROACETIC ACID (UNII: 2KAG279R6R) ISOPENTYLDIOL (UNII: 19NOL5474Q) JOJOBA OIL, RANDOMIZED (UNII: 7F0EV20QYL) BENZYL ALCOHOL (UNII: LKG8494WBH) POLYOXYL 20 CETOSTEARYL ETHER (UNII: YRC528SWUY) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:52205-003-00 221 mL in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 04/18/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part333D 04/18/2019 Labeler - Salient Labs, LLC (130977940) Establishment Name Address ID/FEI Business Operations Pure Source, LLC 080354456 manufacture(52205-003)