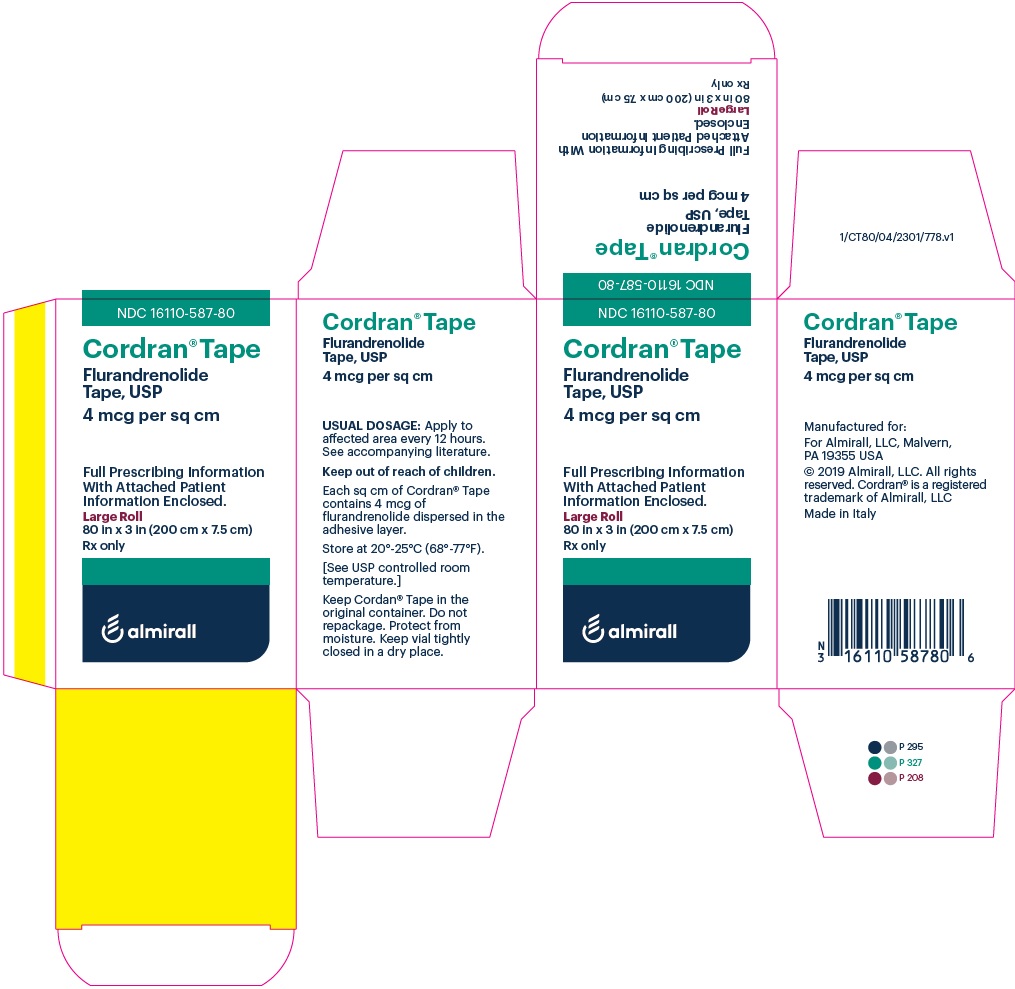
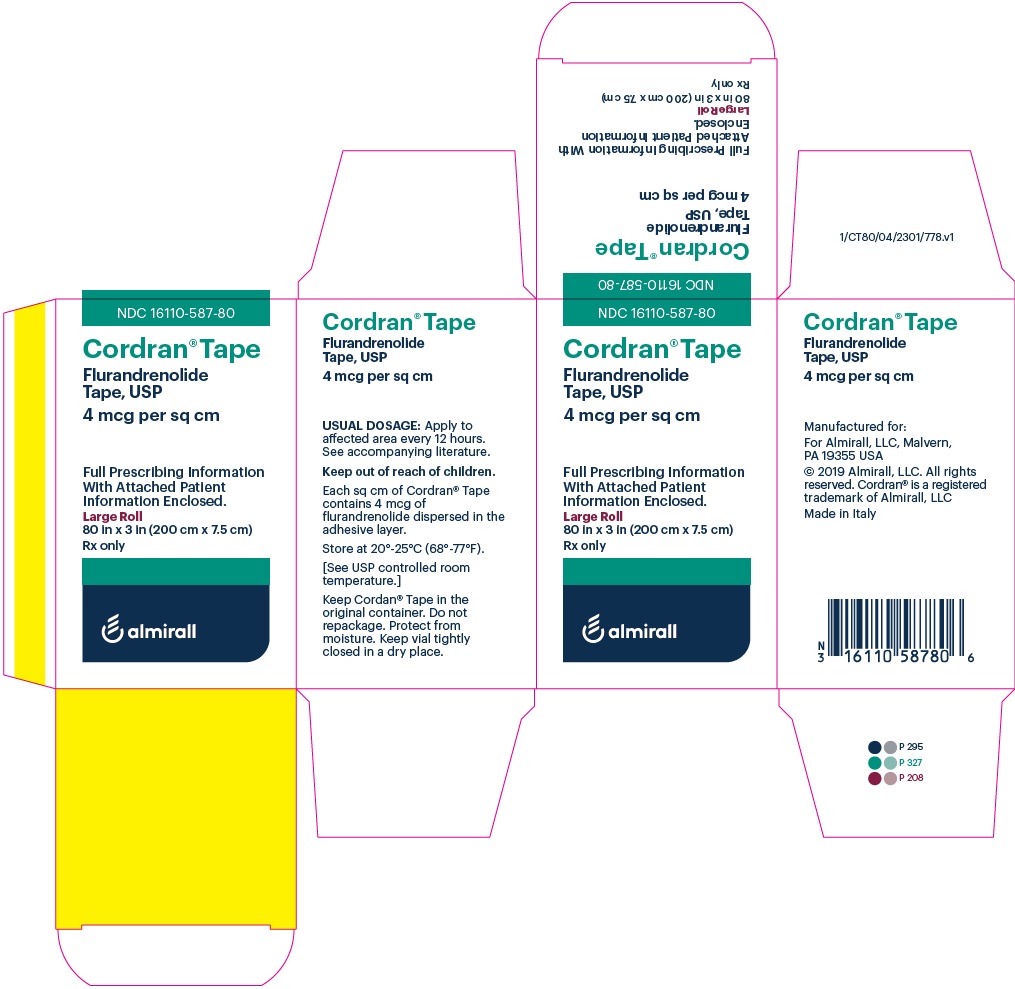
Label: CORDRAN- flurandrenolide tape
- NDC Code(s): 16110-587-24, 16110-587-80
- Packager: Almirall, LLC
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated November 10, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
Cordran Tape (Flurandrenolide Tape, USP) is a transparent, inconspicuous, plastic surgical tape. It contains Cordran (Flurandrenolide, USP), a potent corticosteroid for topical use. Flurandrenolide occurs as white to off-white, fluffy crystalline powder and is odorless. Flurandrenolide is practically insoluble in water and in ether. One gram dissolves in 72 mL of alcohol and in 10 mL of chloroform. The molecular weight of flurandrenolide is 436.52.
The chemical name of flurandrenolide is Pregn-4-ene-3,20-dione, 6-fluoro-11,21 dihydroxy-16,17-[(1-methylethylidene)bis(oxy)]-, (6α, 11ß, 16α)-; its empirical formula is C24H33FO6. The structural formula is as follows:
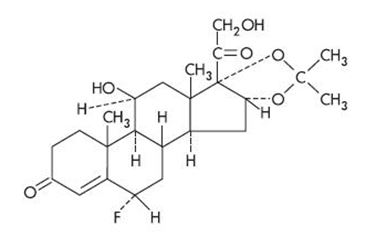
Each square centimeter contains 4 mcg (0.00916 μmol) flurandrenolide uniformly distributed in the adhesive layer. The tape is made of a thin, matte-finish polyethylene film that is slightly elastic and highly flexible.
The adhesive is a synthetic copolymer of acrylate ester and acrylic acid that is free from substances of plant origin. The pressure-sensitive adhesive surface is covered with a protective paper liner to permit handling and trimming before application.
-
CLINICAL PHARMACOLOGY
Cordran is primarily effective because of its anti-inflammatory, antipruritic, and vasoconstrictive actions.
The mechanism of the anti-inflammatory effect of topical corticosteroids is not completely understood. Various laboratory methods, including vasoconstrictor assays, are used to compare and predict potencies and/or clinical efficacies of the topical corticosteroids. There is some evidence to suggest that a recognizable correlation exists between vasoconstrictor potency and therapeutic efficacy in man. Corticosteroids with anti-inflammatory activity may stabilize cellular and lysosomal membranes. There is also the suggestion that the effect on the membranes of lysosomes prevents the release of proteolytic enzymes and, thus, plays a part in reducing inflammation.
The tape serves as both a vehicle and an occlusive dressing. Retention of insensible perspiration by the tape results in hydration of the stratum corneum and improved diffusion of the medication. The skin is protected from scratching, rubbing, desiccation, and chemical irritation. The tape acts as a mechanical splint to fissured skin. Since it prevents removal of the medication by washing or the rubbing action of clothing, the tape formulation provides a sustained action.
Pharmacokinetics—The extent of percutaneous absorption of topical corticosteroids is determined by many factors, including the vehicle, the integrity of the epidermal barrier, and the use of occlusive dressings.
Topical corticosteroids can be absorbed from normal intact skin. Inflammation and/or other disease processes in the skin increase percutaneous absorption. Occlusive dressings substantially increase the percutaneous absorption of topical corticosteroids. Thus, occlusive dressings may be a valuable therapeutic adjunct for treatment of resistant dermatoses (see DOSAGE AND ADMINISTRATION).
Once absorbed through the skin, topical corticosteroids are handled through pharmacokinetic pathways similar to those of systemically administered corticosteroids. Corticosteroids are bound to plasma proteins in varying degrees. They are metabolized primarily in the liver and then excreted in the kidneys. Some of the topical corticosteroids and their metabolites are also excreted into the bile.
- INDICATIONS AND USAGE
- CONTRAINDICATIONS
-
PRECAUTIONS
General -
Systemic absorption of topical corticosteroids has produced reversible hypothalamic-pituitary-adrenal (HPA) axis suppression, manifestations of Cushing’s syndrome, hyperglycemia, and glucosuria in some patients.
Conditions that augment systemic absorption include application of the more potent steroids, use over large surface areas, prolonged use, and the addition of occlusive dressings.
Therefore, patients receiving a large dose of a potent topical steroid applied to a large surface area or under an occlusive dressing should be evaluated periodically for evidence of HPA axis suppression by using urinary-free cortisol and ACTH stimulation tests. If HPA axis suppression is noted, an attempt should be made to withdraw the drug, to reduce the frequency of application, or to substitute a less potent steroid.
Recovery of HPA axis function is generally prompt and complete on discontinuation of the drug. Infrequently, signs and symptoms of steroid withdrawal may occur, so that supplemental systemic corticosteroids are required.
Pediatric patients may absorb proportionately larger amounts of topical corticosteroids and thus be more susceptible to systemic toxicity (see Pediatric Use under PRECAUTIONS).
If irritation develops, topical corticosteroids should be discontinued and appropriate therapy instituted.
In the presence of dermatologic infections, the use of an appropriate antifungal or antibacterial agent should be instituted. If a favorable response does not occur promptly, Cordran should be discontinued until the infection has been adequately controlled.
Information for the Patient -
Patients using topical corticosteroids should receive the following information and instructions:
- This medication is to be used as directed by the physician. It is for external use only. Avoid contact with the eyes.
- Patients should be advised not to use this medication for any disorder other than that for which it was prescribed.
- The treated skin area should not be bandaged or otherwise covered or wrapped in order to be occlusive unless the patient is directed to do so by the physician.
- Patients should report any signs of local adverse reactions, especially under occlusive dressing.
- Parents of pediatric patients should be advised not to use tight-fitting diapers or plastic pants on a patient being treated in the diaper area, because these garments may constitute occlusive dressings.
Laboratory Tests -
The following tests may be helpful in evaluating the HPA axis suppression:
Urinary-free cortisol test
ACTH stimulation testCarcinogenesis, Mutagenesis, and Impairment of Fertility -
Long-term animal studies have not been performed to evaluate the carcinogenic potential or the effect on fertility of topical corticosteroids.
Studies to determine mutagenicity with prednisolone and hydrocortisone have revealed negative results.
Usage in Pregnancy -
Corticosteroids are generally teratogenic in laboratory animals when administered systemically at relatively low dosage levels. The more potent corticosteroids have been shown to be teratogenic after dermal application in laboratory animals. There are no adequate and well-controlled studies in pregnant women on teratogenic effects from topically applied corticosteroids. Therefore, topical corticosteroids should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. Drugs of this class should not be used extensively for pregnant patients or in large amounts or for prolonged periods of time.
Nursing Mothers -
It is not known whether topical administration of corticosteroids could result in sufficient systemic absorption to produce detectable quantities in breast milk. Systemically administered corticosteroids are secreted into breast milk in quantities not likely to have a deleterious effect on the infant. Nevertheless, caution should be exercised when topical corticosteroids are administered to a nursing woman.
Pediatric patients may demonstrate greater susceptibility to topical-corticosteroid-induced HPA axis suppression and Cushing’s syndrome than do mature patients because of a larger skin surface area to body weight ratio.
Hypothalamic-pituitary-adrenal (HPA) axis suppression, Cushing’s syndrome, and intracranial hypertension have been reported in pediatric patients receiving topical corticosteroids. Manifestations of adrenal suppression in pediatric patients include linear growth retardation, delayed weight gain, low plasma-cortisol levels, and absence of response to ACTH stimulation. Manifestations of intracranial hypertension include bulging fontanelles, headaches, and bilateral papilledema.
Administration of topical corticosteroids to pediatric patients should be limited to the least amount compatible with an effective therapeutic regimen. Chronic corticosteroid therapy may interfere with the growth and development of pediatric patients.
-
ADVERSE REACTIONS
The following local adverse reactions are reported infrequently with topical corticosteroids but may occur more frequently with the use of occlusive dressings. These reactions are listed in an approximate decreasing order of occurrence: burning, itching, irritation, dryness, folliculitis, hypertrichosis, acneiform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis.
The following may occur more frequently with occlusive dressings: maceration of the skin, secondary infection, skin atrophy, striae, miliaria.
-
OVERDOSAGE
Topically applied corticosteroids can be absorbed in sufficient amounts to produce systemic effects (see PRECAUTIONS).
-
DOSAGE AND ADMINISTRATION
Occlusive dressings may be used for the management of psoriasis or recalcitrant conditions.
If an infection develops, the use of Cordran Tape and other occlusive dressings should be discontinued and appropriate antimicrobial therapy instituted.
Replacement of the tape every 12 hours produces the lowest incidence of adverse reactions, but it may be left in place for 24 hours if it is well tolerated and adheres satisfactorily. When necessary, the tape may be used at night only and removed during the day.
If ends of the tape loosen prematurely, they may be trimmed off and replaced with fresh tape.
The directions given below are included for the patient to follow unless otherwise instructed by the physician.
-
APPLICATION OF CORDRAN TAPE
IMPORTANT: Skin should be clean and dry before tape is applied. Tape should always be cut, never torn. DIRECTIONS FOR USE:
- Prepare skin as directed by your physician or as follows: Gently clean the area to be covered to remove scales, crusts, dried exudates, and any previously used ointments or creams. A germicidal soap or cleanser should be used to prevent the development of odor under the tape. Shave or clip the hair in the treatment area to allow both good contact with the skin and comfortable removal. If shower or tub bath is to be taken, it should be completed before the tape is applied. The skin should be dry before application of the tape.
- Remove tape from flip-top vial and cut a piece slightly larger than area to be covered. Round off corners.
- Return remainder of tape roll to vial and tightly close the lid.
- Pull white paper from transparent tape. Be careful that tape does not stick to itself.
- Apply tape, keeping skin smooth; press tape into place.
Keep Cordran Tape in the original container. Do not repackage. Protect from moisture. Keep vial tightly closed in a dry place.
REPLACEMENT OF TAPE:
Unless instructed otherwise by your physician, replace tape after 12 hours. Cleanse skin and allow it to dry for 1 hour before applying new tape.
IF IRRITATION OR INFECTION DEVELOPS, REMOVE TAPE AND CONSULT PHYSICIAN.
- HOW SUPPLIED
-
REFERENCES
Bard JW: Flurandrenolide tape in the treatment of lichen simplex chronicus. J Ky Med Assoc 1969;67:668.
Baxter DL, Stoughton RB: Mitotic index of psoriatic lesions treated with anthralin, glucocorticosteroid and occlusion only. J Invest Dermatol 1970;54:410.
Compilation of clinical reports on Cordran Tape received by Eli Lilly and Company.
Halprin KM, Fukui K, Ohkawara A: Flurandrenolone (Cordran) tape and carbohydrate metabolizing enzymes. Arch Dermatol 1969;100:336.
Labow TA, Eisert J, Sanders SL: Flurandrenolide tape in treatment of psoriasis. NY State J Med 1969;69:3138.
Ronchese F: Flurandrenolone tape therapy. RI Med J 1969;52:389.
Sellers FM: Investigative study of flurandrenolone tape in a series of ambulatory outpatients. J Indiana State Med Assoc 1970;63:34.
Weiner MA: Flurandrenolone tape, a new preparation for occlusive therapy, J Invest Dermatol 1966; 47:63.
Manufactured for:
Almirall, LLC
101 Lindenwood Drive, Suite 400, Malvern, PA 19355
© 2019 Almirall, LLC. All rights reserved.
Cordran® is a registered trademark of Almirall, LLC.
Content Updated: July 2019
-
For the Patient
Cordran® Tape
Flurandrenolide Tape, USP
DIRECTIONS FOR USE
APPLICATION OF MEDICATED TRANSPARENT TAPE
Rx only
IMPORTANT: Skin should be clean and dry before tape is applied. Tape should always be cut, never torn. - Prepare skin as directed by your physician or as follows: Gently clean the area to be covered to remove scales, crusts, dried exudates, and any previously used ointments or creams. A germicidal soap or cleanser should be used to prevent the development of odor under the tape. Shave or clip the hair in the treatment area to allow both good contact with the skin and comfortable removal. If a shower or tub bath is to be taken, it should be completed before the tape is applied. The skin should be dry before application of the tape.
- Remove tape from flip-top vial and cut a piece slightly larger than area to be covered. Round off corners.
- Return remainder of tape roll to vial and tightly close the lid.
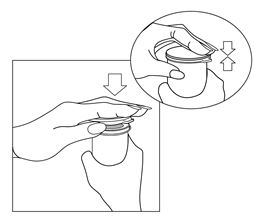
- Pull white paper backing from transparent tape. Be careful that tape does not stick to itself.
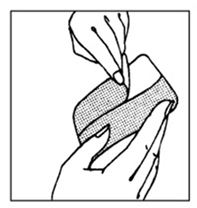
- Apply tape, keeping skin smooth; press tape into place.
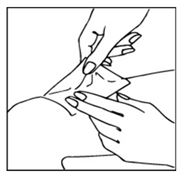
REPLACEMENT OF TAPE
Unless instructed otherwise by your physician, replace tape after 12 hours. Cleanse skin and allow it to dry for 1 hour before applying new tape.
DISPENSING THE TAPE
To use the roll as a dispenser, pull the tape as illustrated.
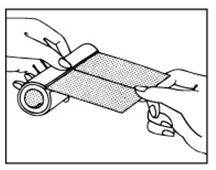
IF IRRITATION OR INFECTION DEVELOPS, REMOVE TAPE AND CONSULT PHYSICIAN.
Store at 20 - 25℃ (68 - 77℉). [See USP controlled room temperature.]
Keep Cordran Tape in the original container. Do not repackage. Protect from moisture. Keep vial tightly closed in a dry place.
Keep out of reach of children.
Rx only
Manufactured for:
Almirall, LLC
101 Lindenwood Drive, Suite 400, Malvern, PA 19355
© 2019 Almirall, LLC. All rights reserved.
Cordran® is a registered trademark of Almirall, LLC.
almirall
Content Updated: July 2019
- PRINCIPAL DISPLAY PANEL - NDC: 16110-587-80 - Carton Label
- PRINCIPAL DISPLAY PANEL - NDC: 16110-587-80 - Vial Label
-
INGREDIENTS AND APPEARANCE
CORDRAN
flurandrenolide tapeProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:16110-587 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Flurandrenolide (UNII: 8EUL29XUQT) (Flurandrenolide - UNII:8EUL29XUQT) Flurandrenolide 4 ug in 1 cm2 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:16110-587-24 1 in 1 Carton 09/10/2019 1 450 cm2 in 1 Bottle; Type 0: Not a Combination Product 2 NDC:16110-587-80 1 in 1 Carton 09/10/2019 2 1500 cm2 in 1 Bottle; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA016455 09/24/2018 Labeler - Almirall, LLC (605425912)