Label: EMINENCE RED CURRANT PROTECTIVE MOISTURIZER SPF 40 SUNSCREEN- zinc oxide cream
- NDC Code(s): 15751-3502-1, 15751-3502-2
- Packager: Eminence Organic Skin Care
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated November 29, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
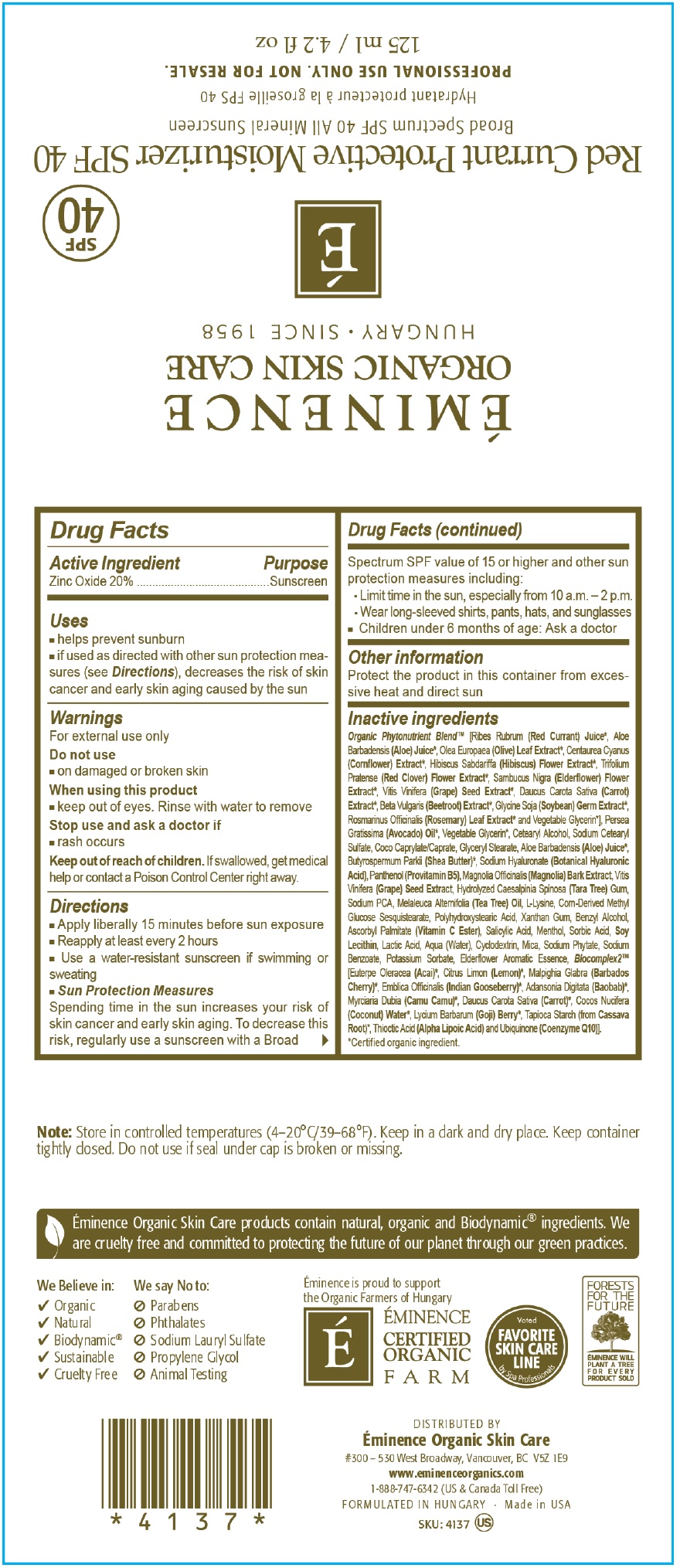
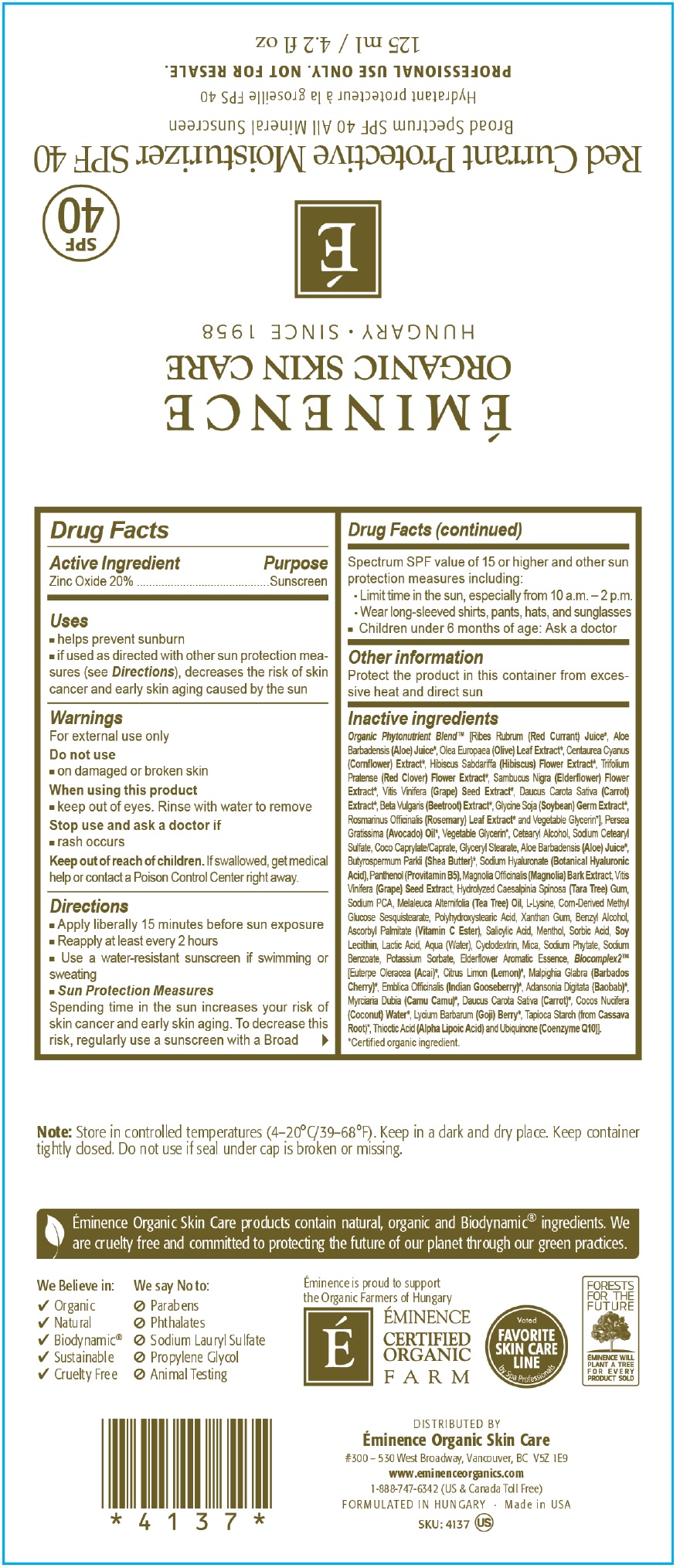
- Drug Facts
- Active Ingredients
- Uses
- Warnings
-
Directions
- Apply liberally 15 minutes before sun exposure
- Reapply at least every 2 hours
- Use a water-resistant sunscreen if swimming or sweating
- Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including: Sun Protection Measures
- Limit time in the sun, especially from 10 a.m. - 2 p.m/
- Wear long sleeved shirts, pants, hats and sunglasses
- Children under 6 months of age: Ask a doctor
- Other information
-
Inactive Ingredients
Organic Phytonutrient Blen [Ribes Rubrum (Red Currant) Juice, Aloe Barbadensis (Aloe) Juice, Olea Europaea (Olive) Leaf Extract*, Centaurea Cyanus (Cornflower) Extract, Hibiscus Sabdariffa (Hibiscus) Sabdariffa (Hibiscus) Flower Extract*, Trifolium Pratense (Red Clover) Flower Extract*, Sambucus Nigra (Elderflower) Flower Extract, Vitis Vinifera (Grape) Seed Extract*, Daucus Carota Sativa (Carrot) Extract*, Beta Vularis (Beet root) Extract*, Glycine Soja (Soybean) Germ Extract*, Rosmarinus Officinalis (Rosemary) Leaf Extract* and Vegetable Glycerin*], Persea Gratissima (Avocado) Oil*, Vegetable Glycerin*, Cetearyl Alcohol, Sodium Cetearyl Sulfate, Coco Caprylate/Caprate, Glyceryl Stearate, Aloe Barbadensis (Aloe) Juice, Butyrospermum Parkii (Shea Butter)*, Sodium Hyaluronate (Botanical Hyaluronic Acid) Panthenol (Provitamin B5), Magnolia Officinalis (Magnolia) Bark Extract, Vitis VInifera (Grape) Seed Extract, Hydrolyzed Caesalpinia Spinosa (Tara Tree) Gum, Sodium PCA, Melaleuca Alternifolia (Tea Tree) Oil, L-Lysine, Corn-Derived Methyl Glucose Sesquistearate, Polyhydroxystearic Acid, Xanthan Gum, Benzyl Alcohol, Ascorbyl Palmitate (Vitamin C Ester), Salicylic Acid, Menthol, Sorbic Acid, Soy Lecithin, Lactic Acid, Aqua (Water), Cyclodextrin, Mica, Sodium Phytate, Sodium Benzoate, Potassium Sorbate, Eiderflower Aromatic Essence, Blocomplex2 (Euterpe Oleracea (Acai)*, Citrus Limon (Lemon)*, Malpighia Glabra (Barbados Cherry)*, Embica Officinalis (Indian Gooseberry)*, Adansonia Digitata (Baobab)*, Myrciaria Dubia (Camu Camu)*, Daucus Carota Sativa (Carrot)*, Cocos Nucifera (Coconut) Water*, Lycium Barbarum (Goji) Berry*, Tapioca Starch (from Cassava Root)*, Thioctic Acid (Alpha Lipoic Acid) and Ubiquinone (Coenzyme Q10)].
Certified organic ingredient
- Note
- Package Labeling:60mL
- Package Labeling:125mL
-
INGREDIENTS AND APPEARANCE
EMINENCE RED CURRANT PROTECTIVE MOISTURIZER SPF 40 SUNSCREEN
zinc oxide creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:15751-3502 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 200 mg in 1 mL Inactive Ingredients Ingredient Name Strength RED CURRANT (UNII: Q5YG47WU3D) ALOE VERA LEAF (UNII: ZY81Z83H0X) OLEA EUROPAEA LEAF (UNII: MJ95C3OH47) HIBISCUS SABDARIFFA FLOWER (UNII: 45TGG6IU6M) TRIFOLIUM PRATENSE FLOWER (UNII: 4JS0838828) SAMBUCUS NIGRA FLOWER (UNII: 07V4DX094T) VITIS VINIFERA SEED (UNII: C34U15ICXA) CARROT (UNII: L56Z1JK48B) BEET JUICE (UNII: IOZ32L9H3O) SOYBEAN GERM (UNII: JBW2VHD14M) ROSEMARY (UNII: IJ67X351P9) GLYCERIN (UNII: PDC6A3C0OX) AVOCADO OIL (UNII: 6VNO72PFC1) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) SODIUM CETOSTEARYL SULFATE (UNII: 7ZBS06BH4B) COCOYL CAPRYLOCAPRATE (UNII: 8D9H4QU99H) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) SHEA BUTTER (UNII: K49155WL9Y) HYALURONATE SODIUM (UNII: YSE9PPT4TH) PANTHENOL (UNII: WV9CM0O67Z) MAGNOLIA OFFICINALIS BARK (UNII: 5M609NV974) SODIUM PYRROLIDONE CARBOXYLATE (UNII: 469OTG57A2) TEA TREE OIL (UNII: VIF565UC2G) LYSINE (UNII: K3Z4F929H6) XANTHAN GUM (UNII: TTV12P4NEE) BENZYL ALCOHOL (UNII: LKG8494WBH) ASCORBYL PALMITATE (UNII: QN83US2B0N) SALICYLIC ACID (UNII: O414PZ4LPZ) MENTHOL (UNII: L7T10EIP3A) SORBIC ACID (UNII: X045WJ989B) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) LACTIC ACID (UNII: 33X04XA5AT) WATER (UNII: 059QF0KO0R) MICA (UNII: V8A1AW0880) PHYTATE SODIUM (UNII: 88496G1ERL) SODIUM BENZOATE (UNII: OJ245FE5EU) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) ACAI (UNII: 46AM2VJ0AW) LEMON (UNII: 24RS0A988O) COCONUT WATER (UNII: 267F5Y81NT) STARCH, TAPIOCA (UNII: 24SC3U704I) THIOCTIC ACID (UNII: 73Y7P0K73Y) UBIDECARENONE (UNII: EJ27X76M46) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:15751-3502-1 1 in 1 BOX 01/01/2021 1 60 mL in 1 JAR; Type 0: Not a Combination Product 2 NDC:15751-3502-2 1 in 1 BOX 01/01/2021 2 125 mL in 1 JAR; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 01/01/2021 Labeler - Eminence Organic Skin Care (205753317)